Leaderboard Ad
Year: 2014
Grants:
Bleeding Disorders Conference
Clinical Research
Real-world dosing of factor in hemophilia A patients
Year: 2014
Grants:
Bleeding Disorders Conference
Clinical Research
Objective:
To analyse real world FVIII dosing and treatment interval patterns in patients with haemophilia A. A secondary objective was to compare the observed dosing patterns with the dosing regimens for rFVIII and rFVIIIFc evaluated in clinical studies.
Methods:
A retrospective analysis was conducted using aggregate Specialty Pharmacy Provider (SPP) records from Nov 2013 through Mar 2014. SPP data included 63 different attributes for each prescription, including trade name, National Drug Code (NDC), drug quantity shipped, prescribed infusion dose, days supplied, and dose frequency. Patients were considered eligible for the analysis if they received a shipment of any FVIII product. Patients were excluded from the analysis if they were being treated episodically, for immune tolerance induction, or their pharmacy records did not specify a prescribed infusion dose. Patients with missing or extremely abnormal weights were also excluded. The patient’s weekly consumption was calculated for each shipment record by multiplying the prescribed infusion dose by the dose frequency and dividing the product by the patient’s weight, resulting in the patient’s average weekly prescribed dose (IU/kg/week). Patients were also categorized according to their dosing interval.
Summary:
The analysis included 520 hemophilia A patients with a median age of 18 (range: 1-77) and median weight of 63.5 kg (range: 8-161 kg). Pharmacy dispensing records represented 227 distinct prescribers across 43 states. FVIII therapies evaluated included Advate®, Recombinate®, Helixate® FS, Kogenate®, Hemofil and Xyntha®. The average weekly consumption across all therapies was 108.0 IU/kg/week (95% CI, 104.6-111.5). Dosing frequency ranged from once-daily to once-weekly with three times/week and every other day as the most common dosing intervals, representing 81.3% of patient records. For patients infusing thrice-weekly, the average infusion dose was 35.1 IU/Kg. Only 15.4% of the population was infusing ≤ two times per week. Clinical trials for Advate report weekly consumption of 110.3 IU/kg (31.4 IU/kg administered QOD). Two prophylactic regimens were evaluated for rFVIIIFc in A-LONG. In the last 3 months of this study, the median weekly consumption was 77.7 IU/kg for the individualized prophylaxis and the median weekly dose of 65.5 IU/kg for the weekly prophylaxis regimen.
Conclusions:
Pharmacy dispensing records support the clinical trial dosing intervals of rFVIII products currently requiring every other day or thrice-weekly dosing; however, real-world dosing (IU/kg/week) may be greater. This may result in unpredictability for payers who are responsible for healthcare budgets.
Does Quality of Life improve with successful immune tolerance induction? An illustrative case report.
Year: 2014
Grants:
Bleeding Disorders Conference
Psychosocial Issues
Objective:
Having a family member with a chronic disease often increases the burden in the family with more hospital visits, treatment administration, and increased expenses. Management of hemophilia patients with inhibitors can be very complex and challenging. Health-related quality of life (HRQoL) has become a recent focus of research in hemophilia. Data on the HRQoL of congenital hemophilia patients with inhibitors and their caregivers is limited.
Methods:
We report a case study of a 36 year old male diagnosed as a neonate with hemophilia A, who, at age 6 months, developed a high titer inhibitor. His titer levels ranged from 99 BU/ml to in the thousands. Throughout a 30 year period, he experienced frequent bleeding episodes with several severe bleeds requiring extended hospitalization and intensive care management. He completed a majority of his schoolwork from a hospital bed and was unable to hold a steady job. He used factor VIII (FVIII) bypassing agents on demand to manage the bleeding episodes. As an adult, immune tolerance induction (ITI) failed with recombinant FVIII (rFVIII). QoL was poor for this husband and father of two children due to extremely limited mobility and inability to provide household income. He needed double knee replacement but insurance coverage for the surgery was denied due to the presence of the inhibitor. ITI therapy was switched to human plasma-derived FVIII with double viral inactivation (Koate-DVI) 10,000 units per day with successful tolerization in 8 months; his inhibitor was undetectable. He is currently on a prophylactic regimen of 3000 units twice a week, has not experienced any adverse events, and has had no major bleeds and only one minor bleed in 4 years.
Summary:
Successful immune tolerance induction (ITI) was achieved in a 32 year old adult male with hemophilia A with daily self-infusion of human FVIII therapy containing naturally occurring von Willebrand factor. This patient’s QoL has significantly improved since initiation of a home-based ITI protocol with Koate-DVI. He has been able to have double knee replacement surgery with major improvement in mobility and started his own successful national business.
Conclusions:
Advances in therapies continue to improve the longevity and quality of life of patients with hemophilia A. Increased or maintained HRQoL are essential goals in health care among patients with a chronic disease. This case study demonstrates the importance of well-disciplined ITI therapy with a human plasma FVIII product for hemophilia A patients with inhibitors.
Retrospective Database Analysis of the Prevalence of Cardiovascular Comorbidities in a US Patient Population with Hemophilia A: Confirmation of Findings
Year: 2014
Grants:
Bleeding Disorders Conference
Clinical Research
Objective:
A previous retrospective study of the MarketScan® claims database reported increased prevalence and earlier onset of cardiovascular (CV) comorbidities in patients with hemophilia A compared with patients without hemophilia. Our study was designed to confirm these findings in a second population of male patients with hemophilia A in the United States.
Methods:
Male patients with hemophilia A and continuous insurance coverage were identified by ICD-9-CM code 286.0 using the PharMetrics LifeLink claims database (IMS Health) of patient records from January 1, 2008 to December 31, 2011. Patients with hemophilia A were matched 1:3 with controls for sex, age, plan type, geographic region, and eligibility months in the study period. The prevalence of CV comorbidities (identified by ICD-9- CM codes) was compared between matched cohorts. Statistical significance was calculated using Fisher’s exact test.
Summary:
Overall, 1050 patients were included in the hemophilia A cohort and 3150 in the control cohort (Table). Prevalence of hemorrhagic stroke, ischemic stroke, coronary artery disease, myocardial infarction, hypertension, hyperlipidemia, arterial thrombosis, and venous thrombosis was significantly higher in the hemophilia A cohort (all P≤0.016). Increased prevalence of CV comorbidities was consistent across most age groups, and patients with hemophilia A experienced CV comorbidities at an earlier age than those without hemophilia.
Table: Cardiovascular comorbidities in patients with hemophilia A
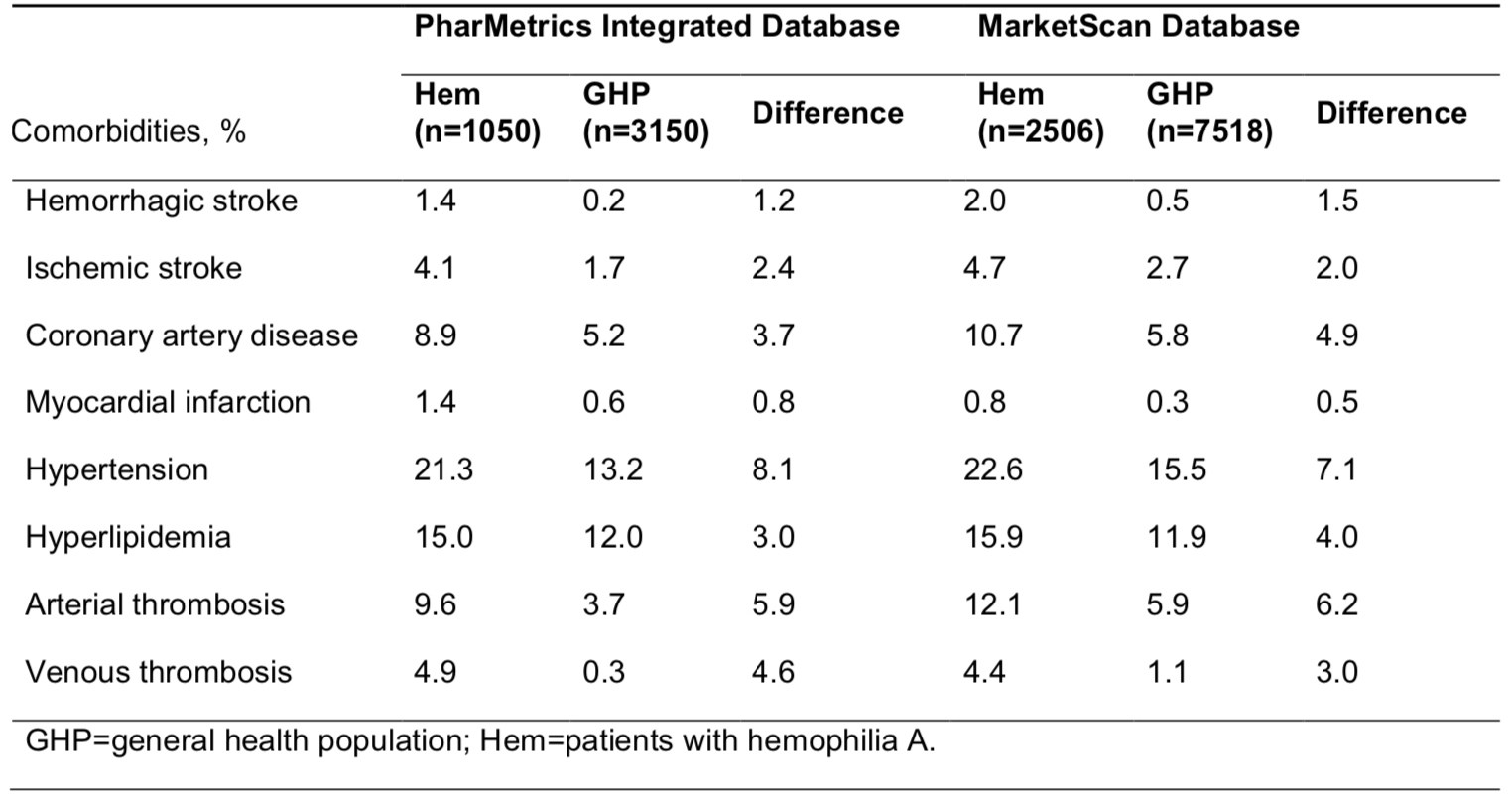
Conclusions:
This second retrospective study of claims databases confirmed an increased prevalence and earlier onset of CV comorbidities in patients with hemophilia A. These findings support increased screening in patients with hemophilia for CV comorbidities at an earlier age than recommended for the general population.
Hemophilia Genotyping Results from the My Life, Our Future Project
Year: 2014
Grants:
Bleeding Disorders Conference
Clinical Research
Objective:
My Life, Our Future (MLOF) is a national project directed by a partnership formed to: 1) conduct wide-scale genetic testing of the U.S. hemophilia community, thereby increasing the rate of patient testing above the currently estimated 20% and allowing carrier detection; 2) establish a repository of associated samples and data to support scientific discovery and treatment advances including informing inhibitor risk and disease severity.
Methods:
A multi-sector partnership was formed to make hemophilia genotype analysis available to the U.S. community. The National Hemophilia Foundation (NHF) educates consumers and supports recruitment. The American Thrombosis and Hemostasis Network (ATHN) provides hemophilia treatment center (HTC) provider education, a secure infrastructure for data collection, and point of access for research proposals. HTCs enroll patients, obtain samples and provide clinical results to patients. Puget Sound Blood Center (PSBC) serves as the central genotyping laboratory and sample repository. Biogen Idec provides scientific collaboration and initiative support.
A pilot study involving 11 HTCs was successfully completed in 2013, and patients continue to be enrolled at those and additional sites. Genotyping is performed through an initial screen by Next Generation sequencing of extracted DNA using a molecular inversion probe-based capture strategy. FVIII and FIX mutations are confirmed in the CLIA-certified PSBC hemophilia genomics laboratory using a separate DNA sample. A clinical laboratory report is returned to the HTC and results transmitted into the ATHN Clinical Manager database accessible only to the patient’s HTC providers. For patients who give informed consent, coded data and samples are stored in a research repository, which can be linked to coded clinical data from the ATHNdataset for future research applications. NHF’s national and local chapter educational programs increased awareness and educated families about MLOF.
Summary:
As of June 13, 2014, 25 HTCs were enrolling patients and 865 patients were enrolled. Of those patients, 168 opted for clinical genotyping only and 697 also gave informed consent to have data and samples entered into the research repository. Mutation analysis has been completed in 707 patients, including 354, 151 and 202 with severe, moderate and mild haemophilia respectively. By comparison to available hemophilia A and B databases, 61 novel mutations have been identified in 68 patients. Lessons learned from the initial stages of the program’s rollout helped us to improve our approach to recruitment and education about the importance of genotyping and research in hemophilia.
Conclusions:
My Life, Our Future is a novel partnership to address unmet needs for hemophilia genotyping services and research. Expanding participation in the program will increase clinical genotyping for patients with hemophilia, increase knowledge of FVIII and FIX mutations present in the U.S. hemophilia population, and provide a robust research repository for future scientific discovery.
SPINART Trial 3-Year Results With Bayer’s Sucrose-Formulated Recombinant Factor VIII: Improved Joint Function and Health-Related Quality of Life in Adults Using Prophylaxis
Year: 2014
Grants:
Bleeding Disorders Conference
Clinical Research
Objective:
Joint status and health-related quality of life (HRQoL) were assessed as part of the 3-year SPINART study, which compared routine prophylaxis versus on-demand treatment in adults with severe hemophilia A. We report SPINART joint outcome results obtained using the Colorado Adult Joint Assessment Scale (CAJAS) and HRQoL data from Haemo-QoL-A assessments.
Methods:
The open-label, randomized, controlled, parallel-group, multinational SPINART study enrolled male patients aged 12–50 years with severe hemophilia A who had ≥150 exposure days to any factor VIII (FVIII) product, no inhibitors, no prophylaxis for >12 consecutive months in the past 5 years, and 6–24 documented bleeding events or treatments in the previous 6 months. All patients were treated with Bayer’s sucrose-formulated recombinant FVIII (rFVIII-FS), either on demand or as prophylaxis (25 IU/kg 3 times weekly, with dose escalation of 5 IU/kg permitted once per year). CAJAS assessments were performed at baseline and years 1, 2, and 3. The physiotherapists performing CAJAS assessments were blinded to patient treatment assignment, bleeding history, and previous joint assessment data. Change from baseline to year 3 in CAJAS total score was prespecified as the second of 2 secondary endpoints; higher CAJAS scores indicate worse joint function. Haemo-QoL-A was completed at baseline, month 6, and years 1, 2, and 3; higher Haemo- QoL-A scores indicate better HRQoL. Between-group comparison was made using constrained longitudinal data analysis. Data are presented for the intent-to-treat (ITT) population.
Summary:
84 patients (42 prophylaxis, 42 on demand) comprised the ITT population; Haemo-QoL-A data were available for 41 and 42 patients, respectively. For CAJAS total score, least squares (LS) mean change from baseline to year 3 was 0.63 for on demand and –0.31 for prophylaxis (LS mean difference, –0.94; 95% CI, –1.61 to –0.26; P=0.0072). LS mean change in CAJAS total score for the on-demand and prophylaxis groups was 0.19 and –0.46 at year 1 and 0.34 and –0.57 at year 2, respectively. For Haemo-QoL-A total score, LS mean change from baseline to year 3 was –6.00 for on demand and 3.98 for prophylaxis (LS mean difference, 9.98; 95% CI, 3.42 to 16.54).
Conclusions:
In adults with severe hemophilia A, joint function and HRQoL improved continuously over 3 years with prophylaxis compared with on-demand use.
Dosing Routines and Bleeding Rates Before and Following Treatment with rFVIIIFc in the A-LONG Clinical Study
Year: 2014
Grants:
Bleeding Disorders Conference
Clinical Research
Safety of BAX 855, a Polyethylene Glycol (PEG) Conjugated Full-Length Recombinant Factor VIII Product
Year: 2014
Grants:
Bleeding Disorders Conference
Clinical Research
Objectives:
Biopharmaceuticals are an emerging branch of therapeutic agents. Their short half-life, rapid elimination and ability to induce a specific immune response, however, may impair their applicability. These disadvantages have been overcome by chemical modification with polyethylene glycol (PEG), which has enhanced the PK and safety of several marketed proteins since the 1990s. PEGylation uses metabolically stable PEG polymers, often with a molecular size of 5-60 kDa.
PEGylated rFVIII candidates include PEG-protein-conjugates with a minimal amount of PEG attached to the protein. Baxter and Nektar are developing BAX 855, a PEGylated full-length recombinant (r) FVIII based on the FVIII molecule used for Baxter’s licensed rFVIII (ADVATE). Due to the high potency of FVIII, the absolute amount of conjugated PEG applied with PEG-FVIII is within the range of μg per kg body weight and week. PEGylation was optimized to retain functionality of the FVIII molecule and improve its pharmacokinetic properties.
PEGs ≤20 kDa are rapidly cleared mainly via the kidneys and excreted into urine. Over time, the protein portion of the PEG-FVIII conjugate is degraded by proteolysis leaving a PEG portion which is rapidly eliminated.
Methods:
Preclinical safety, toxicokinetics and formation of anti-product antibodies were assessed in rats dosed intravenously at 350 or 700U/kg BAX 855 every other day, and in macaques receiving 150, 350 or 700U/kg BAX 855 every five days, for 28 days.
Like other non- degradable entities, physiological clearance mechanisms of PEG may include liver macrophage uptake. Clearance by macrophages in mammals has been reported to cause vacuolization at high cumulative doses. Generally, vacuoles were shown to consistently resolve over time, with no cellular damage, inflammation at the vacuolization site or functional deficits of affected tissues, and are therefore regarded to not affect the safety of PEGylated therapeutics.
Summary:
No systemic adverse effects or vacuolizations were observed after 28-day intravenous administration with BAX 855. Therefore, 700 U/kg was considered the no observed adverse effect level in these studies.
Conclusions:
This favorable safety profile provides the basis for proceeding with human trials.
Prevalence of Depression in US Patients with Hemophilia A Compared with a General Medical Population: A Retrospective Database Analysis
Year: 2014
Grants:
Bleeding Disorders Conference
Psychosocial Issues
Objective:
Prevalence of clinical depression in persons with hemophilia (PWH) has been reported to be from 0%–50%. Most papers studied fairly small numbers of PWH; many had no controls and used instruments not validated for depression. One recent paper, using the Patient Health Questionnaire-9, a validated instrument for depression, reported a prevalence of 37% in 41 adult PWH. Our objective was to determine the prevalence of depression in PWH in the United States.
Methods:
Using the MarketScan® Commercial and Medical Research databases, we compared depression prevalence in 2506 PWH and 7518 controls. Male patients with hemophilia A were identified using an International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) code (ICD-9-CM 286.0) and were matched (based on age, eligibility months in the study, region, and health plan type) in a 1:3 ratio with controls (no hemophilia diagnosis). Evidence of depression was determined using ICD-9-CM 296.20– 296.26, 296.30–296.36, and 311 codes. Chi-square tests were used to compare frequencies.
Summary:
PWH had a statistically significant increase in depression prevalence overall and in age groups 50–59 years and 60–69 years and a numerical increase in all other age groups except 18–29 years (Table). The delta between PWH and controls steadily increased between ages 30 and 69 years.
Table. Patients with Major Depressive Disorder
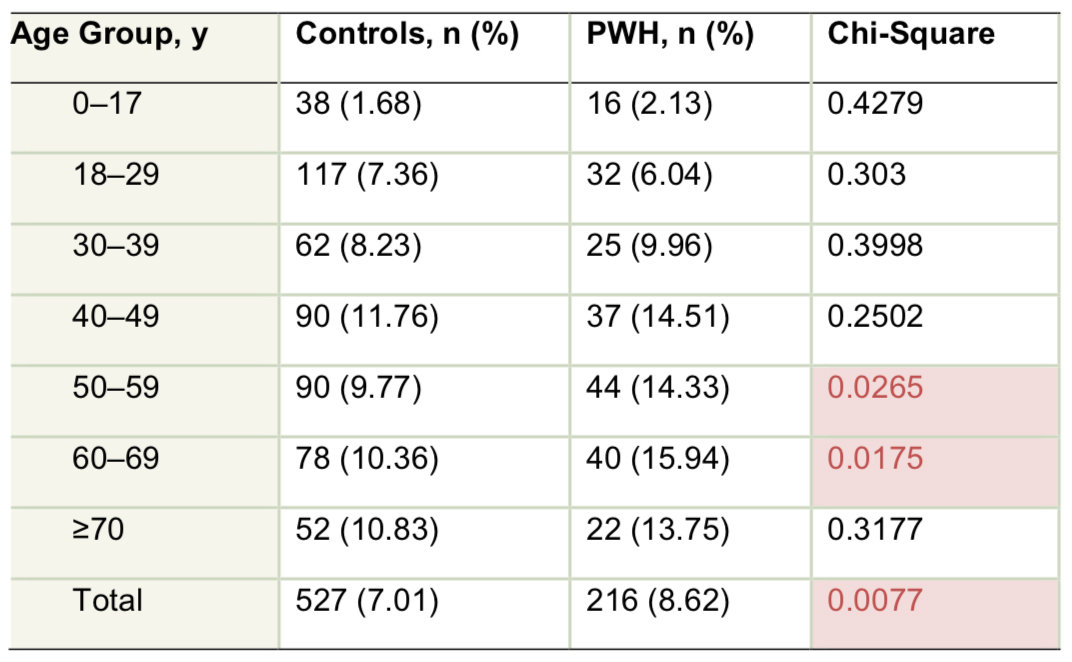
Conclusions:
Commercial claims databases have limitations, including coding errors and inability to verify accuracy of diagnoses. However, in PWH versus a 3:1 control group in a US commercially insured sample that may not be representative of the overall population, depression prevalence was greater in PWH, reaching a peak of 16% in those aged 60–69 years. Awareness of this comorbidity is important as the hemophilia population ages in an era of declining healthcare-delivery resources.
Study Design of a Phase 3, Open-Label Trial of the Safety and Efficacy of Recombinant Factor IX Fc Fusion Protein for the Prevention and Treatment of Bleeding Episodes in Previously Untreated Patients With Severe Hemophilia B
Year: 2014
Grants:
Bleeding Disorders Conference
Available Clinical Studies
Hemophilia of Georgia's Preventive Dental Program
Year: 2014
Grants:
Bleeding Disorders Conference
Psychosocial Issues
Bleeding Risk for the Active Person with Hemophilia: A Comparison of Factor VIII Treatment Regimens
Year: 2014
Grants:
Bleeding Disorders Conference
Available Clinical Studies
Background:
Bleeding risk during physical activities for persons with hemophilia has been shown to be reduced with adequate factor levels (Broderick 2012). With extended half-life Factor VIII and IX products offering opportunities for longer intervals between infusions, it is important to understand the bleeding risk for patients who have an active lifestyle.
Objective:
To evaluate the relative risk of bleeding between prophylaxis schedules using recombinant Factor VIII (rFVIII) vs. rFVIII-Fc among different patient physical activity profiles
Methods:
A mathematical model based on the literature was developed. Factor levels were estimated using a one-compartment pharmacokinetic model (Collins 2010). Half-life and incremental recovery values were taken from a crossover study of rFVIII and rFVIII-Fc (Mahlangu 2014). Five prophylaxis regimens were evaluated: two common to rFVIII (30IU/kg every other day (EOD); 35IU/kg 3x/week) and three studied in the rFVIII-Fc pivotal trial (2x/ week: 25IU/kg covering 3 days and 50IU/kg covering 4 days per week; 50IU/kg every 5 days; 65IU/kg 1x/week). Activities such as swimming, running and wrestling were classified as Type 1, Type 2, and Type 3 in Broderick 2012, derived from the NHF “Playing It Safe Brochure” (Anderson 2005). Risk of bleeding by activity category and factor level at time of activity was calculated using the odds ratio values from Broderick 2012. Three hypothetical patient activity profiles were evaluated: Consistently Active (M-Sun: Type 2 activities), Regular Exerciser (M-F: Type 2 activities, Sat-Sun: Type 1 activities), and Weekend Warrior (TThSun: Type 1 activities, MWF: Type 2 activities, Sat: Type 3 activities). For each regimen, the infusion schedule with the lowest bleeding risk for each patient activity profile was selected. The relative bleeding risk vs. the best prophylaxis regimen was estimated and compared for the activity profiles.
Results:
rFVIII 30IU/kg EOD and 35IU/kg 3x/week achieved the two lowest bleeding risk for all three patient activity profiles. Compared to rFVIII every other day, bleeding risk was increased by 20%, 25% and 46% for the Consistently Active patient prescribed rFVIII-Fc twice per week, every 5 days, and 1x/week, respectively. Compared to rFVIII 3x/week, bleeding risk was increased by 20%, 30% and 44% for the Regular Exerciser and by 21%, 32% and 45% for the Weekend Warrior prescribed rFVIII-Fc 2x/week, every 5 days, and 1x/ week, respectively.
Conclusion:
This model suggests that active patients characterized with the above profiles may have reduced bleeding risk with rFVIII compared to extended half-life FVIII dosing regimens evaluated in this analysis.
A Standardized Approach to Empowering Families with Hemophilia
Year: 2014
Grants:
Social Work Excellence Fellowship
Hemophilia is a rare lifelong condition which can be potentially life-threatening. Parents bare a significant responsibility for delivery of medical care because the treatment for hemophilia begins early in life for children within the home setting. As a result, parents frequently exhibit a heightened level of stress, anxiety, and subsequent trauma around the acceptance of the illness and the administration of medication management. To address the multifaceted nature of chronic illness for patients and their families, the ideal treatment utilizes a multidisciplinary team. Our proposed 3P Patient Parent Power Program aims to standardize care for families with patients of hemophilia using a tiered approach of psychosocial support. The necessary level of support will be provided to parents in order for them to successfully provide in-home prophylactic factor treatment. The goal of the program is to reduce parental stress and anxiety related to this chronic illness and increase feelings of empowerment for the parent and child.
Depression in children with severe Hemophilia - a pilot study
Year: 2014
Grants:
Bleeding Disorders Conference
Psychosocial Issues
Children with severe Hemophilia (CWH) suffer pain and inconvenience due to the required IV factor concentrates' injections and mostly exhibit poor quality of life. These children don't have any hope that their life will improve in the future. Most of them keep the Hemophilia a secret; therefore they are unable to get any emotional support from their peer group. These parameters are known stressors and triggers leading to depression, especially among children and adolescents (due to their lack of mature psychological defense mechanisms). The consequences of depression might be hazardous, since such children may neglect their medical treatment, leading to further deterioration of their medical state.
Objective:
We compared the depression level of children with Hemophilia to healthy children of the same age and background.
Methods:
Depression was evaluated using a standard validated questionnaire of depression that was developed by A.Beck - the CDI. We compared 20 children with severe Hemophilia with 25 non-Hemophiliac children.
Summary:
The calculated score for degree of depression was 7.6 for CWH vs age matched controls with a score of 12.32. The mean of normal populations is around 9. Parametrical T test for Equality of Means = 0.013.
This is the first reported study objectively addressing the issue of depression in CWH.
Conclusions:
We found the opposite of what we had expected: The children with Hemophilia were rated significantly much less depressed then the children without Hemophilia. This finding merits further validation in future larger studies and
must be examined very carefully, due to the complexity of the psychological defense mechanisms.
Using Oral History for Patient Education: The Gift of Experience II: Conversations with Parents about Hemophilia
Year: 2014
Grants:
Bleeding Disorders Conference
Peer Support/Outreach/Integration Models
Modelling the transfer of rFVIIa procoagulant signal from tissue factor to platelets
Year: 2014
Grants:
Bleeding Disorders Conference
Biomedical/Coagulation Research
Ongoing Prospective ADVATE Immune Tolerance Induction Registry (PAIR) Continues to Demonstrate Success Rates Consistent with Published Literature
Year: 2014
Grants:
Bleeding Disorders Conference
Clinical Research
Objectives:
PAIR is an ongoing, global, non-interventional, post-authorization safety surveillance designed to collect information on ADVATE safety and effectiveness in immune tolerance induction (ITI) therapy in routine practice.
Methods:
From July, 2007 to April, 2011, individuals with hemophilia A and inhibitors were enrolled in 10 countries. The primary objective is to assess the incidence of adverse events (AEs) related to ADVATE during ITI therapy. Secondary objectives are incidence of central venous access device (CVAD)-related complications, and success rates of ITI therapy. Maximum observation period for ITI is 33 months plus a 12 month follow-up.
Summary:
As of April 1, 2014, 36 of 44 subjects (81.8%) completed ITI therapy, 28 (63.6%) of which completed the 12 month follow-up. Six subjects withdrew prior to completing ITI therapy. Dosing regimens were: ≥200 IU/kg/day (n=4, 9.1%); 131-199 IU/kg/day (n=3, 6.8%); 90-130 IU/kg/day (n=26, 59.1%) and <90 IU/kg/day (n= 11, 25.0%). During the observation period, 337 bleeding episodes and 273 AEs were reported for all enrolled subjects (N=44). Of these AEs, 52 (19.0%) were serious and none were considered related, while 15 (5.5%) were non-serious and related. CVAD complications were common; 32 subjects experienced one or more CVAD-related AE such as hospitalization, line infection, line malfunction, line removal, and pain following port-a-cath bleed. Of the subjects that completed ITI, 21 achieved negative titer levels, two experienced a high to low titer conversion, seven failed to achieve negative titer, and six were un-assessable per protocol. After 18 months therapy, Kaplan Meier estimates of success for achievement of first negative titer was 65.4% (asymptotic 95% CI: 48.7-81.5%, n=36) for the completer group. Rates were higher for the per protocol analysis set (72.2%, CI: 54.6-87.5%, n=30), and slightly lower for the full analysis set (63.5%, CI: 48.0- 78.7%, n=44).
Conclusions:
These interim outcome results are consistent with previous reports from PAIR and other published data on ADVATE in ITI. No new ADVATE related safety issues have been seen. The last two participating subjects will end observation within the next year.
Safety and efficacy of a recombinant factor IX (BAX326*) in pediatric previously-treated patients with hemophilia B
Year: 2014
Grants:
Bleeding Disorders Conference
Clinical Research
Objectives:
This prospective clinical trial was conducted to assess the safety, hemostatic efficacy and pharmacokinetic (PK) profile of a recently developed recombinant factor IX (BAX326*) in pediatric previously-treated patients (PTPs) with severe or moderately severe hemophilia B.
Methods:
PTPs aged <12 years with severe (FIX level < 1%) or moderately severe (FIX level ≤ 2%) hemophilia B were eligible for enrollment. BAX326 was administered as prophylaxis twice a week over 6-months, and on demand for treatment of bleeds. Efficacy was evaluated by treatment response rating (excellent, good, fair, none) and annualized bleeding rate (ABR). PK assessments after one 75 ± 5 IU/kg infusion of BAX326 were assessed using a non-linear mixed model (population PK) approach. IR was measured as part of the PK evaluation 30 minutes after the initial PK infusion and at 5, 13 and 26 weeks after the initial infusion.
Summary:
Nine subjects (39.1%) had no bleeds during the study. A total of 26 bleeds occurred (mean ABR 2.7 ±3.14, median 2.0), of which 2 were spontaneous. Fewer bleeds occurred in joints than in non-joint sites (19 non joint vs. 7 joint bleeds). Hemostatic efficacy was excellent or good in >96% of bleeds, and the majority (88.5%) resolved after 1-2 infusions. The median IR (IU/dl)/(IU/kg) at the initial PK assessment was 0.685 (range: 0.31- 1.00). As expected, a higher IR was observed in association with increased patient age; IR was slightly lower in subjects < 6 years (median 0.591; range: 0.31-0.75), than in subjects aged 6 to <12 years (median 0.714; range: 0.51-1.00). IR was consistent over time. There were no adverse reactions, no thrombotic events and no hypersensitivity reactions. None of the subjects treated (N=23) developed inhibitory or specific binding antibodies against FIX.
Conclusions:
BAX326 is efficacious and safe as prophylactic treatment as well as for bleed control in pediatric hemophilia B patients.
*Licensed in the USA and Australia (Rixubis®; Baxter Healthcare Corp., USA).
Hemophilia impacts relationships and employment of young adults (ages 18-30) in the us hemophilia experiences, results and opportunities (hero) study
Year: 2014
Grants:
Bleeding Disorders Conference
Psychosocial Issues
Objective:
To assess impact of hemophilia on relationships and employment during the transition to adulthood in young adult (YA) patients with hemophilia (PWH).
Methods:
Analysis of US YA-PWH respondents (aged 18-30 years) in the HERO study.
Summary:
Of 189 US adult PWH HERO respondents, 66 were aged 18-30 years (median: 26). Most lived in large cities (52%) or suburban areas (29%); 73% reported household income of <$60,000/year. Some (32%) were married or in long-term relationships; 20% lived alone. Negative impact on relationships was reported by 32%; 62% predicted an impact in the future, 62% cited difficulty understanding issues, and 52% worry about supporting a family. Only 9% had children; 77% wanted to have children. Only 45% received genetic counseling from the HTC; of those, 60% felt it was helpful. Most were very/quite satisfied with support of partners/spouses (95%), family (92%), and friends (86%). Negative reactions telling friends were reported by 41%; 59% reported most/all of their friends knew about their hemophilia. Most (78%) were employed with 57% reporting office work. One fifth (20%) reported being disabled, and 14% received disability benefits. The majority (74%) reported negative impact on employment; 39% reported moderate/very large impact. Many (43%) reported current treatment allows them to work in most situations, 37% selected a job based on their needs, 29% were helped to obtain a job, 24% had to leave a job, 20% were not hired, and 18% lost a job due to hemophilia. Only 36% received advice from the HTC on work/employment, mostly on what to do if a bleed occurs at work (71%), suitable jobs (67%), when (63%) and what (33%) to tell an employer, and workplace precautions (46%). Fifty-eight percent found the advice helpful. Only 37% reported most/all of their colleagues know about their hemophilia; 38% reported a select few or 1-2 and 26% none. Most were very/quite satisfied with the support of colleagues at work/school (82%). Most YA-PWH (62%) were members of an organization or online (48%) or other (35%) support group.
Conclusions:
During the transition to independent adults, YA-PWH are likely planning for family and careers. Many reported negative impacts on relationships and employment, highlighting a need for career counseling. YA-PWH are supported by friends and colleagues, but this may be a limited group. HTCs are an underutilized resource for addressing these issues, with perhaps online peer-support networks playing a larger role during this transition to adulthood and independence.
Identification of Previously Unreported F8 and F9 Gene Mutations in Hemophilia Subjects From the Phase 3 Clinical Trials of rFVIIIFc and rFIXFc
Year: 2014
Grants:
Bleeding Disorders Conference
Clinical Research
Objective:
Hemophilia A and B are X-linked bleeding disorders caused by the deficiency of clotting factor VIII or IX, respectively. Mutations in the F8 gene can result in hemophilia A while mutations in the F9 gene can lead to hemophilia B. The objective of this analysis was to evaluate the F8 and F9 genotypes of subjects screened for enrollment in the phase III clinical trials of rFVIIIFc in hemophilia A (A-LONG) and of rFIXFc in hemophilia B (B-LONG).
Methods:
The F8 and F9 genotypes of 170 subjects with severe hemophilia A and 114 subjects with severe hemophilia B, respectively, were compared with several genotype databases (HAMSTeRS, [Hemophilia A Mutation, Structure, Test and Resource Site], CHAMP [CDC Hemophilia A Mutation Project], and King’s College London Hemophilia B database), as well as with the NCBI human F8 and F9 sequences.
Summary:
Among 170 subjects with hemophilia A, inversions in intron 22 (Int22inv) were identified in 36%, nucleotide substitutions in 39%, frameshift mutations in 21%, Int1inv in 3%, and an in-frame duplication in 1 subject. Previously unreported mutations (frameshift, missense, nonsense, and splice site changes) were found in 24 subjects, with 2 unrelated subjects having the same mutation, resulting in 23 novel mutations being identified. Among 114 subjects with hemophilia B, the majority (86%) had some form of substitution mutation (missense, nonsense, splice-site change), consistent with previous reports. Thirteen previously unreported mutations were identified, including 10 substitutions (7 missense, 2 nonsense and 1 splice-site change), 1 deletion, and 2 insertions.
Conclusions:
In this analysis, 23 previously unreported mutations in the F8 gene of subjects with severe hemophilia A and 13 in the F9 gene of subjects with severe hemophilia B were identified. Identifying mutations allows for prenatal diagnosis and identification of carrier status. These results will lead to further enhancement of databases for hemophilia A and B mutations and may assist in clarifying the relationship between genotype, phenotype, and pathophysiology in individuals with hemophilia.

