Utilization of Telehealth for Home Infusion Teaching and Support in the COVID Era
Characterization and management of patients with mild or moderate hereditary factor X deficiency: a retrospective chart review
The World Federation of Hemophilia World Bleeding Disorders Registry: A Two-year Update
The Impact of Novel Hemophilia Treatment Products on Inhibitor Testing for the Community Counts Registry for Bleeding Disorders Surveillance
Optimizing language to increase understanding, improve communication, and manage expectations around gene therapy for hemophilia: a qualitative study
Development of the WFH-ISTH Gene Therapy Registry
Continuous infusion of B domain-truncated recombinant factor VIII, turoctocog alfa, for an acute moderate bleeding episode in hemophilia A: a first case report
A Review of Current Patient Reported Outcome Measures Used to Assess Mental Health in People with Hemophilia
A Novel Approach for Rare Bleeding Disorders: Shielded Living Therapeutics
The 2020 World Federation of Hemophilia Guidelines for the Management of Hemophilia
Behavior and cognition in children and young adults with hemophilia A or B: an update on developmental outcome
Characterization of the Early Immune Response to Factor VIII
Unmet Needs in Women with Severe Von Willebrand Disease
The Moti-VIII Study – Factors for Empowering Mobility and Well-being in Hemophilia A
Retrospective review of Hemophilia patients before and after treatment with Emicizumab
Assessing and Responding to the Oral Health Care Needs of Adults in a Bleeding Disorders Population
Redefining Treatment Satisfaction and Its Impact on Treatment Adherence and Value for Persons with Hemophilia: Findings from the HemACTIVE Study
Bone and joint health markers in persons with hemophilia A treated with emicizumab in the HAVEN 3 clinical trial
Summary of thrombotic or thrombotic microangiopathy events in persons with hemophilia A taking emicizumab
Non-severe hemophilia is not benign? - Insights from the PROBE Study
Objective:
There are limited data on the impact of haemophilia on health status and health-related quality of life (HRQL) in people affected by non-severe haemophilia. Aim is to evaluate the health status of people living with mild or moderate haemophilia.
Methods:
A cross-sectional, multinational study was conducted as part of the Patient Reported Outcomes, Burdens and Experiences (PROBE) project. Respondents without bleeding disorder (NoBD) and those with mild or moderate haemophilia were included. Respondents were asked to complete the PROBE questionnaire, which contains haemophilia-related questions, general health questions and HRQL. Results were compared between unaffected individuals and people with mild or moderate haemophilia.
Summary:
A total of 862 respondents, of whom 144 with moderate haemophilia, 143 with mild haemophilia and 575 with NoBD were included. Median age (first-third quartile) was 33 (23-46),42 (25-55) and 43 (35-54), respectively. In relation to bleeding in the previous 12 months, respondents with mild reported less bleeding frequency than those with moderate haemophilia, with similar patterns of bleeding frequency seen in both male and female cohorts. Reporting of acute pain is less in those with NoBD compared to the mild to moderate cohorts for both genders (male - 33%, 67%, 77%; female - 38%, 52%, 67%, respectively). Thirteen percent of those with NoBD reported an impact on activities of daily living compared with mild and moderate haemophilia who reported of 35% and 61%, respectively. The impact on quality of life due to mild haemophilia compared to those with NoBD was a reduction of 5.2%, 5.0% and 9.3% in VAS, EQ-5D-5L and PROBE Score respectively (p≤0.001).
Conclusions:
People affected by mild or moderate hemophilia encountered a significant amount of haemophilia related sequalae. Future research is needed to identify the optimal management of moderate and mild hemophilia patients, with particular focus on early identification of patients with a severe clinical phenotype.
3 apps in 1: MyCBDR, myWAPPS and myPROBE
A systematic review of mortality statistics and causes of death in people with congenital hemophilia A (PwcHA)
Characteristics of persons with hemophilia A treated with emicizumab with or without factor VIII inhibitors
Final Results of PUPs B-LONG Study: Evaluating Safety and Efficacy of rFIXFc in Previously Untreated Patients With Hemophilia B
Final Results of PUPs A-LONG Study: Evaluating Safety and Efficacy of rFVIIIFc in Previously Untreated Patients With Hemophilia A
Evaluating BIVV001, a New Class of Factor VIII Replacement Therapy: A Phase 3 Study (XTEND-1) Design
The Need for Comprehensive Care for Persons with Chronic Platelet Disorders
Treatments and Clinical Outcomes of Bleeding Related to Pregnancy, Surgery, or Spontaneous or Traumatic Bleeds in Women and Girls With Factor VIII and IX Deficiency: Results From a Retrospective Chart Review
An analysis of fatalities in persons with congenital hemophilia A (PwcHA) reported in the FDA Adverse Event Reporting System (FAERS) database
A contemporary framework for understanding mortality in people with congenital hemophilia A (PwcHA)
A single administration of AAV5-hFIX in newborn, juvenile and adult mice leads to stable hFIX expression up to 18 months after dosing
Progress Update on the Development of Etranacogene Dezaparvovec (AMT-061) in Severe or Moderately Severe Hemophilia B
Vector DNA clearance from bodily fluids in patients with severe or moderate-severe hemophilia B following systemic administration of AAV5-hFIX and AAV5-hFIX Padua
An ECHO’d Practice: Utilizing Tele-Mentoring for Enhanced Data Quality Across One Hemophilia Treatment Center Region
Real-world treatment patterns, health outcomes, and healthcare resource use among persons with hemophilia A
Case Report of Surgical Management of a Hemophilia B Gene Transfer Clinical Trial Participant Following Etranacogene Dezaparvovec (AMT-061) Gene Therapy
Supporting patient voice to inform healthcare decision-making: a discrete choice experiment on disability paradox in hemophilia
Analysis of Bleeding and Treatment Patterns in Children and Adolescents before and after Von Willebrand Disease Diagnosis Using Data from a US Medical Claims Database
Objectives:
Von Willebrand disease (VWD) is the most common bleeding disorder in children and adolescents. Its varied clinical presentation contributes to challenges and delays in diagnosis and management. We characterized diagnosis, bleeding, and treatment patterns in children (2-11yrs) and adolescents (12-17yrs) with VWD.
Methods:
This retrospective database analysis utilized data from IQVIA PharMetrics Plus Database of medical insurance claims for VWD patients (ICD-9 286.4) from 01/01/2006 to 06/30/2015. Patients included had ≥2 medical claims for VWD and continuous enrollment for ≥2 years, to ensure higher likelihood of definitive VWD diagnosis, before/after their 1st VWD claim. Pre-diagnosis period included 18mos of data before diagnosis. Post-diagnosis period included 7-24mos post-diagnosis data. Data from the first 6-month post-diagnosis period were excluded due to data variability, suggestive of treatment optimization. Descriptive statistics were used to summarize patient demographic/clinical characteristics, including types of bleeding episode (BE), rates, outcomes; treating physician specialty; and type of VWD treatment, in both pre-/post-diagnosis periods.
Results:
475/1087 patients identified were children (43% female; mean age at diagnosis 6.9yrs; 612 were adolescents (74% female; mean age at diagnosis 14.9yrs). The top 3 treating physician specialties seen by children in the pre-/postdiagnosis periods, respectively, were hospitalists (21%/9%), primary care physicians (16%/7%), and hematologists (11%/3%). Adolescents were mostly seen by hospitalists (30%/15%), primary care physicians (25%/16%), and obstetrician gynecologists (19%/15%). 11% of children and adolescents saw a hematologist prior to diagnosis, compared with 3% and 5%, respectively, post-diagnosis. A 17%/15% decrease in bleed claims in the pre-/post-diagnosis period was observed among children (40%/23%) and adolescents (59%/44%), respectively. The most common type of BE among children in the pre-/post-diagnosis periods was epistaxis (19%/10%). Heavy menstrual bleeding was the most common BE among adolescents in both the pre-/post-diagnosis periods (40%/30%; in females 54%/40%). Epistaxis was the second most common BE among adolescents in both the pre-/post-diagnosis periods (11%/7%), and in females (9%/5%), but highest among males (17%/12%). Overall, VWD-related treatment claims increased between the pre-/post-diagnosis periods for both children (12%/23%) and adolescents (31%/50%). The most prescribed treatments for bleed management in children were aminocaproic acid (ACA), desmopressin (DDAVP) and nasal cauterization (pre-diagnosis: 5%, 4% and 4%, respectively; post-diagnosis: 11%, 13% and 3%, respectively). For adolescents, the most prescribed treatments, pre- and post-diagnosis respectively, were oral contraceptives (22% and 33%, DDAVP (9% and 19%) and ACA (4% and 11%).
Conclusions:
This analysis demonstrates a decrease in BE claims following VWD diagnosis and a rise in ACA and DDAVP treatment claims in both children and adolescents, and in oral contraceptive claims among female adolescents. Nevertheless, a considerable proportion of children and adolescents continue to experience BEs 6mos post-diagnosis. This emphasizes the need for treatment optimization and improvement in care and management of patients in these age groups.
Adding the patient's voice to a hemophilia-specific goal menu to facilitate Goal Attainment Scaling: a qualitative study
Incidence and Prevalence of Diagnosed and Undiagnosed Hemophilia A and Hemophilia B in the USA
Overview of the clinical development of fitusiran
Intra-individual across-study comparison of the pharmacokinetics of rFVIII-FS, BAY 81-8973 and BAY 94-9027 in patients with severe hemophilia A
A US payer database algorithm to identify clinical profiles of hemophilia B for burden of illness assessment
On My Own, A Pilot Transition Program for Teens
Longitudinal trends of patient-focused programs in the bleeding disorders community from 2013-20: a retrospective analysis of Hemophilia Alliance Foundation grants
Fear of pain in people with hemophilia and their families – a pilot study
Application of attention destruction techniques during factor concentrates infusion to children with Hemophilia
Empowering the Future of Hemophilia Through Swimming (Poster Abstract)
Women and girls with hemophilia: Gender-based differences in comprehensive care
von Willebrand Factor in Pregnancy (VIP) Study: A Multicenter Study of Wilate Use in von Willebrand Disease for Childbirth
Specific guidance is lacking for delivery planning in terms of how high a factor level should be achieved for pregnant women with von Willebrand disease (VWD) who, by the third trimester, do not have von Willebrand factor (VWF) (or factor VIII) levels greater than 50-100%. Specifically, guidance is lacking on whether replacement therapy should target a VWF minimum level in the 100–150% range, i.e., a range closer to the 200–250% levels observed in normal pregnancy.
Objectives:
The primary objective is to document the rate of primary postpartum hemorrhage (PPH) and thereby the effectiveness of targeting minimum VWF levels of 100–150% for delivery. The secondary objective is to document further effectiveness outcomes and safety. Patient VWF levels will be maintained at 100-150% for the immediate 72-hour postpartum period, and thereafter maintained at 50-100% target VWF levels through days 5-7 postpartum after normal vaginal delivery or days 7–10 postpartum after caesarean section.
Methods:
This is a prospective, open-label, cohort study of the dosing of Wilate in pregnant patients with VWD to achieve minimum VWF levels of 100–150% for delivery. Outcome parameters will be assessed among patients termed non-correctors and correctors. Patients with a third trimester (gestational week 34–38) VWF level <100% will be enrolled in the non-corrector group. Patients with VWF levels ≥100% at gestational weeks 34–38 will be enrolled in the corrector group. Sample size is based on 65 pregnant VWD non-corrector patients and up to 30 corrector patients. Both correctors and non-correctors will be given tranexamic acid post-partum for 14 days.
Inclusion Criteria: includes VWD patients diagnosed prepartum as type 1 per NHLBI criterion of VWF level <30%, or type 2, or type 3. Exclusion Criteria includes age <18 years, presence of other concurrent disorder of hemostasis, platelet dysfunction, or collagen disorders; presence of liver disease or renal disease, clinical suspicion or diagnosis of preeclampsia or eclampsia, HELLP syndrome, TTP, DIC, or other acquired vasculopathy or coagulopathy, or inability to perform local laboratory monitoring. Primary outcome parameter will be the rate of primary PPH, defined as estimated blood loss ≥1000 mL, or severe PPH defined as estimated blood loss >2000 mL within 24 hours postpartum. Other outcomes are secondary PPH, laboratory measures, and safety. Screening will begin in Q3 2019 and end in Q2 2023, with recruitment ending 6 months before (i.e., Q4 2022).
Summary:
This planned study aims to determine in VWD if VWF levels postpartum should be attained at levels closer to levels achieved physiologically in a normal pregnancy.
Conclusions:
Results from this study will hopefully lead to reduction of the relatively high rate of PPH in VWD women with levels <50-100% in the third trimester.
A retrospective chart review to assess clinical characteristics of women and girls with factor VIII and IX deficiency
Objective:
Evaluate clinical characteristics, hemostasis management, and clinical outcomes regarding menstruation, child birth, surgical procedures, dental care, and spontaneous and traumatic bleeds of women and girls with factor VIII (FVIII; hemophilia A) or factor IX (FIX; hemophilia B) deficiency (WGFD).
Methods:
A retrospective chart review is ongoing at three US hemophilia treatment centers (HTC) to collect data on WGFD (obligate or potential carriers of FVIII or FIX deficiency, with or without genetic confirmation). Data are collected on patients who had at least two HTC visits and underwent medical or surgical interventions for hemostasis management between April 2012 and November 2018, with the outcome available in medical charts.
Summary:
Interim results as of April 5, 2019 include charts from two HTCs on 26 (89.7%) patients with FVIII deficiency and 3 (10.3%) patients with FIX deficiency. The median (range) age at factor deficiency diagnosis was 18.5 (0.1–72.0) years. Twenty-four (82.8%) and 8 (27.6%) patients had a family history of hemophilia and other bleeding disorders, respectively. A total of 17 (58.6%) patients initially visited the HTC due to family history/genetic counseling. Other reasons for visiting an HTC were heavy menstrual bleeding (n=12 [41.4%]) or spontaneous or traumatic bleeds (n=12 [41.4%]), including 7 (24.1%) patients reporting both heavy menstrual bleeding and spontaneous or traumatic bleeds. Of the 12 patients with spontaneous or traumatic bleeds, 4 (33.3%) patients had joint bleeds, 6 (50.0%) patients had excessive nose bleeds, and 9 (75.0%) patients had easy bruising. For those with FVIII deficiency, the median (range) FVIII level at diagnosis was 32.5 (2.0–101.1) IU/dL (n=24), median (range) baseline hemoglobin was 12.9 (5.4–14.8) g/dL (n=19), and median (range) baseline von Willebrand factor ristocetin cofactor was 70 (40–150) IU/mL (n=16). The median (range) number of documented bleeds was 1.0 (0.0–24.0) in the first year at the HTC. Final results of this chart review, including data from those with FIX deficiency, HTC interventions, and outcomes for hemostasis management, will be presented.
Conclusions:
This chart review provides further insights into the clinical presentation and hemostasis management of WGFD evaluated at HTCs in the US. Results may contribute to the design of future prospective studies evaluating treatment options for this patient group.
Female Patients with Hemophilia A: A Claims-Linked Chart Review
Von Willebrand Disease: An international Survey to Inform Priorities for New Guidelines
Background:
Von Willebrand disease (VWD) is an inherited bleeding disorder caused by a quantitative or qualitative deficiency of the protein, von Willebrand factor (VWF). There is a lack of clear guidance on best practices to inform the care of people with VWD.
Objectives:
Identify and prioritize the main topics of a collaborative guideline development effort.
Methods:
A scoping survey to prioritize topics to be addressed in a collaborative guideline for VWD was distributed to international stakeholders including patients, caregivers, clinicians, and allied healthcare professionals. The distribution strategy was coordinated by the guideline chairs and representatives of the American Society of Hematology (ASH), the International Society on Thrombosis and Haemostasis (ISTH), the National Hemophilia Foundation (NHF), and the World Federation of Hemophilia (WFH). The survey was conducted in English, French, and Spanish. The survey focused on both diagnosis and management of VWD, using 7-point Likert-scale response options and open ended comments. Descriptive analysis of participants and comparative analysis of results by stakeholder subtype (patients/caregivers versus healthcare providers [HCP]), gender, and income setting was performed. Qualitative conventional content data was analyzed utilizing both deductive and inductive coding processes.
Results:
601 participants responded to the survey (49% patients/caregivers, and 51% HCPs). The highest priority topics identified were diagnostic criteria/classification, bleeding assessment tools, treatment options for women, and surgical patients. In contrast, screening for anemia and plasma-derived therapy versus recombinant therapies were rated the lowest priority topics (figures 1 – 2).
Conclusion:
The survey results highlighted areas of importance in the diagnosis and management of VWD across diverse groups of stakeholders and will direct future guideline efforts. The large number responses (601) and discrete comments (9,500) attest to the interest and involvement of the VWD community in this effort.
The Patient Reported Outcomes Burdens and Experiences (PROBE) Study Questionnaire Development and Validation
Combining Data from Hemophilia Registries with the World Bleeding Disorders Registry: A Proof of Concept Study with the Czech National Haemophilia Programme Registry
Data is the new currency: The World Bleeding Disorders Registry Data Quality Accreditation Program
World Federation of Hemophilia Annual Global Survey 2017 – 19 years of reporting
World Federation of Hemophilia Annual Global Survey analysis of age distribution of patients with hemophilia
The WFH World Bleeding Disorders Registry – 16-month update
Patient Perspectives on the Impact of Severe or Moderate Hemophilia on Physical Activity: HemACTIVE Survey Findings from the US and Canada
Evaluation of Patient and Physician Reported Reasons for Switching FVIII Replacement Therapies Among Patients With Hemophilia A
Objective:
While a new generation of therapies for patients with Hemophilia A are available, it is unclear what patient characteristics, perceptions, and barriers are associated with the decision to switch FVIII replacement therapies. This study assessed patient characteristics, health history, and reasons for switching from a FVIII product with more frequent dosing (³3x infusions/week) to a product with less frequent dosing (≤2x infusions/week) from patient/caregiver and physician perspectives.
Methods:
Data collection was a mix of qualitative and quantitative procedures. The qualitative portion consisted of two online discussion forums: patients (n=17) and caregivers of patients (n=11) receiving a FVIII product dosed ³3x/week, and patients (n=22) and caregivers of patients (n=5) who switched to a product dosed ≤2x/week. The quantitative portion was a retrospective medical chart review (n=207) which captured variables (e.g., bleed rate, treatment history) 6 months pre- and 6 months post-switching to a product with less frequent dosing.
Summary:
Prominent drivers among patients for starting a FVIII product with less frequent dosing included: 1) experiencing diminished effectiveness while on a product dosed ³3x/week resulting in increased breakthrough bleeding, 2) experiencing vein access issues, and 3) beginning prophylaxis as opposed to on-demand infusions after a bleed.
Key barriers to changing included: 1) fears regarding the process of switching being complicated, time consuming, and costly, 2) perceived risks associated with switching, and, 3) possible lack of healthcare provider support.
Physicians were most likely to report that patients switched products because they sought a newer product with twice weekly dosing or less per FDA-approved dosing recommendations (35.3%), followed by patient requested the switch (30.4%), and patient sought a reduction in infusion frequency to improve adherence (27.5%).
Switching to a product with less frequent dosing was associated with improvements in patient-reported bleeding-related outcomes. Patients were more likely to self-administer the treatment post-switch (63.8%) compared with pre-switch (48.8%; p<0.001) and had fewer infusions per week post-switch (2.8 vs. 3.3; p<0.001). Patients’ annualized bleed rate was lower (5.9) post-switch compared with pre-switch (7.7; p<0.001).
Both the number of spontaneous joint bleeds and joint bleeds after trauma or injury were lower (3.2 and 2.7) post-switch (3.6 and 4.3; p=0.044 and p<0.001). The bleeding event was less likely to be classified as moderate or severe (34.5% and 5.9%) post-switch compared with pre-switch (55.0% and 10.0%; p<0.001 and p=0.049). Fewer infusions were required to resolve the bleeding event post-switch (2.6 vs. 3.2; p<0.001).
A prominent reason why patients switch treatment is to improve bleeding-related outcomes. Indeed, we found that switching to a FVIII product with less frequent dosing was associated with improved patient-reported bleeding-related outcomes. These findings are critical for improving patient outcomes and support the FDA mandate to incorporate patient perspectives in the regulatory process.
Increasing Medical Alert Devices (MAD) Compliance in School Age Children with Hemophilia: A Quality Improvement Project
Patients Report High Satisfaction with US Hemophilia Treatment Centers: National Trends 2014 and 2017
Background:
US Hemophilia Treatment Center (HTC) care reduces mortality and hospitalizations, and guidelines recommend this care model. Yet national data that uniformly and longitudinally monitors patient experience with HTC care is limited.
Objective:
To assess patient satisfaction with HTC services and clinicians over time.
Methods:
The US HTC Network conducted the first ever nationally uniform patient satisfaction surveys on care received in 2014 and 2017. A Regional workgroup devised, piloted, and finalized an electronic, two-page survey for self-administration at clinic, or at home, in English or Spanish. Content was based on national instruments to enhance comparability and scientific robustness. Questions assessed demographics; satisfaction with HTC team members and services; insurance and language barriers. Respondents were anonymous but identified their HTC. Participation was voluntary. Patients with HTC contact in 2014 and 2017 were eligible. Data were collected for 4 months in 2015 and 6 in 2018; on average 130 HTCs (94%) from all US regions participated. Parents completed surveys for children under age 18. Data were entered, analyzed and aggregated at national, regional and HTC levels at a central site.
Results:
5006 and 4767 persons participated, respectively, in 2015 and 2018. In both years, over 1400 (30%) respondents were female, nearly 80% were White, and 10% Hispanic. On average, 3038 had Factor 8 or 9, 1280 Von Willebrand, 186 other factor deficiencies and 369 other bleeding disorders. Respondents reported being ‘always’ or ‘usually’ (A/U) satisfied with HTC staff and services from 90% - 97% of the time in both 2014 and 2017. In both years, >4400 gave these highest A/U ratings for HTC Hematologists and Nurses; 3300 for Social Workers; >2600 for Physical Therapists; 1400 for Genetic Counselors, and >1100 for Psychologists. In both years, 96% were A/U satisfied overall with HTC services. Over 95% gave the A/U satisfaction ratings both years for these services: getting needed care and information, being treated respectfully, spending sufficient time with staff, and involved in shared decision making. 82% and 91% of respondents, respectively, gave the A/U satisfaction ratings for care coordination with primary care providers and other specialists. Over 90% of >700 youth age 12-17 gave HTC teen transition services the A/U satisfaction ratings both years. 96% of >2760 respondents reported A/U satisfaction with their HTC Pharmacy (340B) Factor Program in 2017. Insurance and language barriers to HTC care posed problems A/U for 27% and 15%, respectively both years.
Conclusions:
Patients consistently report high levels of satisfaction with HTCs, documenting HTC value over time. Patient satisfaction influences treatment adherence, can influence reimbursement, and is increasingly required by payers. A national uniform survey is feasible to conduct using a regional structure to implement, is well received by patients, and provides critical information to stakeholders.
Satisfaction with Teen Transition Services at US Hemophilia Treatment Centers by Center – Variation by Pediatric and Lifespan Centers 2014 and 2017
Helping teens with bleeding disorders prepare to manage their care as they transition to adulthood is a national priority for US Hemophilia Treatment Centers (HTC). The National HTC Patient Satisfaction Surveys (PSS) reveal high satisfaction with HTC teen transition services. Yet how satisfaction differs comparing HTCs that primarily care for children to HTCs that care for patients throughout the lifespan is unknown.
Objective:
To assess variation in patient satisfaction with US HTC teen transition services by HTC type.
Methods:
The US HTC Network conducted nationally uniform patient satisfaction surveys in 2015 and 2018 on care received, respectively, in 2014 and 2017. A Regional workgroup devised, piloted, and finalized an electronic, two-page survey for self-administration at clinic, or at home, in English or Spanish. Participation was voluntary. Respondents were anonymous but identified their HTC. Parents completed surveys for children under age 18. The PSS included two teen transition questions for respondents age 12-17 to complete. HTC type was categorized as ‘pediatric’ if >80% of responses were from patients/caregivers of individuals under age 18, and ‘adult’ if >80% were from patients over age 24. All other HTCs were categorized as ‘lifespan’. For both years, approximately 26% of HTCs were classified as pediatric, 52% as life-span, and 22% as adult.
Results:
Over 700 teens age 12-17 (or their parents/guardians) from an average of 130 HTCs (94.0%) from all US regions participated in 2015 and 2018. Approximately 96.5% of teens at pediatric HTCs (96.4% - 96.5%) and 96.2% at lifespan HTCs (95.9% - 96.5%) reported being ‘always’ or ‘usually’ (A/U) satisfied with HTC services overall. On average, 90.4% of teens at pediatric HTCs (90.1% - 90.7%) and 91.0% at lifespan HTCs (90.3%–91.6%) reported being A/U satisfied with how HTC clinic staff talked about how to care for the bleeding disorder as they became an adult. Similarly, 92.5% (92.0%– 92.9%) of teens at pediatric HTCs and 92.5% (92.3%-92.7%) reported being A/U satisfied with how the HTC clinic staff encouraged them to become more independent in managing their bleeding disorder.
Conclusions:
HTC patients age 12-17 years consistently report very high levels of satisfaction with HTC teen transition services, regardless if the HTC primarily cares for patients up to age 17, or throughout the life-span. This suggests teens receive support and tools to successfully transition to adult care across the US HTC Network. A national uniform HTC Patient Satisfaction Survey provides vital information, is feasible to conduct using a regional structure, and well received nationwide.
An evaluation of health utility and quality-of-life in hemophilia: a systematic literature review
Objective:
Hemophilia may negatively impact a patient’s health utility and quality of life (QoL). Health state utility values (HSUVs) and QoL are important inputs to the evaluation of novel treatment being developed in hemophilia, including gene therapies. This systematic literature review identified and evaluated HSUVs and QoL for people with hemophilia (PWH) type A and/or type B, as well as utility decrements relevant to the experience of PWH, by treatment and health state.
Methods:
Building on a review undertaken in 2014 (Grosse et al. 2015), we conducted a systematic literature review to March 2019 through a search of electronic medical databases, including MEDLINE®, Web of Science, Cochrane Library databases and the School of Health and Related Research Health Utilities Database (SCHARRHUD). Major clinical, patient, and pharmacoeconomic conferences in 2016-2019 were also queried. Studies were independently double screened by independent reviewers, after which data extraction was performed. The information extracted included study design, description of treatment and health state, respondent details, instrument and tariff, HSUV and QoL estimates, quality of study, and appropriateness for use in economic evaluations of novel treatment.
Summary:
Of 1,511 titles and abstracts screened, 20 studies and 12 conference abstracts were included. The studies identified applied a mix of direct and indirect health utility elicitation techniques. Two studies applied direct time trade-off (TTO) methodology and the remaining 30 studies adopted indirect valuation methodologies. HSUVs were found to decrease with increasing disease severity. For example, in Hoxer et al. (2018), mean (standard deviation) HSUV were 0.80 (0.21), 0.73 (0.22) and 0.67 (0.25) in people with mild, moderate, and severe hemophilia, respectively.
Utility values were also found to vary by severity of musculoskeletal damage, frequency of bleed episodes, inhibitors, hemophilia subtype, treatment regimen, treatment adherence and other disease-related complications. Interestingly, HSUVs derived from valuations from the general public were found to be valued lower than those derived from PWH for similar health states. For example, in Carlsson et al. (2017), general population participants consistently rated significantly lower HSUVs for hemophilia disease states compared to PWH (range: 0.54-0.60 vs. 0.67-0.73).
Several hemophilia-specific QoL instruments were used alongside HSUV evaluations. These QoL findings further contribute to improving the understanding of the impact of hemophilia on PWH.
Conclusions:
This systematic review shows significant impact of hemophilia on health utilities and QoL among PWH. The substantial humanistic burden experienced by PWH highlights unmet needs remaining in hemophilia. Our review findings also suggest potential disease state adaptation among PWH, which warrants further research using robust patient preference studies.
Quality of life and health in patients with Haemophilia in Mexico
A look from within: a needs assessment of educational support for the Rare Bleeding Disorders Community
Objective:
The National Hemophilia Foundation Education team partnered with an evaluator to conduct a needs assessment of the rare bleeding disorder (RBD) community to help inform the development of programming tailored to the community’s unique experiences and needs.
Methods:
A guided discussion with the attendees of a Bleeding Disorder Conference (BDC) session titled, “The Lonely Island: Dealing with Being Rare” in 2018 as well as brief surveys at the end of the session were compiled as part of the needs assessment. Additionally, 12 one on one interviews of those part of the RBD community (either affected themselves or a close relative to someone that is affected) were conducted.
Summary:
Various challenges for this population were identified, including: connecting with others who have the same RBD; healthcare providers’ lack of knowledge/understanding of specific RBDs; accessing knowledgeable hematologists and RBD experts; accessing the latest science specific to their RBD; scarcity of treatment resources; difficulty getting diagnosed. Other secondary challenges were also expressed. While challenges were identified, those that participated in the needs assessment also highlighted the ways in which they see the RBD community can best be served. Common suggestions included: the addition of RBD-specific programming at NHF’s Bleeding Disorder Conference (BDC); continuing to make NHF and Chapters inclusive; creating more opportunities for the RBD community to connect with others with the same RBD (at NHF’s BDC and other events); creating targeted educational materials and opportunities for the RBD community; creating opportunities for members of the RBD community to identify and engage with the medical community.
Conclusions:
By conducting this needs assessment, NHF took an important step in asking the RBD community directly how they can best be supported given their unique experiences and needs. While challenges for the RBD community were identified, several opportunities to support the RBD community were also identified.
Identification of Orthopedic and Genetic Needs Reported by Persons with Type 3/Severe Von Willebrand Disease
Objective:
To determine the medical and educational needs reported by persons with Type 3 and other severe types of Von Willebrand Disease (VWD) who attended the second USA National Type 3/Severe VWD Conference held in Florida in June 2018.
Little research has been done concerning medical issues and education in Type 3/Severe VWD. As patient identification increases, it is vital that education, support and resources are available for these patients.
Methods:
A survey of 48 questions was developed and administered to 74 vetted patient attendees. Responses for any individual question varied between 62-66. The survey was administered through an Audience Response System (ARS) utilizing handheld clickers. The responses were compiled and immediately visually available to the respondents via a projector screen.
The multiple-choice questions were used to identify basic demographics, medical and psychosocial concerns, and educational needs.
Summary:
In this self-reported ARS survey, basic demographic data was obtained. This sample of VWD patients reported a need for more education on several issues related to their medical and psychosocial issues including depression/mental health issues, lab results and product choices. In addition, subjects reported significant needs for care, treatment and education in the fields of orthopedic services and genetic counseling.
Respondents' answers expressed a lack of orthopedic care despite a need for it. Only 8 (13%) patients reported having an orthopedic surgeon attend his/her bleeding disorder clinic. Forty-two (67%) did not know of any orthopedic resources. However, 18 (28%) reported that he/she had already had at least one joint surgery/procedure due to VWD and 5 (*%) plan to have surgery in the future. Eight (12%) had had joint replacements.
Only 25 (40%) of respondents knew that they had undergone genetic testing related to their bleeding disorder, 30 (48%) have not had genetic testing, 8 (13%) were unsure. When asked, “Were your parents diagnosed with a bleeding disorder before your birth?” of the 63 who answered, 51 (81%) stated “no, neither parent”. When asked if a parent was diagnosed with a bleeding disorder after the respondent’s birth, 24 (38%) responded “yes” to one or both parents. Twelve (19%) respondents have had their diagnosis change since first being identified with a bleeding disorder.
Conclusion:
Orthopedic care, genetic testing and education are vital services wanted by Type 3/Severe VWD patients. The community should further evaluate these needs and take action to respond. These results may also empower persons with Type 3/Severe VWD to seek support from professional and social members of their community.
Optimizing language for effective communication of gene therapy concepts: A qualitative study
The Effect of Bleeding Disorder Characteristics on Patient Perceived Challenges and Management Strategies
Objective:
To investigate how bleeding disorder characteristics influence patient perceived challenges and management strategies.
Methods:
This is a mixed-method, retrospective, cross-sectional continuation of a pilot study identifying themes of self-perceived challenges and management strategies for persons with bleeding disorders. Sixty-one participants with a bleeding disorder (BD), either hemophilia (PWH) or Von Willebrand disease (PWVWD), were asked what their most significant challenge was in managing their BD and how they managed that challenge. Data were collected from March, 2017 through December, 2018, coded for themes and uploaded to NVivo. Similar themes were grouped for analysis. Subject-level data was extracted from the electronic medical record including demographics, disease type, severity and presence of joint disease (JD). Pain interference was determined from participant response to the Brief Pain Inventory (BPI).
Results:
- The mean age of the cohort was 31.4 years, with a median of 25 years, and range of 7 to 75 years. 87% were PWH, 13% PWvWD.
- There were 26% mild, 25% moderate, and 49% severe PWH.
- 54% had JD.
- Identified challenges included: participation restriction (24%), acute bleeding (22%), infusion (19%), bleeding sequelae (10%), other’s unfamiliarity with bleeding disorder (other’s unfamiliarity) (10%), no challenges (10%), and other (6%). Management strategies reported were: acceptance (37%), learning through experience (25%), education/advocacy (11%), seeking help (9%), other (9%), and no challenges (9%).
- Severe PWH greatest reported challenges were participation restriction (27%) and infusion (27%). Management strategies were acceptance (41%) and experiential learning (31%).
- Mild-moderate PWH greatest reported challenges were acute bleeding (35%), infusion and no challenge (17% each). Management strategies were acceptance (30%) and seeks help (26%).
- Seek help was not identified as a strategy among severe PWH.
- Reports of no challenges was higher among those <18 years than those ≥ 18 years.
- Mean pain interference was 13.2 out of 70 based on the composite score of BPI measures.
- As age increased, the likelihood of JD and interference increased.
- Regardless of challenge, people with JD reported interference averaging 18% (range 0-27%).
Conclusions:
Gaining insight to patient-perceived challenges and management strategies is important to be able to tailor an effective treatment approach that is individualized and meets the changing needs of PWBD across circumstance and life-course.
Using Photovoice with the Bleeding Disorder Population: A Pilot Project
Introduction:
Photovoice is a qualitative research method that has been used for communities to share pictures as a tool for discussion that is often used at a grassroots advocacy level. Photovoice can show both strengths about a topic or concerns. Photovoice can create empowerment by sharing perspectives and can also create a foundation to advocate for awareness and change.
Long-term Goal:
To create more awareness surrounding bleeding disorders during the month of March, which is bleeding disorders awareness month.
Objectives:
- To apply Photovoice methodology to the use of social media among the hemophilia population in the Cincinnati, Ohio geographical area.
- To engage people with hemophilia and their families in sharing their stories related to their bleeding disorder by sharing photographic images on social media during bleeding disorders awareness month.
Methods:
People that follow the Tri-State Bleeding Disorder Foundation on social media as well as members of a closed social media group that consist of parents of children who are patients at Cincinnati Children’s Hospital were asked to participate in the pilot Photovoice project. Participants were asked to share pictures on their own social media pages and to use hashtags to link the photos to the Tri-State Bleeding Disorder Foundation’s page. The project was promoted by sharing an infographic that explained Photovoice and the details of the project. Several community stakeholders were identified as people active on social media and they were personally asked to participate so that examples of the project could be shared with others. There were weekly themes and a weekly contest for pictures that best exemplified that week’s theme with the winners winning a small gift card.
Summary:
This innovative pilot project applied the methodology of Photovoice to social media to generate awareness and advocacy during bleeding disorders awareness month. The theme of this Photovoice project was “Living with Hemophilia”. Weekly themes consisted of: living with a new diagnosis, living with treatment, living and learning about a bleeding disorder, and living with health and being physically active.
Conclusion:
Utilizing Photovoice and applying this methodology to social media as a pilot project with the bleeding disorder population is an innovative idea. This grass roots level movement is a modern way for people to share their story of living with a bleeding disorder. To date, the use of this methodology with the bleeding disorder population has not been documented in the literature. Participants in this project reported satisfaction with being a part of the project. The project’s authors reported that it was a positive and creative way to create more awareness on a personal level about bleeding disorders and they plan to repeat the project in the future.
Quality Improvement: An Initiative to Foster Mental Health Wellness among a Hemophilia Treatment Center Patient Population
The impact of face-to-face social work meetings in bleeding disorder care
HemoFOCUS Screener for Inattention, Hyperactivity and Impulsivity: A Quality Improvement Intervention for Children with Severe Hemophilia
Pain assessment and treatment in bleeding disorder care: The need for social work specific education
Objective:
Persons with bleeding disorders experience pain in association with needle pokes, joint and muscle bleeds and permanent tissue damage. The impact of this pain on patients can include time off school or work, a change in career, income, stress, mental health concerns and change in relationships. Comprehensive pain management includes strategies from the “Four P’s of Pain Management” which include pharmacological, physical, psychological and prevention.
The aim of the project was to examine current psychological knowledge and management of pain within our patient population. This study asked the following research questions: 1) What is currently understood about pain and bleeding disorder care among social workers (CSWHC)? 2) What specific pain knowledge and training is prioritized by social workers in Hemophilia Treatment Centres?
A scoping review was conducted concurrently with the qualitative study. Medline and SocIndex were searched with the terms “social work” and “pain management” and a second search was conducted with the term “social work and hemophilia/von willebrand's or platelet disorders”. A total of 105 articles were examined by three independent reviewers. Eleven articles have been included for the purpose of examining the role of social work in pain management.
Methods:
Qualitative interviews were conducted and recorded with 12 social workers from the CSWHC between September 2018 and February 2019. Five provinces were represented. Social work participants were deployed within paediatric, adult or within combined clinics. The interviews were approximately 20-45 minutes. Transcribed interviews were coded with NVivo by two independent reviewers with Thematic Analysis.
Summary:
Social workers identified the roles of social work to include completion of psychosocial assessments and meeting the practical needs of patients, while supporting patients in medical decisions. Barriers to pain management and the impact of pain on patients were described as having an impact on individuals and families. Social workers also discussed their understanding of acute and chronic pain in patients, which has indicated an increase of knowledge is required. Skills development in multi-dimensional nature of pain and pain assessment were determined to be most likely to produce positive impact on practice outcomes. Initial themes include hope, relationship of trust, stigma (diagnosis vs. pain), defining multidisciplinary roles.
Conclusion:
Study results, first, will contribute to the literature supporting the need for social work education for those practicing in bleeding disorder care. Secondly, they will provide recommendations for an educational pain curriculum for social workers in bleeding disorder care. This education will reflect the need for pain knowledge in acute and chronic pain dimensions which will facilitate dialog with other professionals in pain management. Pain assessment will also be a focus in order for social workers to be able to support and provide appropriate referrals for pain management.
Gender Differences in Parenting Stress and Social Support Among Hemophilia Families
Parents Empowering Parents (PEP) Community Survey: How does the community want to stay engaged, communicate, and receive information?
Behavioral Health and Substance Use Screening Practices among Hemophilia Treatment Centers
Objective:
To examine the frequency and methods used to screen patients for substance use and behavioral health disorders in Hemophilia Treatment Centers (HTC). We hypothesized that inconsistencies in methods utilized and frequency of utilization exist.
Methods:
Marshall University (MU) Physical Therapy faculty along with MU addiction education staff developed a 26-question survey using Qualtrics. The survey included questions on demographics, validated screening tools utilized, screening frequency, and team member responsible for screening. The HTC email addresses were obtained from the Hemophilia Treatment Center Directory on the CDC’s website. Following approval from MU IRB, the survey was disseminated via an online link. Descriptive analysis was performed on the data.
Summary:
Health professionals from 19 HTCs, representing 8 different regions, completed the survey. The overall response rate was 13.6%. Social workers (12, 63.2%), nurses (6, 31.6%)) and counselors/psychologists (1, 0.05%) submitted responses. On average HTCs reported 34.5% (0-92%) of their patients experience chronic pain with an average 22.4% (0-56%) receiving prescription opioids for pain management. Adverse consequences related to opioid use existed in all of the HTCs including overdose (31.5%), withdrawal symptoms (42.1%), increased dose due to tolerance (63.2%), and increased bleeding episodes (26.3%). The majority of HTCs (57.9%) reported being the primary provider of pain management for people with hemophilia (PWH). Standardized screening for substance use disorders is occurring 31.6% of the time with marijuana and illicit drugs (100%) being most commonly screened followed by alcohol and prescription drugs (83%) and tobacco at 33%. Frequency of screening for substance use varied widely from every comprehensive visit to initiation of an opiate contract to suspicion of misuse. Screening for behavioral health is more common (81.3%) with a variety of validated screening tools being utilized. Over 60% of the time, screening for anxiety and depression occurs either annually or every visit.
Conclusions:
PWH often develop chronic pain related to joint arthropathy.
Based on our findings, the incidence of chronic pain in PWH is relatively equal to the national average. HTCs are often the primary provider of pain management and are challenged to find safe treatment methods. PWH are often prescribed opioids which may place them at increased risk for potentially developing an opioid use disorder.
The presence of a behavioral health disorder may further enhance one’s risk. Although behavioral health screenings appear to be more consistently utilized in HTCs, substance use screenings are rare. Our research suggests that universal screening for substance use and behavioral health conditions should be considered, as a standard of care in HTCs, to better inform healthcare providers of patient risk, need for referral and to guide prescriber’s decision making with regard to pain management options.
Depression in hemophilia and von Willebrand using the Beck Depression Inventory
Evaluation of Joint Bleeds Using Portable Ultrasound and Its Impact on Treatment of Persons With Hemophilia in a Resource Limited Setting
Physical Therapy and Extensions for Community Healthcare Outcomes (ECHO): Western States Hemophilia Regional Project
Objective:
Report on the utilization of a multi-point videoconferencing platform, Extensions for Community Healthcare Outcomes (ECHO), in providing a clinical learning opportunity to physical therapists (PTs) involved with people with bleeding disorders (PWBD) within Western States Region Hemophilia Treatment Centers (WSR HTC).
Methods:
WSR HTC includes thirteen HTC’s located in California, Nevada, Hawaii, and Guam. Monthly one-hour evidence based case presentations with a facilitated discussion were conducted using the ECHO platform. Each session was recorded, so all the therapists invited to participate have access to the information.
Data were collected from the WSR participating PTs by using anonymous on-line surveys, Qualtrics software (Qualtrics, Provo, UT), and prior to the start of the physical therapy ECHO session and upon completion of each session.
Descriptive statistics were calculated to evaluate the educational value of presentations.
Results:
Thirteen PTs, surveyed prior to the first PT ECHO session had reported > 6 years of experience as a PT. Twenty-three percent reported < 5 years of experience working with PWBD and over half of PT surveyed had > 16 years of experience working with PWBD.
Eight topics were presented in 2018 included musculoskeletal ultrasound imaging, invasive surgery rehab outcomes for patients with inhibitors, kinesiology taping, knee arthroplasty and stiffness, iliopsoas bleeding, myofascial decompression, chronic pain, knee bleed, and ankle joint impact from bleeding. An average of nine HTC PTs attended each session (range 4 to 18). Ten (11.2%) non-HTC PTs (outpatient PTs, HTC nurse, HTC Nurse Practitioner) attended some of the PT ECHO sessions. Table 1.
Ninety-five percent of respondents reported strong agreement with the program’s educational value and appropriateness for a practicing PT. Thirty-seven (94.9%) of responses reported agreement that the PT ECHO program improved their knowledge of physical therapy and bleeding disorders. Table 2.
Conclusion:
Videoconferencing platforms such as ECHO allows PTs in the WSR HTC, who are geographically separated to successfully share clinical knowledge to facilitate best practice in the area of specialty care for PWBD.
Please see files attached for tables and figures.
Use of Return to Sport Testing for Prevention of Bleeding Episodes Following an Acute Injury in the Hemophilia Patient
Tackling a New Era of Treatment in Hemophilia A: One Institution's Experience of Integrating Emicizumab into Practice
Acute Lymphoblastic Leukemia in a Pediatric Patient with Hemophilia B: A Rare Clinical Challenge
Background:
There are no reported cases of acute lymphoblastic leukemia (ALL) in patients with hemophilia B. There is one case report of a young adult with hemophilia B and acute myeloid leukemia (1). Currently, there is no best practice recommendation for the management of patients with hemophilia B and ALL.
Objectives:
To report our experience in managing a pediatric patient with congenital hemophilia B and ALL, which presents a rare and unique clinical challenge.
Design/Method:
Retrospective chart review
Results:
This is a 2y/o male with hemophilia B diagnosed at birth from a cord blood sample showing a factor IX level <1%. Mother is a known carrier and maternal grandfather has severe hemophilia B. Patient started prophylaxis with a standard half-life product through central venous access around 8months of age, following a spontaneous wrist bleed. The schedule was 35 units/kg twice a week. He had no spontaneous joint or soft tissue bleed on this regimen. 3mo ago he presented with pain and swelling of the right wrist. He fell on an outstretched hand the day before and received 100% factor infusion. X-ray showed metaphyseal lucencies with overlying soft tissue swelling. No evidence of fracture. Due to this finding, additional labs were done. WBC 4.8K, hemoglobin 7.1 g/dL, platelets 18K and 31% blasts. Flow cytometry confirmed the diagnosis of preB-ALL. On exam, he had pallor, scattered petechiae and cervical lymphadenopathy.
Based on recovery studies and thrombocytopenia, the prophylaxis was changed to 50 units/kg every third day. The platelets are kept above 30K at baseline. For lumbar punctures, he has been corrected to 100% factor level and platelets kept above 50K. However, due to the risk of port infection with frequent accessing, he was switched to long-acting albumin fusion factor IX product on day 22 of induction. The current prophylaxis regimen is 75units/kg weekly and the schedule is adjusted to coincide with lumbar puncture days whenever needed. He has tolerated all his procedures well without increased bleeding, including end of induction bone marrow aspiration, biopsy and lumbar puncture with intrathecal chemotherapy. He is currently in remission and is in interim maintenance phase of treatment per COG protocol AALL0932.
Conclusion:
Long-acting factor IX products could potentially decrease the number of infusions and need for frequent central venous access in immunocompromised patients with hemophilia B. In addition, a higher trough level with a weekly schedule could provide better bleed control in patients with severe thrombocytopenia due to underlying malignancy. A baseline platelet count of at least 30K is recommended during treatment. More treatment guidelines need to be established.
(1) Clark C et al, Pediatric Blood and Cancer Jan 2011
NHF’s State Advocacy and the Bleeding Disorders Community
Objective:
NHF Chapters must develop, support, and sustain influential advocacy programs to protect and enhance access to health care for the bleeding disorders community.
Method:
NHF supports the development of state health care policy advocacy programs with in-kind and financial assistance. NHF’s State-Based Advocacy Coalition (SBAC) program and the standard advocacy expectations our Chapters are the pillars of a robust framework of state advocacy programs. The standard advocacy expectations provide clear expectations and identify best practices for advocacy programming. These include: an advocacy committee of staff and volunteer advocates that meets monthly, multi-year strategic planning, clear engagement with legislative and administrative policymakers, an advocacy annual budget, and more. Chapters are also expected to actively engage their network of Hemophilia Treatment Centers, and all Industry partners.
The SBAC program provides funding and in-kind support from NHF for chapters to either start an advocacy program or grow and maintain an advanced one, using the standard advocacy expectations as a guide. NHF provides extensive in-kind support for SBAC grantees, as well as assisting all other chapters with their specific advocacy needs.
Summary:
Since the inception of the SBAC program NHF has seen participation by and enthusiasm among community members in advocacy events and programming grow. Nearly 500 volunteers participated in NHF’s Washington, DC Days in recent years. Chapter run State Advocacy Days are more prevalent and well-attended. At least 1000 advocates now travel to their state Capitol each year to participate in chapter Advocacy Days. There are 15 grantees participating in the SBAC program covering 20 states. Collectively, NHF has trained more than 5,000 volunteer advocates across the country.
Increasing numbers of volunteer advocates have turned out in state capitols for several years to make their voices heard and influence health care access. In addition, NHF’s investments in public policy resources have allowed us to encourage and support state regulatory advocacy, e.g. with state Medicaid offices. These new efforts have demonstrated tangible results in the form of protecting and enhancing patients’ access to care.
The promotion and support of state chapter advocacy programs by NHF, especially among the SBAC-participating states, has advanced the public policy interests of the bleeding disorders community. Objectively, we’ve seen the increased numbers and enthusiasm of volunteer advocates. Anecdotally, there are many stories of chapters playing a key role in state health care policy,
Conclusion:
Successful state advocacy work has led to improved access to quality healthcare. Start-up and ongoing support from NHF have played a critical role in that success. The continued refinement of our Chapter standard advocacy expectations and in-kind support of NHF policy staff will continue to be key to advancing state advocacy programs uniformly around the country.
Validation of a FVIII Chromogenic Nijmegen Bethesda Assay for the Detection of Inhibitors in the Presence of Emicizumab (ACE-910)
Short-term efficacy of recombinant porcine factor VIII in patients with acquired factor VIII inhibitors
A Retrospective Study Evaluating Immune Tolerance Induction (ITI) with a Plasma-derived Factor VIII for Patients with Hemophilia A and High Titer Inhibitor
Objective:
The formation of inhibitors to clotting factors is a serious complication in hemophilia A. Immune tolerance induction (ITI) therapy remains the primary method for eradicating inhibitors. This multicenter retrospective data collection project evaluated patient- and treatment-related factors associated with outcomes following primary or rescue ITI with an antihemophilic factor (Human) concentrate in patients with hemophilia A and high titer inhibitors.
Methods:
Medical records of nine inhibitor patients treated with antihemophilic factor (human) for primary or rescue ITI therapy between January 1, 2012 and July 31, 2017 were evaluated in four US hemophilia treatment centers. Data were de-identified and analyzed descriptively. Outcome measures were defined per the International Immune Tolerance Induction Study: successful (eradication of FVIII inhibitor and normal FVIII recovery), partial success (near normal FVIII recovery), and failure.
Results:
A total of nine patients between the ages of 10 months and 39 years at time of ITI were evaluated. Six out of nine patients (66.7%) had successful ITI; three with complete success (ages 27, 32, 32 years) and three with partial success (ages 5, 5, 21 years). Three patients failed ITI (ages 1.5, 10.5, 39 years) (Table 1.) Six of the patients had a combined previous ten attempts at ITI with other products (plasma derived and/or recombinant). Of these six rescue patients, ITI with antihemophilic factor (human) was successful in one and partially successful in three.
Conclusions:
While retrospective data has limitations, real-world evidence demonstrates that ITI with antihemophilic factor (human) concentrate can be successful or partially successful in diverse populations of moderately complex patients with hemophilia A and high titer inhibitor.
rFVIIIFc for first-time immune tolerance induction therapy: interim results from the global, prospective verITI-8 study
Objective:
Immune tolerance induction (ITI) is the standard of care for inhibitor eradication and restoration of factor VIII (FVIII) responsiveness in subjects with severe hemophilia who develop high-titer inhibitors. Retrospective data support the use of recombinant FVIII Fc fusion protein (rFVIIIFc) in ITI (Carcao et al. Haemophilia. 2018) but this has yet to be confirmed in prospective studies. This study presents preplanned interim results of verITI-8 (NCT03093480).
Methods:
VerITI-8 is a single-arm, nonrandomized, open‐label, ethics-approved study of rFVIIIFc (200 IU/kg/day) for first-time ITI. Eligible subjects had a history of high-titer inhibitors (historical peak ≥5 Bethesda units [BU]/mL) and provided informed consent. The primary endpoint is time to tolerization, defined by negative inhibitor titer (<0.6 BU/mL) at two consecutive visits; incremental recovery ≥66% of expected at two consecutive visits; and rFVIIIFc half‐life ≥7 hours. ITI failure is defined as not meeting the above criteria by Week 48. This interim analysis was planned when ≥10 subjects had received ≥6 months of rFVIIIFc ITI.
Summary:
Fifteen subjects were screened as of the December 5, 2018 cutoff, while 14 subjects enrolled and had received ≥1 dose of rFVIIIFc for ITI. The median (range) age at start of ITI was 2.6 (0.8–16.0) years and historical peak inhibitor titer was 29.6 (6.2–256.0) BU/mL. Six subjects have been successfully tolerized, with a median (range) time to first negative titer, normal incremental recovery, and tolerization of 2.3 (1.7–15.6), 6.0 (4.3–28.1), and 11.7 (8.1–32.0) weeks, respectively. Seven subjects continue to receive rFVIIIFc ITI (median [range] time on ITI: 16.0 [0.1–35.6] weeks) and 1 subject has failed. No adverse events related to rFVIIIFc have been reported.
Conclusions:
Early results from this prospective/ongoing study of first-time ITI indicate that rFVIIIFc may offer rapid time to tolerization in some subjects with severe hemophilia A and high-titer inhibitors. Achieving tolerance faster can improve quality of life and reduce costs.
A survey among patients with hemophilia and inhibitors seeking treatment in non-hemophilia treatment centers
Objective:
Acute bleeds in patients with rare bleeding disorders (RBDs), including congenital hemophilia with inhibitors (CHwI), acquired hemophilia, congenital factor VII deficiency, and Glanzmann’s thrombasthenia (GT) must be treated as quickly as possible. This study evaluated the obstacles and experiences of patients with CHwI, or their caregivers, for the early treatment of acute bleeds in non-hemophilia treatment centers (HTCs).
Methods:
Patients in the United States (aged 18–65 years [or caregivers of patients <18 years]) with CHwI, who currently have or have had inhibitors in the last 3–4 years, and who sought treatment in a non-HTC, underwent an interactive online qualitative discussion over 7 days.
Summary:
The survey was completed by 23 respondents (seven patients and 16 caregivers; all patients with CHwI). Respondents were aware of the need to treat bleeds quickly, which had been taught to them by physicians and learned from experience. Delays in respondents initiating their treatment were typically due to: technical issues (e.g., 7/23 respondents had difficulty gaining access to a vein or port); delay in diagnosis (e.g., 5/23 respondents’ child does not inform caregiver of the bleed); convenience (e.g., 3/23 respondents were unwilling/unable to take treatment out of the home); or financial issues (e.g., one respondent had inadequate insurance). Respondents tended to visit a non-HTC as a last resort, often due to the long distance to an HTC when emergency treatment was needed, unsuccessful pain management, or unsuccessful factor administration at home. Most patients/caregivers (20/23) reported treatment delays in emergency departments (EDs). Delays in EDs were often due to healthcare professional’s (HCP) lack of knowledge (16/23 respondents; 4 hours average wait until treatment) and four reported delays due to lack of available treatment (14 hours average wait for treatment). All patients/caregivers reported that they had dealt with uneducated/unaware HCPs, having to spend significant time educating the ED staff. Three respondents reported not waiting for treatment—partly because they chose hospitals very carefully, and because they had educated their closest hospital prior to needing an emergency service. Patients/caregivers with negative experiences reported that HCPs were unwilling to listen to them, did not seek consultation quickly, dismissed their instructions, and directed care that forced an outcome. When patients had satisfactory experiences, HCPs listened intently, immediately called an HTC/patient’s physician, and provided care in consultation. Respondents highlighted the need for HCPs education on hemophilia.
Conclusions:
Patients/caregivers are aware of the need to treat an acute bleed fast, but sometimes delay their treatment, and experience delays when attending non-HTCs. The lack of experience of HCPs in managing acute bleeds contributes to these delays. Improved education of HCPs at non-HTCs and provision of protocols or guidelines would be beneficial for patients with CHwI.
A multidisciplinary approach to the successful transition of a complex patient with severe hemophilia A with inhibitor to Emicizumab (Hemlibra®): A Case Study
Objective:
Demonstrate the success of collaborative efforts between the specialized multidisciplinary Infusion Pharmacy Provider (IPP), the prescriber, patient and payer, in achieving improved outcomes.
Methods:
A Case Study including chart review, cost analysis, and interviews with patient and prescriber.
Summary:
Patient is a 23-year-old male with severe hemophilia A and an inhibitor, followed by a Hemophilia Treatment Center (HTC). Patient developed a high titer inhibitor with a Bethesda Titre of 1000 BU/ml as a child. Several complex treatment plans including: Immune Tolerance Therapy (ITT) utilizing plasma derived and recombinant factor products, immunosuppressive therapy, and prophylaxis with bypassing agents failed. Complications with implanted ports resulted in hospitalizations and replacement of approximately twenty ports. Numerous hospitalizations for uncontrolled bleeding episodes and pain management contributed to a disruptive childhood/adolescence and suboptimal quality of life for the patient and family.
Patient was unable to attend school regularly, develop socially, or participate in normal age-appropriate activities. Repeated uncontrollable bleeding episodes led to the development of target joints and hemarthrosis. The complex nature of the patient’s treatment regimen, his psychosocial issues, bleed history, and cost of therapy resulted in frequent communication and collaboration between all stakeholders to maximize therapy outcomes.
Inhibitors presents a significant management challenge.2 Emicizumab (Hemlibra®) was approved for the treatment of hemophilia A with inhibitors in November 2017. Well in advance of the transition, the IPP and prescriber discussed the benefits with the patient. Although understandably reluctant due to his history of failed therapies, the patient agreed to try Emicizumab. Initial doses were administered at the IPP’s Alternate Infusion Suite (AIS) under clinical observation, per prescriber’s request. The patient and caregiver received extensive education regarding potential adverse events, self- administration, and bleed treatment regimen during these visits.
Conclusion:
The coordination of care, communication, and goal alignment by all stakeholders resulted in positive outcomes for this patient. Following eighteen months of therapy with Emicizumab, the patient reports improved over-all quality of life as evidenced by his ability to maintain employment, attend college, and engage in social events/ activities. Twenty-two hospitalizations in the twelve months prior to changing therapies decreased to one in the eighteen months after transitioning. His bleeding events decreased from six to eight bleeds per month to one bleed in the past eighteen months and this bleed was attributed to a missed dose. Education on the importance of adhering to prescribed dosing schedule was reinforced by both the IPP and HTC. His port has been removed. Along with his significant increase in quality of life, the dramatic decrease in overall cost of care will be highlighted.
Navigating the Emergency Department: A Collaboration Among Hemophilia Treatment Center Staff, Emergency Department Staff & Bleeding Disorder Chapter Staff
The Positive Impact of CME on Healthcare Providers’ Knowledge of Gene Therapy Studies in Hemophilia
Online CME as a Tool to Increase Clinicians’ Knowledge of Clinical Trial Data for Gene Therapy in Hemophilia
Four-year safety and efficacy of N8-GP (ESPEROCT®) in previously treated adolescents/adults with hemophilia A in the completed pathfinder 2 trial
Objective:
The adolescent/adult pivotal phase 3 pathfinder 2 trial assessed N8-GP (turoctocog alfa pegol, ESPEROCT®) use for routine prophylaxis and treatment of bleeds in previously treated patients (PTPs).
Methods:
pathfinder 2 was a multi-center, multi-national, single-arm study evaluating safety, efficacy and pharmacokinetics. Adolescents/adults (aged ≥12 y) with severe hemophilia A were administered prophylaxis (50 IU/kg Q4D) in the main phase with option for eligible patients (0-2 bleeds in prior 6 months) to randomize (2:1) to 75 IU/kg Q7D or 50 IU/kg Q4D during extension 1 (24 weeks) and continue treatment into extension 2. An on-demand group was included throughout. Current analysis covers January 2012 through December 2018.
Summary:
Of the 186 PTPs (including 46 [25%] from the US) enrolled in the main phase, 150 (81%) started extension 1, 139 (75%) completed extension 1, and 128 (69%) completed the study. Mean age was 31.1 years, weight 75 kg and BMI 24.3.
The complete trial covers 785 patient-years of treatment (66,577 exposure days [ED]) during which there were 2,758 bleeds, including 1,807 (66%) spontaneous bleeds and 1,735 (63%) joint bleeds. Twelve patients treated on-demand for a mean 3.1 years reported nearly half of all bleeds (1,270, 46%), including 971 (54%) spontaneous bleeds and 627 (36%) joint bleeds. Hemostatic efficacy was rated excellent/good in 2,470 (90%) episodes; 2,614 bleeds (95%) were treated with 1-2 injections.
Of 175 patients on prophylaxis, 55 of 110 eligible were randomized in extension 1. For 177 patients treated with 50 IU/kg Q4D prophylaxis for 613 years (57,723 ED), 126 (71%) experienced 1,312 bleeds. For 61 low-bleed patients with 134 years (7,255 ED) on 75 IU/kg Q7D prophylaxis, 53 (87%) experienced 176 bleeds. Median ABRs are shown in the TABLE.
| 50 IU/kg Q4D | 75 IU/kg Q7D | |
| n | 177 | 61 |
| Mean treatment | 3.5 years | 2.2 years |
| Median ABR | 0.8 | 1.7 |
N8-GP mean trough levels were 3.1 IU/dL on 50 IU/kg Q4D and 1.0 IU/dL on 75 IU/kg Q7D.
A total of 1,827 adverse events were reported over 785 exposure years, including 63 serious adverse events. One patient with an intron 22 inversion developed a low-titer inhibitor at 93 ED and was withdrawn when it progressed to >5 BU. Non-neutralizing anti-PEG antibodies were seen at baseline in 12 patients (6.5%) prior to first N8-GP exposure and 11 (5.9%), who had negative anti-PEG at baseline, had positive antibodies after exposure.
Conclusion:
These data support the safety and efficacy of N8-GP in a controlled phase 3 trial setting in adolescents/adults. Prophylaxis with N8-GP with a consistent dose/interval (50 IU/kg Q4D) was effective in preventing bleeds; extended dosing was evaluated as successful for a subgroup of low-bleed patients. No significant safety issues were identified.
Five-year safety and efficacy of N9-GP (REBINYN®) in previously treated children with hemophilia B in the ongoing paradigm 5 trial
Objective:
The ongoing pediatric phase 3 paradigm 5 trial is assessing N9-GP (nonacog beta pegol, REBINYN®) use for routine prophylaxis and treatment of breakthrough bleeds in previously treated children with hemophilia B (FIX ≤2%). This analysis presents 5-year safety and efficacy data in a group of children treated with weekly prophylaxis to a higher mean FIX trough (≥15%).
Methods:
paradigm 5 is a multinational, single-arm study evaluating safety, efficacy, and pharmacokinetics. Children (aged ≤12 years at enrollment) were administered weekly prophylaxis (N9-GP 40 IU/kg) through a 52-week main phase followed by an ongoing extension study. Mild/moderate bleeds were treated with 40 IU/kg. Prophylaxis, bleed treatments, and hemostatic efficacy were captured in electronic diaries. Current analysis extends from May 2012 through October 2018.
Summary:
Of the 25 children enrolled in the main phase (12 ages 0-6, 13 ages 7-12), 24 completed the main phase and 22 entered the extension (11 per age group). At the time of this analysis, 17 remain in the trial. No patients withdrew due to adverse events. Ten participants remaining in the trial have become adolescents (mean 2.6 adolescent-years of exposure).
The cumulative exposure in the study was 116 patient-years (6,194 exposure days). The median (range) time in study was 5.2 (0.2-6.1) years representing 290 (10-325) N9-GP doses per patient. The median/mean prophylactic dose was 43.1 IU/kg/wk.
A total of 573 adverse events were reported, including 4 serious adverse events, all of which were considered unlikely related by the investigator. No patients developed anti-FIX inhibitory antibodies (primary endpoint). There were 7 medical events of interest, including 6 allergic reactions (no anaphylaxis).
Age-related increase in trough FIX levels was seen; the mean FIX trough levels were 0.179 IU/mL (overall), 0.166 IU/mL (younger), and 0.192 IU/mL (older). Mean PEG plasma concentration reached steady state after ~6 months.
Overall, 20 patients (80.0%) experienced 115 bleeds, the majority of which were traumatic (64%) or spontaneous (33%) and in joints (43%). Most (93%) were treated with 1-2 doses with 89% rated as excellent/good. Median individual ABRs are shown in the TABLE; 64% of patients were spontaneous-bleed-free throughout the study.
| Median ABR | Age 0-6 | Age 7-12 | Total |
| Overall | 0.41 | 0.99 | 0.66 |
| Spontaneous | 0.00 | 0.00 | 0.00 |
| Traumatic | 0.41 | 0.50 | 0.47 |
Conclusion:
These data support the safety and efficacy of N9-GP 40 IU/kg weekly over a median of 5 years in a controlled phase 3 trial setting in children. N9-GP prophylaxis with a trough of ~18% was effective in preventing bleeds with low reported ABR and with 64% of patients reporting no spontaneous bleeds during the entire study period. No unexpected safety issues were identified.
Modeling of Daily Administration of N8-GP (ESPEROCT®) vs Standard Half-life FVIII for Patients With Hemophilia A Participating in Sports Activities
Objective:
Daily administrations of FVIII products are considered useful for providing high FVIII coverage for active patients with hemophilia A. This analysis was performed to determine the daily dose levels required of N8-GP (turoctocog alfa pegol, ESPEROCT®) vs standard half-life (SHL) FVIII (N8, turoctocog alfa, Novoeight®) to normalize risk of activity-related bleeding for patients with hemophilia participating in daily sports activities (practices, games) of varying risk profiles.
Methods:
Patients with hemophilia engaging in physical activity have associated increased bleeding risk with sports that have increased potential for contact injuries as classified by Broderick et al (JAMA. 2012): Class 1 - no contact (eg, swimming); Class 2 - contact might occur (eg, basketball); and Class 3 - inevitable contact (eg, American football). To normalize the risk of bleeding, nominal targets of FVIII activity levels for at least 2 h/d based on Broderick et al were chosen: above 30% (Class 1), above 50% (Class 2), and above 70% (Class 3).
Pharmacokinetic (PK) simulations were performed using a one-compartment model with first-order elimination. FVIII PK profiles were simulated for the extended half-life (EHL) N8-GP based on the pathfinder 1 PK trial showing 60% prolonged half-life compared with prior SHL FVIII. For PK simulations of an SHL, N8 was used due to 104⁰F stability with PK based on the guardian clinical trial program.
Summary:
Daily doses to sustain at least 2 h/d of 30%/50%/70% activity were estimated for N8-GP (9, 15, and 21 IU/kg) and N8 (14, 23, and 33 IU/kg). Steady-state PK profile simulations of once-daily administration are shown in the Figure.
| Broderick Class 1 (>30%) | Broderick Class 2 (>50%) | Broderick Class 3 (>70%) | |
| N8-GP | |||
| Daily (weekly), IU/kg | 9 (63) | 15 (105) | 21 (147) |
| Peak/trough activity | 32%/13% | 54%/21% | 76%/29% |
| Difference from 50 IU/kg Q4D | -28% | 21% | 69% |
| N8 | |||
| Daily (weekly), IU/kg | 14 (98) | 23 (171) | 33 (231) |
| Peak/Trough activity | 36%/6% | 59%/10% | 87%/14% |
| Difference from 25 IU/kg QD | 13% | 97% | 166% |
| N8-GP vs N8 | |||
| Utilization, IU/kg | -36% | -39% | -36% |
Conclusion:
Experience with routine prophylaxis with EHL/SHL FVIIIs towards guideline recommended >1% activity does not readily translate to HCP understanding of the PK with high daily or much higher every-other-day administration required to minimize risk for the active patient. With a 1.6x (60%) prolongation in half-life for adolescents/adults, this model shows daily N8-GP to be a more efficient strategy compared with daily SHL FVIII (N8) to cover the active patient; N8-GP achieves higher trough levels with a smaller increase in overall factor consumption compared with standard prophylaxis with SHL FVIII.
Five-year safety and efficacy of N8-GP (ESPEROCT®) in previously treated children with hemophilia A in the completed pathfinder 5 trial
Objective:
The completed pediatric phase 3 pathfinder 5 trial assessed the safety and efficacy of N8-GP (turoctocog alfa pegol, ESPEROCT®) use for routine prophylaxis and treatment of breakthrough bleeds in previously treated children.
Methods:
pathfinder 5 was a multicenter, multinational, single-arm study evaluating safety, efficacy, and pharmacokinetics. Children (aged <12) with severe hemophilia A were administered prophylaxis (target 60 [50-75] IU/kg twice weekly) in the main phase (26 weeks) followed by an extension phase. Current analysis covers study initiation (February 2013) through completion (September 2018).
Summary:
Of the 68 children (34 aged 0-5, 34 aged 6-11) enrolled, 63 completed the main phase and 62 completed the extension. Most (95%) were previously on prophylaxis. The total study period amounted to 306 patient-years (32,138 exposure days); median (mean) patient exposure was 4.9 (4.5) years.
Overall, 838 adverse events (AEs) were reported; 18 serious AEs included 2 possibly/probably related to N8-GP (severe allergic reaction [1] and increasing bleeding symptoms [1]). No inhibitor development was observed in the trial. Two AEs resulted in withdrawal; a third patient with severe allergic reactions (after 4 doses) that resolved after 2 hours without any treatment met preestablished withdrawal criteria. There were no anti-PEG antibodies of clinical significance; however, 21 (31%) patients had anti-PEG antibodies at baseline (prior to exposure), and 1 patient had a single positive measurement after exposure at a titer <1.
Overall, 55 patients (81%) reported 330 bleeds during the study; most were traumatic (67%). The success rate for hemostasis was 84% (excellent/good); 71% were treated with 1 injection, and 88% of patients were successfully treated with 1-2 injections. Median (mean) utilization for bleeds was 68 (95) IU/kg.
Median ABRs are shown below; estimated mean ABR was 1.1. Forty-seven percent of children had no spontaneous bleeds throughout the trial. Of 13 children with 17 target joints at baseline, 77% (main phase) and 46% (complete trial) reported no bleeds in their target joints. For those previously on prophylaxis, the mean observed ABR was 2.3 compared with the historical ABR of 6.4. The mean prophylaxis dose was 64.7 IU/kg with an interval of 3.5 days.
| Median ABR | Age 0-5 y | Age 6-11 y | Total |
| Overall | 0.6 | 0.9 | 0.8 |
| Spontaneous | 0.1 | 0.2 | 0.2 |
| Traumatic | 0.3 | 0.8 | 0.5 |
N8-GP prolonged single dose half-life by 1.9x compared with the child’s prior FVIII product. The mean trough levels on twice-weekly dosing were 0.019 IU/mL (0.016 ages 0-5, 0.024 ages 6-11).
Conclusion:
These data support the safety and efficacy of N8-GP in a controlled phase 3 trial setting in children. Prophylaxis with N8-GP using a consistent dose/interval (65 IU/kg twice weekly) was effective in preventing bleeds. No unexpected safety issues were identified.
Factor VIII deficiency is associated with abnormal brain volumes
Three-year efficacy and safety results from a phase 1/2 clinical study of AAV5-hFVIII-SQ gene therapy (valoctocogene roxaparvovec) for severe hemophilia A (BMN 270-201 study)
Objective:
Hemophilia A (HA) is an X-linked disorder caused by mutations in the gene encoding Factor VIII protein (FVIII). Gene therapy is increasingly being viewed as a viable treatment option for hemophilia. Herein, long-term clinical safety and efficacy are presented from a Phase 1/2 study of an AAV-mediated gene therapy for severe HA.
Methods:
Valoctocogene roxaparvovec is an adeno-associated virus-mediated gene therapy that delivers a functional, codon-optimized, B domain-deleted, human FVIII gene under the control of a liver-specific promoter (AAV5-hFVIII-SQ). An ongoing Phase 1/2 study continues to evaluate the safety and efficacy of valoctocogene roxaparvovec in thirteen males with severe HA. Study participants received a single intravenous injection of valoctocogene roxaparvovec at one of two dose levels (6×1013vg/kg, n=7; 4×1013vg/kg, n=6).
Summary:
Participants who received 6×1013vg/kg valoctocogene roxaparvovec showed a reduction in annualized bleeding rate (ABR) of 96%, from a pre-treatment median(mean) of 16.5(16.3) to 0.0(0.7) at year three. Participants demonstrated an absence of target joints and target joint bleeds, with 86% experiencing zero bleeds requiring FVIII treatment. ABR diminished by 92% in 4×1013vg/kg participants, from a pre-treatment median(mean) of 8(12.2) to 0(1.2) at year two. Sixty-seven percent of 4×1013vg/kg participants experienced zero bleeds requiring FVIII treatment.
FVIII usage demonstrated a reduction from pre-treatment median(mean) of 139(137) infusions to 0(5.5) at year three in 6×1013vg/kg participants, and from 156(147) to 0.5(6.8) at year two in 4×1013vg/kg participants.
In 6×1013vg/kg participants, FVIII levels reported by chromogenic assay reached a median(mean) of 60.3(64.3), 26.2(36.4), and 19.9(32.7) IU/dL at the end of one, two, and three years post-infusion, respectively. In 4×1013vg/kg participants, FVIII levels reported by chromogenic assay reached a median(mean) of 22.9(21.0) IU/dL and 13.1(14.7) IU/dL at the end of one and two years post-infusion, respectively. Although FVIII levels were measured and will be presented using both the chromogenic substrate assay and the one-stage assay, chromogenic assay results appear to more accurately represent the true level of circulating FVIII.
The safety profile of valoctocogene roxaparvovec remains favorable and unchanged, with transient, asymptomatic ALT elevations and no FVIII inhibitor development reported to-date.
Conclusions:
Following a single administration of valoctocogene roxaparvovec, participants showed sustained, clinically relevant FVIII activity that reduced self-reported bleeding and exogenous FVIII replacement use at 156 weeks and 104 weeks post-administration in 6×1013vg/kg and 4×1013vg/kg dose cohorts, respectively.
Baseline patient characteristics in ReITIrate: A prospective study of rescue ITI with recombinant factor VIII Fc fusion protein (rFVIIIFc) in patients who have failed previous ITI attempts
Objective:
Inhibitor development is the most serious complication of hemophilia A therapy. Immune tolerance induction (ITI) is the gold standard for inhibitor eradication, restoring factor VIII (FVIII) responsiveness. Retrospective data on ITI therapy using rFVIIIFc have been reported (Carcao et al. Haemophilia. 2018). The ReITIrate study (NCT03103542) was designed to prospectively evaluate success of rescue ITI with rFVIIIFc.
Methods:
ReITIrate, a prospective, interventional, multicenter, open-label study, enrolled patients with severe hemophilia A and inhibitors, who failed previous ITI attempts. The primary purpose is to describe the outcome of ITI performed with rFVIIIFc (200 IU/kg/day) within a maximum of 60 weeks. Here, patient baseline characteristics are reported using descriptive statistics and listings.
Summary:
Sixteen subjects were included in the study between November 2017 and December 2018. The median (range) age at study enrollment was 7.5 (2–46) years. Seven subjects had a known family history of inhibitors. The median (range) number of prior ITI attempts was 1 (1–3) and the median (range) total ITI duration was 51.5 (12–155) months. All subjects had previously received high-dose ITI, with 3 subjects receiving plasma products, 6 subjects receiving recombinant products, and 7 subjects receiving both recombinant and plasma products for previous courses of ITI. Four subjects received prior immunomodulatory therapy. The median (range) inhibitor titer at screening and historical peak were 11 (0.9–635) BU/mL and 127 (8–3000) BU/mL, respectively. During the 12 months prior to enrollment, the median (range) number of bleeds was 5 (0–24); 11 subjects used activated prothrombin complex concentrate (aPCC) for treatment of bleeds, 5 subjects received recombinant factor VIIa (rFVIIa), and 1 subject each received FVIII/von Willebrand factor, recombinant FVIII, and tranexamic acid. Twelve subjects received prophylaxis with bypassing agents during this period (10 aPCC, 1 rFVIIa, and 1 both products).
Conclusions:
This is the first prospective study describing rescue ITI with an extended half-life recombinant FVIII product. Enrolled subjects had multiple risk factors for poor ITI outcomes and a long duration of previous ITI. There is an unmet need for successful tolerization in such patients, allowing regular FVIII prophylaxis and potentially leading to improved clinical outcomes and quality of life.
Clinical Study to Investigate the Efficacy and Safety of Wilate During Prophylaxis in Previously Treated Patients With Von Willebrand Disease (VWD)
Objectives:
This study has a primary objective to determine the efficacy of VWF/FVIIII concentrate (Wilate) in the prophylactic treatment of previously treated patients with type 3, type 2 (except 2N), or severe type 1 VWD.
Secondary objectives of this study will be to collect data to 1) Assess the VWF:Ac and VWF:Ag incremental IVR of VWF/FVIIII concentrate over time, 2) Assess the safety and tolerability of VWF/FVIIII concentrate in this indication.
Also the study will examine, the efficacy of VWF/FVIIII concentrate in the treatment of breakthrough bleeding episodes (BEs), and in surgical prophylaxis, as well as the quality of life (QoL) during prophylaxis with VWF/FVIIII concentrate.
Methods:
The study is planned to enrol 28 PTPs aged ≥6 years and with VWD type 1, 2A, 2B, 2M, or 3. Eligible patients must be receiving on-demand treatment with a VWF-containing product, with at least 1, and an average of ≥2, documented spontaneous BEs per month in the preceding 6 months requiring treatment with a VWF-containing product. This will be assessed as part of a run in observational study to collect bleeding rate prior to the start of prophylaxis.
From the beginning of the study, patients will receive prophylactic treatment with VWF/FVIIII concentrate for 12 months and record all BEs in a patient diary. Based on these data, the frequency of BEs and the annualized bleeding rate (ABR) under prophylactic treatment will be calculated.
Treatment efficacy of BEs will be assessed by the patient (together with the investigator in case of on-site treatment) using a 4-point scale (excellent, good, moderate, none)
In case patients undergo surgeries, efficacy of VWF/FVIIII concentrate will be assessed at the end of surgery by the surgeon and at the end of the postoperative period by the haematologist. In both cases, predefined assessment criteria will be used. In addition, an overall assessment of efficacy will be made at the end of the postoperative period by the investigator.
Summary/conclusions:
Prophylactic treatment in other congenital bleeding disorders is widely accepted as the standard of care to prevent bleeding and preserve quality of life in patients. This form of treatment in VWD is not well characterized prospectively as yet. This study will provide data on the efficacy of prophylactic treatment in reducing the rate of bleeding and on the impact of prophylaxis on the quality of life in VWD patients.
Clinical experience with BIVV001, the first investigational factor VIII (FVIII) therapy to break through the von Willebrand factor (VWF) ceiling in hemophilia A
AMT-061 (AAV5-Padua hFIX variant) an Enhanced Vector for Gene Transfer in Adults with Severe or Moderate-Severe Hemophilia B: Follow-up up to 9 Months in a Phase 2b trial
No evidence of germline transmission of vector DNA following intravenous administration of AAV5-hFIX to male mice
Background:
Recombinant adeno-associated viruses (rAAV) are replication-deficient, non-integrating viruses commonly used as vectors for gene therapies currently in clinical development. Systemic administration of gene therapy raises the possibility of vertical germline transmission of the vector DNA.
Aim:
Here, we investigated the possibility of germline transmission following IV administration of an AAV serotype 5 vector designed for the liver-directed expression of human Factor IX which is being studied in clinical trials for hemophilia B.
Methods:
Since hemophilia B predominantly occurs in male patients, paternal germline transmission was investigated in mice in a GLP compliant study, according to current gene therapy guidelines (EMEA/273974/2005). Male C57Bl/6 mice (n=15) each received a single intravenous infusion of 2x1014 gc/kg AAV5-hFIX and were mated 6 days later with untreated female mice (n=30). On day 20 post-treatment, males were sacrificed and the seminal vesicle, epididymis, testes and a sperm sample were collected. Successfully mated females were necropsied on day 17 of gestation and the uterus, placenta and fetuses collected for each female. Each fetus was examined for viability and externally visible abnormalities. All samples were analyzed for vector DNA by QPCR.
Results:
No effect of treatment was observed on male mating performance, fertility indices, maternal body weight, food consumption, pregnancy performance, external fetal abnormalities, or fetal weights. Vector DNA levels of up to 2x106 gc/μg gDNA were detected in male reproductive tissues (epididymis, seminal vesicle, sperm, and testes), but not in female uterus, placenta and offspring. Although vector DNA was detected in the reproductive tissues of males, there was no evidence of transmission of vector DNA to female reproductive tissues or to the fetuses.
Conclusion:
The risk of paternal germline transmission following AAV5-based vector administration is therefore considered to be low.
Bleeding types and treatments in patients with von Willebrand disease before and after diagnosis
Objective:
Von Willebrand disease (VWD) is the most common inherited bleeding disorder, however, initial diagnosis and subsequent management of patients after diagnosis remains a challenge. The aim of this study was to characterize the specialists who are treating patients before and after diagnosis of VWD. We also identified the most common bleeding types and the treatments given to these patients.
Methods:
This retrospective study analysed data from a US medical claims insurance database (IQVIA PharMetrics Plus Database) for patients who had made insurance claims for VWD (International Classification of Diseases, ninth edition [ICD-9] code: 286.4). The claims were made from January 01, 2006 to June 30, 2015. Patients with ≥2 medical claims for VWD and who were continuously enrolled for a 2-year period before and after their 1 st VWD claim were included in this study. Descriptive statistics were used to summarize patient demographic and clinical characteristics, which included bleed types, treating physician specialty, and type of VWD treatment, in both the pre- and post-diagnosis periods.
Summary:
A total of 3,756 patients were included: 73% were female, and the median age at VWD diagnosis was 34 years old (age range 2–82 years). Pre-diagnosis, the top 3 treating physician specialties were hospitalists (22%), primary care physicians (14%) and obstetrician-gynecologists (13%). Post-diagnosis, the top 3 treating physician specialties were hospitalists (14%), primary care physicians (8%) and obstetrician- gynecologists (10%). Only 6% of patients saw a specialist hematologist before VWD diagnosis for a bleeding event and this decreased to 3% after diagnosis. The number of claims made by patients for bleeding events decreased from 45% pre-diagnosis to 34% post-diagnosis. In females, heavy menses were the most common bleed type, representing 29% of pre-diagnosis claims and 21% of post-diagnosis claims. In males, epistaxis was the most common bleed type, representing 13% of pre-diagnosis claims and 8% of all post-diagnosis claims. Overall, insurance claims for medical treatments associated with VWD increased from 19% pre-diagnosis to 27% post-diagnosis. The most prescribed treatments in women were oral contraceptives, desmopressin (DDAVP) and aminocaproic acid (ACA) (pre-diagnosis: 18%, 5% and 2%, respectively; post- diagnosis: 20%, 11% and 5%, respectively). In men, the most prescribed treatments were DDAVP, ACA and von Willebrand factor (VWF) concentrates (pre-diagnosis: 5%, 4% and 2%, respectively; post-diagnosis: 9%, 6% and 4%, respectively).
Conclusions:
These data show an overall reduction in the frequency of bleeding event insurance claims after VWD diagnosis. This was coupled with an increase in treatment insurance claims for DDAVP, ACA and VWF after diagnosis. These results highlight the importance of diagnosis of VWD and treatment optimization in these patients. Also, only a minority of patients received care from a hematologist, which may impact treatment and care.
Correcting Bleeding Disorders Using Blood Clotting Factors Produced in vivo by Encapsulated Engineered Allogeneic Human Cells
Optimizing signal strength and suppressive potential of FVIII specific CAR Tregs for tolerance induction in a murine model of hemophilia A
A Unique Combination of Severe Congenital Factor XIII Deficiency and Type 2M Von Willebrand Disease – A Case Report
Factor XIII deficiency is classified under rare bleeding disorders and is in fact, the rarest with an incidence of 1 in 2 to 3 million births and is inherited in an autosomal recessive manner. On the other hand, Von Willebrand Disease (VWD) is the most common bleeding disorder occurring in about 1% of the US population. Type 2M Von Willebrand Disease (Type 2M VWD), a subtype of Type 2 VWD, has an autosomal dominant pattern of inheritance and is characterized by decreased activity of Von Willebrand Factor (VWF) and its failure to interact with platelets. This report describes an interesting case of an individual with a combined severe congenital factor XIII deficiency and a recently diagnosed Type 2M VWD.
A 43 year old male diagnosed with severe congenital factor XIII deficiency at birth following umbilical stump bleeding has been on regular prophylaxis since 2007. Frequent breakthrough bleeds despite receiving prophylactic infusions of Factor XIII concentrate prompted a re-evaluation of the patient’s coagulation profile. Clinical laboratory parameters consistent with a diagnosis of Type 2M VWD were observed in addition to his underlying severe Factor XIII deficiency. DNA sequencing identified a novel missense variant p.Arg1136Trp (c.3406C>T) as the possible causative mutation.
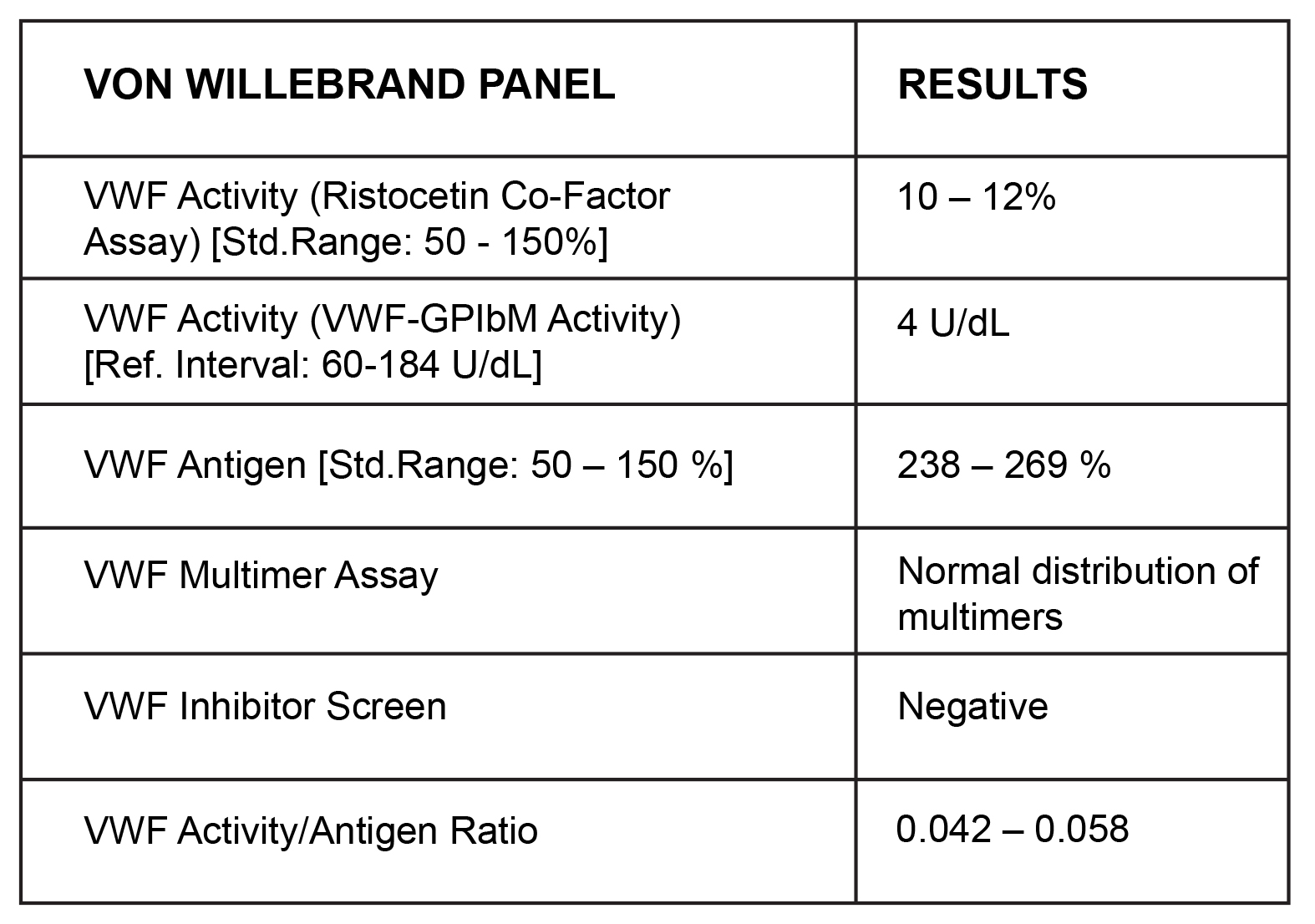
Weekly blood draws overlapping 3 cycles of prophylactic infusions of Tretten (recombinant FXIII-A subunit concentrate) revealed peak and trough factor XIII activity levels of ~80% and ~10% respectively. The subject had a largely sedentary life style over this period of 12 weeks indicating, based on previous experience, that any moderate to vigorous physical activity could necessitate maintenance of a higher trough level. Additionally, the potential utility of VWF concentrates to effectively manage the second hemostatic defect and prevent breakthrough bleeds needs to be explored.
To our knowledge, this is the first report describing a unique combination of severe congenital factor XIII deficiency and type 2M VWD.
Surveying Nurses’ Knowledge and Confidence of Discussing Oral Health with Patients with Bleeding Disorders
PiggyBac mediated gene transfer for prevention of anti-factor VIII antibodies in hemophilia A
Impact of hemophilia on employment - Insights from the PROBE Study
Congenital afibrinogenemia: a case report of perioperative hematological management during difficult orthopedic surgery
Head-to-head pharmacokinetic comparisons of N9-GP with standard FIX and rFIXFc in patients with hemophilia B
Annual Bleed Rates Compared Before and After Changing to Extended Half Life Products in Home Infusion Patients with severe hemophilia
NHF’s State Based Advocacy Coalitions (SBAC) Program
Long-term clinical outcomes of rFIXFc prophylaxis in adults 50 years of age or older with severe hemophilia B
Long-term clinical outcomes of rFVIIIFc prophylaxis in adults 50 years of age or older with severe hemophilia A
HOPE-B: Study design of a Phase III trial of an investigational gene therapy AMT-061 in subjects with severe or moderately severe hemophilia B
Joint health in patients with hemophilia A: analysis from the CHOICE survey
Do Hemophilia Treatment Centers Want Or Need A Regional Ethics Committee?
Identification of Challenges and Coping Strategies in the Management of Bleeding Disorders, From the Patient Perspective
Objective:
We conducted a pilot study using a single open-ended question to elicit patient-perceived challenges and management strategies in individuals with bleeding disorders. The de-identified responses and themes expressed in this study were analyzed. The identification of perceived challenges and management strategies in individuals with bleeding disorders offers the opportunity to improve value based care.
Methods:
This retrospective, cross-sectional cohort study used a sample of convenience at The Hemophilia Center at OHSU. Study population included 20 participants. Inclusion criteria included: ages seven to eighty nine, diagnosed with a bleeding disorder and seen during any outpatient Hemophilia Center clinic visit between March 1, 2017 and April 30, 2018. Participants were included if, during the course of their clinical care, they answered the question, “What is the most significant (or greatest) challenge you have in managing your bleeding disorder and what do you do about it?” Data extracted included question response, age, type and severity of bleeding disorder. Responses were analyzed for themes by the investigators and using qualitative data analysis software. Coded demographic data was correlated.
Summary:
Seven challenge themes were identified: activity restrictions, infusions, emotion/stress, pain, future plans, education and access. Management themes included: self-advocacy, parent directed, activity modification or avoidance, acceptance, inquiry, asking for assistance, planning ahead, resiliency, and peer supports. Younger participants’ (9-17 years) challenges included activity restriction and infusions with management strategies of self-advocacy, activity avoidance and modification. Participants aged 18-57 years highlighted challenges with access to care, infusions, emotion/stress, pain, education and future planning. Management strategies in this group were focused on acceptance, planning ahead, peer support and resiliency. Analysis based on severity of bleeding disorder revealed that subjects with severe hemophilia reported infusions and activity restriction as their most significant challenge, with self-advocacy and activity modification management strategies. Participants with moderate hemophilia reported challenges centered on activity restriction and education of peers, with management strategies being self-advocacy and planning ahead. There were no differences in themes identified when analyzed based on type of bleeding disorder.
Conclusions:
This study characterizes the unique challenges and management strategies described by individuals with bleeding disorders. The themes highlighted the importance of patient voice and can be used to inform individual care decisions.
Bringing families affected by Factor XIII deficiency together for a novel educational program
Factor X deficiency consumer education program’s inaugural year
Giving men with vwd a voice
Introduction and Objectives:
Von Willebrand disease (VWD) is the most common bleeding disorder, affecting men and women equally. Despite this, awareness surrounding VWD is low. Outreach efforts often only target women, leaving many to assume VWD does not affect men. To begin changing this perception, the National Hemophilia Foundation (NHF) conducted a needs assessment to understand men’s awareness of VWD and experience getting a diagnosis for future programming.
Materials and Methods:
On behalf of NHF, The Harris Poll conducted a nationally representative online survey. From 2015 to 2016 1,002 adult men in the US were interviewed to learn about their health behaviors and awareness of VWD. A second online survey was conducted by NHF targeting men who were diagnosed with VWD to learn about their path to diagnosis and the impact of VWD on their lives. This survey was given to adult men in the US from 2016 to 2017 and 49 responses were included in the analysis.
Results:
The Harris Poll found that only 28% of men say they are aware of VWD and 68% are not sure of the symptoms. Medical providers (69%) are the main sources the men turned to for information about their health, followed by internet sources (40%). The second survey found an average of 8 years from first symptoms to final diagnosis, with almost 60% of respondents diagnosed at 18 years of age and older. When asked what motivated them to seek medical care, 45% cited a significant bleeding incident. Over half reported limitations to work, physical and social activity. Medical providers were one of the most common sources of information and support for men with VWD and the first place men went for information.
Conclusion:
More awareness of VWD is needed and outreach focused online and to medical providers. Diagnosed men need more education and support surrounding their disorder. NHF will continue to pursue outreach efforts and creation of resources for men with VWD.
Inhibitor Teams: building stronger connections and deeper learning
Understanding and finding symptomatic undiagnosed women
Use of platelet microcapsule hybrids loaded with factor VIII to treat hemophilia A mice
The Relationship of Hope and Self-Compassion with Quality of Life among Individuals with Bleeding Disorders
Bleeding Disorders Education Day for School Nurses
Surgical Experience in Two Multicenter, Open-label Phase 3 Studies of Emicizumab in Persons with Hemophilia A with Inhibitors (HAVEN 1 and HAVEN 2)
Persons With Hemophilia Reinforce Their Desire to be More Active: US Findings From an International Patient Survey
BIVV001 – a novel, weekly dosing, VWF-independent, extended half-life FVIII therapy: first-in-human safety, tolerability, and pharmacokinetics
Patient perspectives on the value of reduced infusion frequency and longevity of protection for prophylactic treatment of hemophilia A
Retrospective review of unplanned hospitalizations and perceived pain in children and adults with a diagnosis of factor ten deficiency receiving home infusions of commercially available factor ten
Background:
Inherited factor X deficiency is an autosomal recessive bleeding disorder with an estimated occurrence rate of 1:1,000,000¹. Historically, bleeding symptoms have been treated with topical therapies, antifibrinolytic agents, fresh frozen plasma (FFP) or plasma-derived FIX concentrates (PCCs). In 2015, the first factor X (FX) concentrate was approved in the U.S.
Objective:
This organization was interested in reviewing clinical outcomes such as perceived pain and unplanned hospitalizations of adults and children with FX disease currently being treated in the home with Coagadex®. METHODS: This organization conducted a retrospective review of a population of seven adult and pediatric patients. Patients were surveyed for pain, bleeding episodes, hospitalizations/ ER visits, dosing parameters and administration methods pre/post initiation of FX therapy. There were 3 children, 12 years old and under and four adults. Ages ranged from 5-60 years old with the average age of 27.9. There were five males and two females. The average length of treatment was 6.4 months. One patient was naïve, six converted from other therapies. Dose ranges administered by caregivers or self-infusion were 750 -2800 IU (26-61 IU/Kg). One patient was on-demand and six were administering prophylaxis therapy.
Results:
There were two converted prophylaxis patients reported pain with PCC’s and none with FX; one on-demand naïve patient stated his pain was markedly improved with prn administration of FX; four converted prophylaxis patients with no prior pain history reported no changes in pain on FX therapy. For on-demand patients treating bleeding episodes, three reported a decrease in the number of bleeding episodes, three were unchanged and one reported one additional bleeding episode. A total of ten hospitalizations or emergency room visits were reported during the six months prior to initiation of FX treatment and only one in the six months following initiation of treatment.
Conclusion:
Early recognition and home treatment with FX concentrate allows for prompt resolution of bleeding symptoms, decreased pain and decreased hospitalization or emergency room visits. Further investigation is needed to determine cost-savings for decreased hospitalization/ ER visits.References:Brown, D.L. & Kouides, P.A. (2008). Diagnosis and treatment of Inherited Factor X deficiency. Haemophilia. (14). 1176-1182. Retrieved from: https://www.hemophilia.org/sites/default/files/document/files/DiagnosisAndTreatmentOfInheritedFact…
Gender Differences in Parenting Stress and Social Support in Hemophilia Families
Real-world bleeding outcomes and adherence metrics among persons with hemophilia A and B receiving standard or extended half-life factor replacement products
Objective:
We sought to explore real-world outcomes, such as annualized bleeding rate (ABR) and markers of adherence, in persons with hemophilia A (PwHA) or with hemophilia B (PwHB) who receive standard half-life (SHL) or extended half-life (EHL) factor VIII (FVIII) or factor IX (FIX) replacement products.
Methods:
We analyzed de-identified data from the Adelphi Disease Specific Programme (DSP) database, a patient health record–based survey of hematologists in the US and 5 European countries (France, Germany, Italy, Spain and the UK). Data were collected from May–November 2017 for male patients with moderate or severe HA or HB. Outcomes in the two groups of patients (SHL vs EHL) were compared and descriptive statistics were used to summarize results.
Summary:
A sample of 595 patients with HA or HB met the inclusion criteria (US, n=123; Europe, n=472). Age, weight, and body mass index (BMI) were similar between SHL and EHL groups for PwHA and PwHB on both continents. Higher ABR was noted consistently in Europe vs the US. Hemophilia A: Analysis included 101 patients from the US (SHL, n=64; EHL, n=37) and 360 patients from Europe (SHL, n=340; EHL, n=20). The ABR was similar between both groups on both continents (median: US, 1.0 SHL and 1.0 EHL; Europe, 1.0 SHL and 1.5 EHL; mean: US, 1.3 SHL and 1.2 EHL, P=0.68; Europe, 1.8 SHL and 1.7 EHL, P=0.76). The mean of the physician-reported ‘number of doses missed of the last 10 doses’ appeared to be numerically higher in the EHL vs the SHL group in the US (mean: 0.4 SHL and 1.6 EHL, P=0.13), whereas in Europe, the trend was reversed (mean: 0.7 SHL and 0.0 EHL, P=0.29). Hemophilia B: Analysis included 22 patients from the US (10 SHL; 12 EHL) and 112 patients from Europe (91 SHL; 21 EHL). The median ABR for PwHA and PwHB in the US was 1.0 (SHL) and 1.0 (EHL), and the mean was 1.6 SHL and 0.8 EHL (P=0.25); in Europe, the median ABR was 2.0 SHL and 1.0 EHL, and the mean was 2.2 SHL and 1.6 EHL, P=0.25. The mean of the physician-reported ‘number of doses missed of the last 10 doses’ was 0.8 SHL and 0.5 EHL (P=0.63) in the US and 0.6 SHL and 0.1 EHL (P=0.33) in Europe.
Conclusions:
These preliminary real-world data, unadjusted for treatment regimen and inclusive of US and ex-US sampling, showed no clinically meaningful difference in ABR or adherence markers in PwHA or PwHB who received SHL versus EHL FVIII or FIX products. These observations may challenge assumptions regarding adherence and or clinical outcomes associated with SHL/EHL product selection among PwHA and PwHB. Further analyses should be explored.
Efficacy of on-demand treatment of bleeding episodes in hemophilia B patients with extended half-life N9-GP in pivotal trials: an in-depth analysis of treatment
Improving the screening for and evaluation of bleeding disorders in the primary care setting
An update on cognitive and behavior function in children and young adults with hemophilia: a 25-year journey from the Hemophilia Growth and Development Study to the current eTHINK study
Objective:
The Evolving Treatment of Hemophilia’s Impact on Neurodevelopment, Intelligence and Other Cognitive Functions (eTHINK) study aims to evaluate the impact of hemophilia on neurodevelopment and cognitive function through the use of validated instruments and to identify covariates that drive differences in neuropsychological performance.
Methods:
A sample of at least 510 males aged 1-21 years (~25 per age) with hemophilia A or B (any severity, with or without inhibitors) will be enrolled in a cross-sectional, non-interventional study. Following ethics review and informed consent, data collected will include a structured developmental and hemophilia history interview, a standardized neurologic examination, and a comprehensive neuropsychological assessment of cognitive/motor development (Bayley-III), intelligence (WPPSI-IV/WASI-II), attention/processing speed (CogState™), executive function (BRIEF-P/BRIEF2/BRIEF-A), mood and behavior (BASC-3), and adaptive behavior (ABAS-3). Assessments will include objective tests as well as parent and patient self-report rating scales. Z scores will be derived from published general population norms for each instrument and analyzed to develop hemophilia population specific norms. Secondary analysis for predictors of outcome will include regression modeling and chi-square tests of top vs bottom quartile responses.
Summary:
Initiated in the early 1990s under Centers for Disease Control, Maternal and Child Health Bureau, and National Institutes of Health, the Hemophilia Growth and Development Study (HGDS) evaluated the impact of hemophilia on neurodevelopment, executive function, and intelligence. The 4-year observational study enrolled 333 patients from 14 US centers, aged 6-18 years at baseline (62% HIV+), who underwent annual/semi-annual comprehensive assessments including neurologic examination, neuroimaging (MRI), and neuropsychological assessment. Results suggested that hemophilia and HIV had independent effects at baseline and follow-up. Baseline neurologic examination findings were common, as were progressive abnormalities of gait/coordination. Imaging showed baseline CNS bleeds in 12% of patients and new CNS bleeds (2% per year), which often occurred in the absence of reported head trauma. HIV+ children were more likely to show lower scores on neuropsychological assessments. Academic/adaptive skills were lower than expected based on mean IQ, and more behavioral/emotional problems were seen, including attention abnormalities related to known/silent CNS bleeds. There was a large shift in mean scores in IQ and achievement for the children with more severe hemophilia. Six small studies published between 1996 and 2009 reported impacts on academic achievement, attention, and behavior.
Conclusions:
HGDS established 25 years ago that hemophilia and HIV have independent effects on cognitive and behavioral function in children with hemophilia. Since then, standards of care in hemophilia treatment have changed significantly, but no follow-up studies have investigated whether these changes have affected the profile of neurocognitive outcomes in hemophilia. We therefore designed the eTHINK study to provide valuable insights into whether subgroups of children and young adults with hemophilia remain at risk for impaired neuropsychological outcomes.
Effects of Factor VIII Prophylaxis on Vascular Remodeling and Synovial Gene Expression Changes Associated with Hemarthrosis in FVIII-Deficient Mice
PROTECT VIII Extension Trial Interim Data: Safety of >5 Years of Treatment With BAY 94-9027
Effective Long-term Prophylaxis with BAY 94-9027 in Previously Treated Children: Interim Results of the PROTECT VIII Kids Extension Study
Objective:
BAY 94-9027 is an extended–half-life recombinant factor VIII (FVIII) product. In the PROTECT VIII Kids trial, BAY 94-9027 was efficacious for the prevention and treatment of bleeding episodes in previously treated children with severe hemophilia A. We report interim long-term efficacy and safety data from the PROTECT VIII Kids extension study.
Methods:
In PROTECT VIII Kids, previously treated patients (PTPs) aged <12 years with severe hemophilia A received BAY 94-9027 prophylaxis twice weekly (25‒60 IU/kg), every 5 days (45‒60 IU/kg), or every 7 days (60 IU/kg). Patients completing ≥50 exposure days (EDs) and ≥6 months in the main study or a 12-week safety substudy (part 2) that enrolled PTPs aged <6 years could continue in the optional extension for an additional ≥50 EDs.
Summary:
Fifty-nine of 73 patients treated with BAY 94-9027 in PROTECT VIII Kids (main study or part 2) continued in the extension (median [range] age at enrollment in the main study, 5.0 [2–11] years). At data cutoff (January 2018), patients had a median (range) of 1456 (351–1665) days in the trial (main study or part 2 plus extension). Patients in the extension received prophylaxis twice weekly (n=20), every 5 days (n=20), every 7 days (n=8), or switched prophylaxis frequency during the extension (variable frequency; n=11). Median (range) dose/infusion was 52.1 (19–62) IU/kg. Median annualized bleeding rate (ABR) for total bleeds was 1.8 for all patients and 0.8, 1.1, 2.1, and 3.2 for those treated twice weekly, every 5 days, every 7 days, or with varying frequency, respectively. Median ABR for joint bleeds in all patients was 0.7. During the extension, 3 patients (5.1%) experienced treatment-related adverse events (AEs) classified as mild (n=1), moderate (n=1), or severe (n=1). One patient discontinued because of a serious AE that was not related to treatment. No confirmed FVIII inhibitors or anti-PEG antibodies were observed; no patients had sustained levels of detectable PEG in plasma.
Conclusions:
Long-term treatment (up to ~4.5 years) with BAY 94-9027 prophylaxis was efficacious and well tolerated in previously treated pediatric patients with severe hemophilia A.
Long-term Benefit of BAY 81-8973 Prophylaxis in Children With Severe Hemophilia A: Interim Analysis of the LEOPOLD Kids Extension Study
Effective Protection for >5 Years With BAY 94-9027 Prophylaxis: PROTECT VIII Extension Trial Interim Results
BAY 94-9027 Maintains Hemostasis During Major Surgery in Adults and Adolescents With Severe Hemophilia A: PROTECT VIII Results
Achievement of therapeutic levels of factor VIII activity following gene transfer with valoctocogene roxaparvovec (BMN 270): Long-term efficacy and safety results in patients with severe hemophilia A
rFVIIIFc for immune tolerance induction in severe hemophilia A subjects with inhibitors: a follow-up retrospective analysis
Bypassing agent (BPA) use for the treatment of bleeds in persons with Hemophilia A (PwHA) with inhibitors before and after emicizumab prophylaxis in the HAVEN 1 study
Change in cost and units consumed by people with factor VIII and factor IX deficiency after switching from a standard half-life product to an extended half-life product
Patient Satisfaction with US Hemophilia Treatment Centers: National Trends 2017
Objective:
Patient satisfaction with healthcare services is a measure of patient centeredness, influences treatment adherence, and increasingly affects reimbursement. In 2018, the US Hemophilia Treatment Center Network (USHTCN) launched the second national Patient Satisfaction Survey (PSS).
Methods:
A Steering Committee and Regional HTC Coordinator work group updated, piloted, and finalized the two-page survey for patient self-administration online, at clinic, or at home, in English or Spanish, and mailed to households. Survey content and format were based on national health surveys to enhance comparability and scientific robustness. Questions included assessed patient demographics, satisfaction with team members, and care processes aligned with HRSA goals. An open ended question sought qualitative data. Respondents were anonymous but the HTC where they received care was identified. Participation was voluntary. Persons with genetic bleeding disorders who had HTC contact in 2017 were eligible. Since March 2018, HTCs sent surveys to approximately 31,650 households. Parents were asked to complete surveys for children under age 15. No reminders were sent. Data were entered and analyzed at a central site and aggregated at national, regional and HTC levels. Survey remains open through summer 2018.
Results:
4042 individuals (12.8%) from 125 (90%) of the 139 Centers in the USHTCN returned surveys by June 12, 2018. National analyses on 4042 surveys reveal that 93% - 98% were ‘always’ or ‘usually’ satisfied with HTC care processes: shared decision making (97%); care coordination (97%); obtaining understandable information (97%); getting timely services (95%); enough time with staff (97%); being treated respectfully (98%); and HTC Factor Program/Pharmacy (340b) (96%). 95% - 97% were ‘always’ or ‘usually’ satisfied with core HTC team members. 91% of 12-17 year olds were ‘always’ or ‘usually’ satisfied with HTC encouragement regarding becoming more independent, and 92% with how the HTC discussed caring for a bleeding disorder upon reaching adulthood. Insurance and language were ‘always’ or ‘usually’ a problem for 14% and 9% respectively. 31% of respondents were female and 10% Hispanic. 80% were Caucasian, 5% African-American, 4% Asian/Pacific Islander or Native Hawaiian, 4% Multiple races, and 7% did not respond. Over 60% had severe or moderate FVIII or FIX deficiency or VWD Type 3. Ages ranged from newborns to over 90 years: 37% under 18, 18% age 18 – 34, and 45% over age 35.
Conclusions:
Implementing a National Patient Satisfaction Survey for the USHTCN remains feasible, is supported by HTCs nationally, and provides valuable information. Satisfaction with HTC services including 340B pharmacy is high. Insurance and language pose problems for 9-14%. Future analyses will examine additional national data and regional variation, and identify trends from the first national PSS conducted in 2014.
Integrated efficacy and safety analysis of Phase 2 and 3 studies with glecaprevir/pibrentasvir in patients with a history of bleeding disorders and chronic hepatitis C virus genotype 1–6-infection
Ultrasound-mediated Therapeutic Gene Transfer for Hemophilia
Discrepant Hemophilia A: Single Institution Experience
Pharmacokinetics, Efficacy, and Safety of High-Purity Factor X for Prophylactic Treatment of Hereditary Factor X Deficiency
A Multicenter, Retrospective Data Collection Study on the Compassionate Use of a Plasma-Derived Factor X Concentrate to Treat Patients with Hereditary Factor X Deficiency
My Life, Our Future: Development of the World’s Largest Genetic Research Repository for Hemophilia
Objective:
My Life, Our Future (MLOF) seeks to advance understanding of hemophilia by developing the MLOF Research Repository, a resource for use in scientific study. To strengthen the database and improve clinical care for females, MLOF expanded in 2016 to collect data and samples from potential and confirmed carriers.
Methods:
MLOF is a collaboration between the American Thrombosis and Hemostasis Network (ATHN) (educates HCPs, collects and protects genetic data, manages ATHNdataset, manages research review committee), Bloodworks Northwest (BWNW) (performs genetic testing and analysis, manages MLOF Research Repository), National Hemophilia Foundation (educates patients and the community), and Bioverativ (provides scientific collaboration and sponsorship). Patients enrolled in MLOF can contribute their de-identified clinical information and specimens (DNA, RNA, plasma and serum) to the MLOF Research Repository. The patients’ hemophilic genetic data (F8 or F9 variant) and specimens are stored at BWNW and their clinical data in the ATHNdataset. For carrier project participants, an ISTH Bleeding Assessment Tool score is determined, and for confirmed carriers factor levels are obtained and recorded in ATHN Clinical Manager. Upon enrollment of 5,000 participants, the MLOF Research Repository was opened to U.S-based investigators in February 2017. Investigators were encouraged to partner with a participating hemophilia treatment center (HTC) on projects for clinical translational support. An independent, international, multidisciplinary panel of experts was convened to review research proposals, evaluating project feasibility, scientific merit and potential contribution to the bleeding disorders community.
Summary:
To date, 97 HTCs are actively participating in MLOF and have enrolled 8,246 patients. Of those, 83% (6,857), including 723 confirmed carriers, have consented to research. The samples of 427 additional females consenting to research are pending evaluation; 1,643 females have participated in MLOF. The first MLOF Research Repository cycle received 9 Letters of Intent and 7 were chosen for full proposal review. The final selection of studies will be announced to the community in June 2017.
Conclusions:
Combining and analyzing genetic and clinical data via this database may allow researchers to solve unmet needs in patients with hemophilia, including understanding inhibitor development, bleeding severity or aiding in identifying new therapeutic targets. By considering molecular drivers of disease and genetic variability, this approach could lead to more individualized treatment through advancement of precision medicine. Global expansion of the MLOF Research Repository is planned for 2018. Carrier testing may help females manage their bleeding disorder and may aid in family planning. Related to research, females provide a unique control group for males with hemophilia on natural history, modifier genes inside and outside the coagulation system, and epigenetic factors affecting outcomes.
Staying on TRAQ: Determining transition readiness from pediatric to adult care in adolescents and young adults with hemophilia
Qualitative findings from bleeding disorders camp
Living with hemophilia B: examining quality of life and associated characteristics in the Hemophilia Utilization Group Studies (HUGS Vb) cohort
Objective:
To identify sociodemographic, clinical and treatment characteristics associated with the quality of life (QoL) of individuals with hemophilia B (HB) using longitudinal data in the HUGS Vb cohort.
Methods:
Between 2009-2012, 148 persons with HB were enrolled into HUGS Vb, a prospective cohort study examining individuals seen at ten federally supported U.S. hemophilia treatment centers (HTCs). Participants or parents of pediatric enrollees completed periodic surveys; data from 107 individuals with at least three follow-up surveys, clinician charts and dispensing records were included in the analyses. Data were analyzed at baseline and 6-month intervals across 2 years, yielding 5 time points. Periodic QoL assessments (SF-12 for adults and PedsQL for children), self-reported pain, employment and insurance status, time lost from work/school and treatment regimen were collected. Descriptive statistics and Spearman’s correlation coefficient test were used to examine the associations.
Summary:
Forty-six percent of the sample had severe HB; 50% were children (2-17 years). Among those with severe HB, 64% of children and 50% of adults treated prophylactically. 58% of adults were employed full-time. Individuals with mild hemophilia missed more work/school days due to disease-related issues (8 days) than those with moderate hemophilia (2 days) or severe hemophilia (3 days, P=0.03). QoL scores were similar over time among those using prophylactic and on-demand treatment for both adults and children. Median adult Mental Component Scores (MCS) and Physical Component Scores (PCS) measured at 5 time points ranged from 53.0 to 55.1 for MCS and 45.5 to 50.5 for PCS, with no significant changes observed over time. However, adults employed full-time had significantly higher median PCS at each time point than those working less than full-time (all Ps<0.05). Adults who reported pain had significantly lower median PCS than those who reported no pain/pain only when bleeding at each time point (all Ps<0.03). Median MCS remained similar between the two groups. Overall, we observed no longitudinal differences in children’s total PedsQL scores (range of median: 81.2-92.4) or in functioning subscales. However, among 18 children with QoL scores at both baseline and 24 months, missed school days were significantly correlated with decreased social functioning over time (rho=0.73, P<0.001). 8% of children who reported pain had consistently lower median total QoL scores than those reporting no pain/pain only when bleeding, despite having access to insurance and prophylactic treatment.
Conclusions:
Longitudinal data collected by HUGS Vb provide a valuable opportunity to examine the association of HB patient characteristics with measures of QoL in a multi-state sample. These data demonstrate that lower QoL was consistently associated over time with multiple factors, including absence from school, unemployment and pain. Continued analysis of this cohort will increase our understanding of the challenges faced by persons with HB.
Efficacy, safety and pharmacokinetics of once-weekly prophylactic emicizumab (ACE910) in pediatric persons (<12 years) with hemophilia A with inhibitors: interim analysis of single-arm, multicenter, open-label, phase 3 study (HAVEN 2)
Efficacy, safety and pharmacokinetics of emicizumab (ACE910) prophylaxis in persons with hemophilia A with inhibitors: randomized, multicenter, open-label, phase 3 study (HAVEN 1)
What Symptoms of Hemophilia Most Impact Quality of Life – A Quantitative Survey of People Living with or Caring for Someone with Hemophilia A
Objectives:
To better understand what symptoms beyond bleeds are experienced, as well as the depth of impact of these symptoms and how they uniquely impact people living with hemophilia on a daily basis. Additionally, the study aims to better understand patients’ satisfaction with current treatments in addressing their hemophilia needs.
Design/Method:
An email invitation was sent to all U.S. members affiliated with hemophilia A of MyHemophiliaTeam, a social network of people diagnosed with or caring for someone with hemophilia. 54 members responded to a 24 question survey between April 19 and May 1, 2017.
Results:
Hemophilia had a significant negative impact on the day-to-day life of adults (72%) and children (52%). Pain was the most broadly and acutely experienced symptom: 60% of adults and 28% of caregivers felt that pain had a major impact on their lives and 33% of adults and 25% of caregivers considered mobility to be significantly impacted by hemophilia.
For adults, both pain and mobility limitations impacted sleep (71% and 45%, respectively), being able to perform chores (71% and 65%), and the ability to work (48% and 45%). For children, these conditions impacted school attendance (61% and 58%) and participation in high impact activities like running or playing soccer (56% and 75%).
Depression and anxiety were also common symptoms that impacted sleep across adults (71%, 61%) and children (60%, 55%). Adults most commonly reported feeling negative ones: stress (38%), fatigue (38%) and annoyance (35%).
81% of adults and 86% of caregivers were extremely or very satisfied with current treatment. However, needs beyond treating bleeds are currently not being met. Few felt their pain was adequately addressed by current therapies (74% of adults and 57% of children reported no relief). Mobility impairment issues were also not being adequately addressed. Time spent on treatment impacted people with hemophilia (39% of adults and 43% of children, respectively were not satisfied with the frequency of treatment).
Background:
While people with hemophilia are known to suffer from bleeding, numerous concomitant symptoms also burden these patients, including pain, mobility impairments, depression, and anxiety. These symptoms can have a significant impact on quality of life, limiting work and school attendance, causing social withdrawal, and encouraging inactivity. Additionally, available treatment options can sometimes fall short in treating the totality of hemophilia symptoms.
Conclusions:
People with hemophilia have many challenges beyond bleeds that are not currently being well addressed. This is particularly true for the pain experienced. As such, a more holistic approach to treating hemophilia beyond bleeds would be beneficial to patients living with hemophilia. Additionally, therapies that reduce the need and frequency for treatment could potentially lower the burden of disease.
A Cumulative Review on Four Decades of Thrombo-Embolic Events Reported with the Use of Activated Prothrombin Complex Concentrate (APCC) in Congenital Haemophilia
Estimating the prevalence of symptomatic, undiagnosed von Willebrand disease: analysis of medical insurance claims data
Assessment of numeracy, genetic knowledge and perceptions of genetic testing in carriers of Hemophilia A and B
Prevalence of gross motor delays in boys with hemophilia ages 4-14: single site study
Lessons Learned in the Assessment of Functional Status in US Adults With Hemophilia in the Pain, Functional Impairment, and Quality of Life (P-FiQ) Study: Importance of More Formalized Assessment of Function in the Comprehensive Care Setting
Lessons Learned From the Assessment and Prevalence of Anxiety and Depression in US Adults With Hemophilia in the Pain, Functional Impairment, and Quality of Life (P-FiQ) Study: Importance of Routine Screening and Comprehensive Approaches to Management
Objective:
Pain and functional impairment resulting from joint disease in patients with hemophilia (PWH) may impact emotional well-being, resulting in consistent reports of anxiety/depression. The P-FiQ study formally evaluated patient-reported anxiety/depression symptoms and treatment as well as responses to standardized patient-reported outcomes (PROs), and evaluated reliability, validity, and consistency of responses.
Methods:
At a comprehensive care visit, adult male PWH with a history of joint bleeding or pain completed a hemophilia history and 3 patient-reported outcomes (PROs) assessing anxiety/depression and quality of life (QoL): EQ-5D-5L, Brief Pain Inventory (BPI), and SF-36v2. PROs were assessed for reliability, consistency, and correlation with factors including patient-reported characteristics.
Summary:
A total of 381 PWH (median age, 34 years) were enrolled in P-FiQ; 77% had hemophilia A, 23% had hemophilia B, and 9% had inhibitors. Fewer than half (44%) were currently receiving routine infusions to prevent bleeding. More than half were employed full-time (53%), and 65% were married or had a long-term partner. Depression was reported by 19% and anxiety by 14%. On the EQ-5D-5L anxiety/depression item, 43% reported feeling anxious or depressed “today.” On BPI, most participants indicated that pain interfered in the previous week with mood, sleep, and enjoyment of life, and more than half (54%) indicated interference with relations with other people. On SF-36v2 (range 0- 100, higher scores indicate better QoL), median mental health summary score was 50.7; subdomains were similar (vitality, 49.0; social functioning, 45.6; role emotional, 55.9; mental health, 52.8). Emotional problems resulted in reduced time spent on work/activities (40%) and accomplishing less than they would like (47%). More than half (55%) had felt downhearted or depressed, and a large majority (93%) had felt tired in the past 4 weeks. Sixty percent of participants indicated that their physical or emotional problems had interfered with their normal social activities with family, friends, and other contacts. Similar items across PROs correlated with one another, and PRO scores (EQ-5D-5L anxiety/depression, SF-36 mental health) were significantly (P<0.05) correlated with self-reported anxiety/depression.
Conclusion:
Anxiety and depression in adults with hemophilia have been consistently reported in other studies and were identified in P-FiQ by self-report and across several PRO instruments. Emotional problems were reported to interfere with normal social activities and productivity. While the unmet need to address mental health in PWH has received increased recognition, it is not typically assessed formally. When compared with pain, management strategies and/or referral relationships may also not be as formally established. The findings presented here highlight the potential value of simple screening tools (eg, EQ-5D-5L) and opportunities to encourage patient dialogue about mental health within the comprehensive care setting and in referral networks.
Lessons Learned in the Assessment of Pain in US Adults With Hemophilia in the Pain, Functional Impairment, and Quality of Life (P-FiQ) Study: Importance of More Formalized Discussions Around Pain in the Comprehensive Care Setting
Management of Hemophilia B in US Women and Its Impact on Education, Employment, and Activities: Results From the Bridging Hemophilia B Experiences, Results, and Opportunities Into Solutions (B-HERO-S) Study
Objective:
The B-HERO-S study assessed the impact of hemophilia on US adults with hemophilia B, including affected females. Here we describe the symptoms, management, and impact on education, employment, and engagement in recreational activities in women.
Methods:
Adults aged ≥18 years with hemophilia B (factor IX <40%) completed a survey including questions about bleeding, treatment, and psychosocial impact.
Summary:
Of 299 respondents, 86 were women (median age, 29 years; 6% were aged >45 years) with mild (29%), moderate (65%), or severe (6%) hemophilia. The majority reported arthritis (66%) and anxiety/depression (57%); 19% reported acute and/or chronic pain. Most (86%) were treated with some form of “routine infusions to prevent bleeding”; 16% reported self-infusing, 31%/17% by partners/family. Self-infusion was initiated at median age of 18, and learned from HTCs (86%), camps (71%), or parents (57%). Most reported difficulty with access to factor due to affordability/availability in the past 5 years (72%) or expected in the next 5 years (85%). Women reported a median (mean) of 4 (3.86) bleeds in prior year; 83% bled within the last 4 weeks with most recent bleeding in joints (73%) or muscles (21%). Twenty-five percent reported one specific “bad” joint, most commonly the knee (69%). Nearly all were seen in HTCs (85%) with a median (mean) of 3 (3.5) HTC visits per year. Most (88%) went past high school with 55% completing 4-year college and 24% graduate degrees. Most (94%) reported some negative impact on completing their education (7% very large/69% moderate/19% small impact), most commonly due to difficulties concentrating at school due to bleeds or pain (81%). Most were employed full-time (71%) or part-time (10%), commonly in office work (71%). Most (94%) reported a negative impact on their working life (3% very large/71% moderate/20% small impact); 36% reported leaving a job because of their hemophilia. Nearly all (99%) indicated some negative impact on their ability to engage in recreational activities (3% large/83% moderate/13% small impact). From a list of predetermined activities, dancing (50%) and walking (48%) were the most common current activities; bicycling (20%), dancing (17%), and swimming (16%) were the most common discontinued activities. Most reported treatment changes around activities (21% started prophylaxis, 52% revised time of doses, 38% added doses, 34% changed amount of doses); 8% reported no change and 3% no moderate/vigorous activities.
Conclusion:
Clinical presentation and treatment in women mirrored that reported by men except for greater anxiety/depression and more issues with access to treatment. Nearly all affected women reported a negative impact on their education, employment, and activities. While these results may be limited by bias in recruitment selecting those most symptomatic, the data reveal opportunities to improve awareness of and guidance around management and counselling of affected women.
The WFH Launches World Bleeding Disorders Registry to Expand Knowledge Base Worldwide
The WFH Annual Global Survey: Gender Distribution
Mild-Severe Hemophilia B Impacts Relationships for US Adults and Children With Hemophilia B and Their Families: Results From the Bridging Hemophilia B Experiences, Results, and Opportunities Into Solutions (B-HERO-S) Study
Objective:
The B-HERO-S study evaluated the impact of mild-severe hemophilia B on the lives of affected adults and children. Here we assess the impact of hemophilia B on relationships.
Methods:
US adults with hemophilia B and caregivers of affected children completed separate 1-hour online surveys that included questions regarding impact on interpersonal relationships.
Summary:
Most (88%) of the 299 adults completing the survey had mild-moderate hemophilia B. Of those, 54% were married or in a long-term relationship, and 44% were single. Most adults (87%) reported that hemophilia impacted their ability to form close relationships with partners or prospective partners; 35% were very/quite dissatisfied with the support received from a previous partner. Ninety percent reported that their experiences in a prior relationship, including satisfaction with support from their previous partner, impacted their decision to enter a relationship with their current partner. Nearly all participants (98%) were very/quite satisfied with the support received from their current partner. The top reason for satisfaction was “my partner takes the lead in providing financial security” (45%). Most were very/quite satisfied with the support from family (87%) and friends (96%). Most participants reported a negative reaction or experience as a result of disclosing their hemophilia (friend/colleague/employer, 76%/80%/82%); some reported having felt bullied by peers/colleagues (69%/66%). Majorities reported that past experiences impacted which friends/colleagues they told about their hemophilia, and how or when they disclosed their hemophilia status to these individuals (97%/95%). Seventy-four percent of participants indicated that hemophilia has affected the quality of their sex life; only 54% were extremely/moderately satisfied with their overall sexual relationship. Many reported having had a bleed during sex (40%). Of 150 caregivers of children with mostly mild-moderate hemophilia (74%), 89% were married or in a long-term relationship, and most felt very/quite supported by their partner (98%) and family (87%). Impact on unaffected siblings was more often mixed (49%) than negative (18%). Most felt very/quite satisfied with support of teachers (91%), children at school (80%), and other adults in regular contact (72%). Most caregivers reported negative experiences telling a friend (76%) or having their child tell a friend (69%) about the child’s hemophilia; 43% reported that their child was bullied as a result of having hemophilia.
Conclusion:
While the impact of severe hemophilia on relationships has been reported in HERO and other studies, B-HERO-S suggests that mild-moderate hemophilia B also significantly impacts relationships of affected men/women and boys/girls, especially in terms of disclosure, intimacy, and feeling bullied by peers/colleagues. Opportunities may be explored to more proactively counsel individuals with mild-moderate hemophilia B and families in the setting of comprehensive care to better navigate interpersonal relationships and improve quality of life.
Impact of targeted education on obesity in children with hemophilia-a single HTC quality project
Real-World Specialty Pharmacy Dispensation and Expenditures Associated with Prophylactic Regimens Using Standard and Extended Half-Life Recombinant Factor IX Products in Severe Hemophilia B
Sports/Recreational Activity-Specific Range and Drivers of Risk in People With Hemophilia: Results of the Activity-Intensity-Risk (AIR) Survey and Consensus Meeting of US Physical Therapists
Objective:
Limited evidence supports activity-associated bleeding risk assessment for people with hemophilia (PWH), and consumer materials generically describe types of risk and a single activity risk score based on input from a few physical therapist (PT) authors. The aim of AIR was to assess activity-specific risk ranges, bleed-specific risks, and inherent/modifiable factors that increase risk based on survey/consensus of PT experts.
Methods:
Peer- nominated PTs from US hemophilia treatment centers (HTCs) were invited to participate in a survey regarding ~100 sports/recreational activities. For each activity, respondents provided a low (minimum) and high (maximum) risk assessment on a 5-point scale (low=1, high=5). Position-specific assessments were made for some team sports (eg, baseball pitcher, catcher, and field positions). Sports with distinctly different rules for contact were evaluated separately (eg, flag/tackle football, boys/girls lacrosse, Frisbee/ultimate). Drivers of risk were identified from free text comments and explored at a consensus meeting. Drivers of risk were categorized as inherent, modifiable, activity-driven, and patient-driven.
Summary:
Of 32 invited PTs, 17 responded to the survey with median 26.5 years as a PT and 15.5 years at an HTC; 8 participated in the full-day consensus meeting. Of the survey participants, majorities reported treating adults (94%) and treating children (88%), and most worked in the HTC full- time (29%) or nearly full-time (41%). Overall, few activities had low and high risk assessments both fall within the lower (1) or upper (5) end of the response range. For example, swimming is associated with low risk scores, even when including maximum risk with year-round competitive teams (median low 1, high 2), and tackle football had consistently high scores (median low 5, high 5). Risk scores (median low, high) for some common sports were as follows: baseball pitcher (3,4), catcher (3,4) or other position (2,3); basketball (2,4); hockey (4,5); skiing-downhill (2,4); soccer goalie (2,4) or other position (2,4); snowboarding (3,5). Risks for joint injuries were consistent with position and motion requirements for each sport, while head and muscle bleeds were associated with contact. Key drivers of risk that were identified included progression from seasonal participation to year-round play, overtraining, competitive level, participation in tournaments, and improper body mechanics. Inherent risks included impact with surface/ball/equipment (eg, soccer goalie), impact with players (eg, football), or falls (eg, horseback riding). Modifiable risks included tricks/stunts (eg, skateboarding) and use of safety equipment when not required.
Conclusion:
AIR provides insights into activity-specific risk for PWH including types of bleeding risk and drivers of increased risk. The results may provide a broader framework for assessing activities with respect to bleed site-specific risks and for recognizing how certain activities may be modified to decrease risk or to identify those with non-modifiable inherent risks for injury.
Physician practice patterns in the US show significant variation in how PK parameters are currently used to personalize care for US hemophilia A patients
Introduction & Objective:
Standard approaches to prophylaxis may be further improved by taking into account individuals’ pharmacokinetic (PK) profile, thereby increasing the likelihood of therapeutic success [Valentino 2012, Valentino 2014, Lissitckov 2017]. One study demonstrated an association between more time spent above higher FVIII levels (20 and 30%) and lower bleeding rates [Valentino 2016]. Furthermore, a consensus statement proposes to target specific factor levels to tailor treatment for different patient profiles [Iorio 2017]. Additionally, new extended half-life treatments provide physicians with another option to personalize therapy. As more data and therapeutic options become available, it is important to understand physicians’ current perceptions and practice patterns with respect to personalizing haemophilia A care in the US.
Materials & Methods:
Physicians in the US who treat persons with severe hemophilia A and provided informed consent were eligible to complete a cross-sectional, double-blinded web-based survey to evaluate physicians’ perceptions and practice patterns with respect to measuring PK and if/how they personalize FVIII treatment with this information, when and to what extent. This abstract presents results from the first half of the survey which focused on ways in which physicians measure FVIII pharmacokinetics and personalize care.
Results:
Ninety physicians completed the survey. The top three most important considerations for personalizing therapy in general were bleeding history, patient goals, and physical activity. The most commonly cited reason for conducting a PK assessment was product switch (74%) while the most common barrier was patient willingness/availability (60%). Physicians reported using a PK-based approach to personalize treatment in 25% (median) of their severe patients. Of physicians who use PK, trough levels (91%), half-life (58%), peak levels (56%) and Area Under the Curve (22%) were used. While 12% of these physicians reported using all PK parameters, 18% only used trough levels. 23% reported using peak, trough and half-life in combination. Most physicians (89%) indicated using PK data to adjust dose and 50% also used it as a patient education opportunity.
Conclusions:
There was significant variability across respondents as to how PK is assessed and how PK parameters are used in treatment decisions, suggesting an opportunity to increase awareness and use of PK-guided personalization to ultimately improve patient care. Additional education on the definitions and details of PK-guided dosing could help improve overall adoption of this treatment strategy and align the community on what it means to personalize therapy using PK information. More research studying the association between PK-guided prophylaxis and outcomes is encouraged to better understand how best to personalize PK-based prophylaxis for different patient scenarios.
Real-World Pharmacy Dispensation and Expenditures Associated with Standard and Extended Half-Life Recombinant Factor VIII Products in Hemophilia A
CHESS – Improving research and advocacy through an improved understanding of the economic and social burden of hemophilia
Assessing the Availability and Use of Resources to Support Youth with Hemophilia, their Families and Care Teams during Transition to Adult Health Care
Centralized Inhibitor Testing in the United States: Laboratory Methods Used for the Community Counts Registry for Bleeding Disorders Surveillance
Management of Bleed Events in the Phase 2 Study of Fitusiran, an Investigational RNAi Therapeutic Targeting Antithrombin for the Treatment of Hemophilia A and B with and Without Inhibitors
Background:
Hemophilia is an inherited bleeding disorder caused by an impairment in the body’s ability to accomplish the natural clotting process. People with hemophilia experience bleeds because there is an inadequate amount of thrombin due to a deficiency in factor VIII or IX. Thrombin is critical to clotting and sealing the wound by converting fibrinogen into fibrin, reinforcing the primary platelet plug. Thrombin activity is regulated by antithrombin.
Fitusiran is a subcutaneously administered investigational RNA interference (RNAi) therapeutic targeting antithrombin with the goal of improving thrombin generation to promote clotting in people with hemophilia A or B with and without inhibitors. Fitusiran is currently being evaluated as a prophylactic agent for hemophilia A and B with and without inhibitors in an ongoing Phase 2 extension study. Breakthrough bleeds on the study are being managed using replacement factors (non-inhibitor patients) or bypassing agents (BPAs; inhibitor patients). The use of replacement factors or BPAs in the background of fitusiran treatment is of clinical interest and will be described.
Methods:
The Phase 2 extension study (NCT02554773) includes people with hemophilia A or B with and without inhibitors, previously dosed in the Phase 1 (NCT02035605) study. Participants receive monthly, fixed subcutaneous doses of fitusiran, 50 mg or 80 mg. Data on breakthrough bleeds and how they were treated were collected by patient diary.
Results:
As of May 2017, 33 participants were enrolled in the study. Previously reported data demonstrated that fitusiran was generally well tolerated and led to dose-dependent antithrombin lowering, thrombin generation increase, and decrease in bleeding frequency in participants with hemophilia A and B with or without inhibitors. Among those achieving target antithrombin lowering of >75%, few bleed events occurred during the observation period. Bleed events were treated with factor concentrates (FVIII or FIX) or bypassing agents (rFVIIa or aPCC), respectively, in doses according to or lower than recommended by the WFH guidelines. Detailed analyses of the frequency and management of bleed events in the Phase 2 study will be presented.
Conclusions:
Clinical data suggest that fitusiran may be a promising investigational prophylactic therapy to promote appropriate clotting and reduce the frequency of bleeding in people with hemophilia A or B with and without inhibitors. Further, the initial limited experience in treating breakthrough bleeds with replacement factor or BPAs has been encouraging, demonstrating good treatment effect in the absence of identified safety concerns.
Fitusiran, an Investigational RNAi Therapeutic Targeting Antithrombin for the Treatment of Hemophilia A and B with and Without Inhibitors: Interim Results from a Phase 2 Extension Study
Background/Objective:
Hemophilia is a bleeding disorder caused by the body’s inability to accomplish the natural clotting process. People with hemophilia experience bleeds because there is an inadequate amount of thrombin due to a deficiency in factor VIII or IX. Thrombin is critical to clotting and sealing the wound; it converts fibrinogen into fibrin, establishing a network to help more platelets accumulate. Thrombin activity is regulated by antithrombin. Fitusiran is a subcutaneously administered investigational RNA interference (RNAi) therapeutic targeting antithrombin with the goal of improving thrombin generation to promote clotting in people with hemophilia A or B with and without inhibitors. Interim data from the Phase 1 study showed fitusiran was generally well tolerated and administration of monthly fitusiran led to dose-dependent antithrombin lowering, improvement in thrombin generation, and decrease in bleeding frequency. We will report interim safety, pharmacodynamics, and clinical activity of fitusiran from the Phase 2 extension study.
Methods:
The Phase 2 extension study (NCT02554773) includes people with hemophilia A or B with and without inhibitors, previously dosed in the Phase 1 (NCT02035605) study. Participants receive monthly, fixed subcutaneous doses of fitusiran, 50 mg or 80 mg. Primary endpoints include safety and tolerability; secondary endpoints include antithrombin activity, thrombin generation and exploratory evaluation of bleed events.
Summary of Results:
As of May 2017, 33 participants were enrolled in the study and had received continuous dosing of up to 14 months. Previously reported data showed that fitusiran was generally well tolerated, with no serious adverse events related to study drug and no thromboembolic events. Once-monthly subcutaneous dosing achieved dose-dependent antithrombin lowering of ~80% and thrombin generation levels approaching levels similar to participants without hemophilia. Exploratory post-hoc analysis of bleed events showed median annualized bleed rate (ABR)=1 in participants without inhibitors and median ABR=0 in participants with inhibitors. Bleed events were successfully managed with either replacement factors or bypassing agents. Updated safety, tolerability and clinical activity will be presented.
Conclusions:
Emerging clinical data suggest that fitusiran may be a promising investigational prophylactic therapy to promote appropriate clotting and reduce the frequency of bleeding in people with hemophilia A or B with and without inhibitors.
Management of Hemophilia Carriers Around The Time of Their Delivery: Phenotypic Variation Requiring Customization of Management
An Integrated Safety and Efficacy Analysis of Sofosbuvir-Based Regimens in Patients with Hereditary Bleeding Disorders
Objective:
Patients with hereditary bleeding disorders have been included in Phase 2 and 3 clinical trials of sofosbuvir (SOF), ledipasvir/sofosbuvir (LDV/SOF), sofosbuvir/velpatasvir (SOF/VEL), and sofosbuvir/velpatasvir/voxilaprevir (SOF/VEL/VOX) as well as in a dedicated study (n = 120) in this patient population. This integrated analysis evaluates the safety and efficacy of SOF-based regimens in HCV-infected patients with hereditary bleeding disorders.
Methods:
HCV-infected patients with a medical history of a hereditary bleeding disorder who participated in a SOF-based Phase 2 or 3 study were included in this pooled analysis. Medical history term(s) used to identify patients with bleeding disorders included variations of Hemophilia A or B, Von Willebrand’s Disease, Factor Deficiencies, or conditions associated with hemophilia.
Summary:
A total of 201 patients (74 GT1, 10% GT2, 14% GT3, 2% GT4, <1% GT5) with bleeding disorders were identified across 19 studies. The majority were male (91%), Caucasian (82%), IL28B non-CC (70%), HCV treatment-naïve (55%), and without cirrhosis (74%). Hemophilia A (65%) and B (26%) were the most common bleeding disorders. SVR12 results are shown in the below table by treatment regimen and genotype. The most frequently reported adverse events (>10%) were fatigue and headache; majority were mild or moderate in severity. One patient (<1%) discontinued LDV/SOF due to an adverse event and 11 patients (5%) experienced a serious adverse event. Hemarthrosis, muscle hemorrhage, epistaxis, hematoma, and hematuria were the only hemorrhagic events that occurred in >1 patient. Grade 3 or 4 laboratory abnormalities were infrequent with anemia and hyperbilirubinemia the most frequent Grade 3 laboratory abnormality consistent with RBV administration.
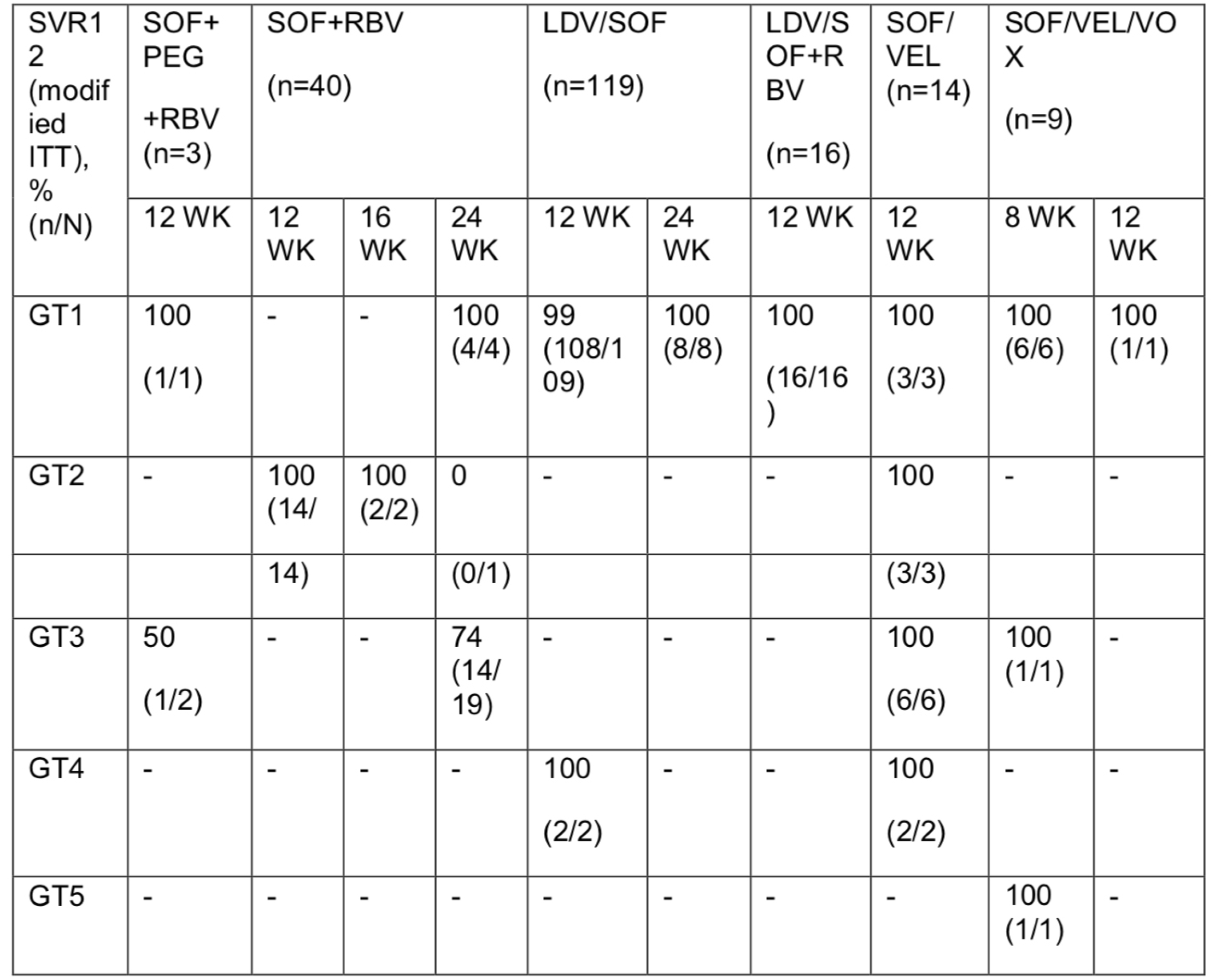
Conclusions:
SOF-based regimens led to high rates of SVR in genotype 1–5 HCV infected patients with bleeding disorders. SOF-based regimens were safe and well tolerated with no new toxicity specific to patients with bleeding disorders emerging
.
Efficacy, safety, and pharmacokinetics of a high-purity plasma-derived factor X (pdFX) concentrate in the prophylaxis of bleeding episodes in children <12 years with moderate to severe congenital factor X deficiency (FXD)
Background:
Congenital FXD is a rare bleeding disorder characterized by spontaneous joint and mucocutaneous bleeding and gastrointestinal or intracranial hemorrhage. pdFX is a US- and EU-approved treatment for congenital FXD, but data in children <12 years have been unavailable.
Aims:
To investigate pdFX efficacy, safety, and pharmacokinetics in children <12 years with moderate to severe congenital FXD.
Methods:
In this 6-month, open-label, multicenter, phase 3, prospective study in children <12 years, all subjects had a confirmed diagnosis of moderate to severe congenital FXD (basal FX:C <5%), severe bleeding history, or an F10 gene mutation causing a documented severe bleeding type. Subjects received routine prophylaxis at recommended 40–50 IU/kg twice weekly to maintain trough FX:C levels ≥5%. Each investigator assessed efficacy based on standardized criteria and presence of breakthrough bleeding. All subjects provided informed consent and the protocol was approved by appropriate independent ethics committees.
Results:
Mean age of the 9 trial completers was 6.8 years. Eight subjects had severe and 1 had moderate FXD. Overall, 537 prophylactic infusions were administered; mean dose/child was 38.6 IU/kg. Ten bleeds in 3 of 9 children were reported: 6 minor, 3 major, 1 unassessed. Investigators rated overall pdFX efficacy as excellent in all subjects. Overall mean incremental recovery was 1.74 IU/dL per IU/kg. FX trough levels were maintained >5% after visit 4 (days 29–42) in all subjects.
A total of 28 treatment-emergent adverse events (TEAEs) were reported in 8 children; none were considered pdFX related. No significant changes were noted in vital signs, physical exams, or laboratory measurements. No evidence of inhibitor development was seen.
Conclusion:
pdFX is efficacious in the prophylaxis of bleeding episodes in subjects <12 years with moderate to severe FXD. Safety profile in this population is consistent with previous results in subjects ≥12 years.
Funding: Bio Products Laboratory
Hereditary factor X (FX) deficiency in women and girls: treatment with a high purity plasma-derived factor X concentrate
Patient and Clinician Experience of Using Goal Attainment Scaling for Hemophilia (GAS-Hēm), an Innovative Patient-Centered Outcome Measure
Perceptions of Vulnerability, Protective Behaviors, and Reported Stress in Mothers of Sons with Hemophilia
Depression levels in patients with Hemophilia and von Willebrand
IMPACT QoL II - The relationship of depression and anxiety to control of chronic pain and adherence to clotting-factor treatment
Objectives:
The primary aims of this study are to 1) evaluate the prevalence of depression and anxiety among adult persons with hemophilia (PWH) and 2) explore relationships between depression, anxiety, chronic pain, and adherence to clotting-factor treatment.
Method:
This study used a subset of data from the IMPACT QoL II, a one-time, cross-sectional survey of 200 adults (age >18) with self-reported diagnosis of either Hemophilia A or B who were able to read, write, and speak English. The study was approved by the IRB in the primary investigator’s institution. Participants were a convenience sample recruited at bleeding disorders conferences in 2013-2014 and issued a randomly generated identification number to ensure anonymity. The 139-item survey was completed electronically through SurveyMonkeyTM on a study computer tablet. Participants were given $20 upon completion of the survey, which took 15-45 minutes to complete. The subset of variables evaluated for this analysis included the Faces of Pain Scale-Revised (FPS-R) (chronic pain), level of control over pain each participant feels they have, adherence to clotting factor measured by total scores (higher score=lower adherence) on the VERITAS-PRO or VERITAS-PRN, anxiety measured by the GAD- 7, depression measured by the PHQ-9, and self-report of ever receiving a diagnosis of depression and/or anxiety. The cut-off score for presence of moderate to severe depression or anxiety was 10 on both the PHQ-9 and the GAD-7, scores which have been validated by previous literature in other populations. Lower vs. higher adherence was defined by VERITAS scores in the highest and lowest quartile respectively.
Summary:
Participants with lower treatment adherence (VERITAS score >57) were more likely to have PHQ-9 scores >10 (P=0.02). GAD-7 scores >10 also demonstrated a trend to be associated with lower treatment adherence (P=0.15). 28% of participants reported a diagnosis of depression, but 53% with PHQ-9 scores >10 had not been diagnosed with depression. Similarly, 22% reported a diagnosis of anxiety but 52% with GAD-7 scores >10 had not been diagnosed. Participants who reported non-control of pain were more likely to have PHQ-9 scores or GAD-7 scores >10 (P=0.001 and P=0.013 respectively). There were significant correlations between PHQ-9 and GAD-7 (Rho=0.76, P<0.0001), PHQ-9 and FPS-R (chronic pain) (Rho=0.31, P=0.003), and GAD-7 and VERITAS (P=0.005). These data indicate that depression and anxiety are associated with greater severity of chronic pain, and depression is associated with poorer adherence to clotting factor treatment regimen (prophylaxis or other regimen). Both depression and anxiety appear to be under-diagnosed in PWH.
PROTECT VIII: Can Eligibility for Less-Frequent Prophylaxis Dosing Regimens Be Predicted by Patient Characteristics?
Ethics of Compensated Plasma Donation
Assessment of Current Clinical Practices in Integrating Treatment Guidelines for Hemophilia
Objective:
This study assessed current clinical practices of clinicians related to hemophilia treatment guidelines to identify knowledge, competency, practice gaps and barriers to optimal care of patients with inhibitors.
Methods:
A continuing medical education (CME)-certified clinical practice assessment survey was developed comprising 24 knowledge- and case-based, multiple-choice questions. The survey assessed knowledge, attitudes, and confidence with regard to newly-developed hemophilia treatment guidelines emphasizing integrated care for patients with inhibitors, and the application of these guideline-based recommendations. The survey launched on the Medscape Education website on December 5, 2016 with participant responses collected through January 26, 2017. The data sample includes responses from 170 physicians who participated during the study period.
Summary of Results (n=170 physicians):
Responses to questions on the screening for, and management of, inhibitor formation in patients with hemophilia undergoing prophylaxis, showed that: the majority of hematologists/oncologists correctly identified the factors that increase risk of inhibitor formation (71%), while less than half of pediatricians did so (46%); when asked regarding exposure days (EDs) and the formation of inhibitors, half of hematologists/oncologists correctly identified within 50 EDs, while only 25% of pediatricians did so; and both hematologists/oncologists (21%) and pediatricians (28%) incorrectly identified how often a patient should be tested for inhibitors. When surveyed specifically regarding immune tolerance induction (ITI), a slight majority of hematologists/oncologists and pediatricians correctly chose the time frame during which to initiate ITI (55% and 51%, respectively), and 50% of hematologists/oncologists knew the most powerful predictor of ITI success, while only 42% of pediatricians did so; only 14% of hematologists/oncologists and 4% of pediatricians knew that there is no optimal rFVIII to initiate for ITI; only 10% of hematologists/oncologists and 8% of pediatricians knew that there is not optimal dose of rFVIII to initiate for ITI.
Conclusions:
The need for further education was observed for the following topics: best practices in the integrative care of patients using evidence-based guidelines and recommendations; current and emerging clinical data guiding acute and prophylactic management; risk factors for the development of inhibitors during prophylaxis; screening and management of inhibitor formation, including ITI. Further educational efforts tailored to address these gaps are warranted.
Verification of Effective Zika Virus Reduction by Production Steps Used in theManufacture of Plasma-Derived Medicinal Products
A Feasibility and Usability Study of a Nursing Orchestrated, Customized 3 Dimensional Virtual Reality Environment in Children with Hemophilia Undergoing Routine Intravenous Procedures
Optimal dosing strategies evaluated using a model of the terminal half-life curves for 11 rFVIII products
Objective:
Physicians prescribing Factor VIII for haemophilia A patients are presented with an array of dose and dose frequency in the package insert making it difficult to precisely prescribe. To clarify the treatment outcome across products, a model of the published half-life was used to provide the expected % IU/dl +/- SD at 24, 48, 72, and 96 hours post dose at a normalized dose and at the high end of the recommended dosage (dose and freq.).
Methods:
Eleven rFVIII products recently marketed in the US were assessed using a model of the halflife +/- SD to calculate the expected %IU/dl +/- SD at 24, 48, 72, and 96 hrs post-dose. Terminal half-life data for adults was obtained from each published package insert (PI). The one-stage clotting assay data was used for all except Afstyla© (chromogenic assay only). Variance reported as coefficient of variation or confidence intervals was converted to standard deviation.
The first comparison was made using a standard dose of 50 IU/kg across all products. The second comparison used the maximum recommended dose and frequency for routine prophylaxis defined in the PI. Full data sets and curves of %IU/dl+/-SD under standard doses of 50 IU/kg and under maximum dose and frequency for routine prophylaxis have been generated.
Summary:
Graphing the single dose comparison (50 iu/kg) revealed 3 general clusters and one outlier at 24 hrs post dose. The first cluster had 4 products with a mean range of 21-25 %IU/dl at 24 hrs post dose. The second cluster had 5 products with a range of 30-32 %IU/dl at 24 hrs. The third cluster had 1 product (Eloctate©) with 43% IU/dl at 24 hrs. The outlier (Nuwiq©) showed exceptional variance compared to others and removed from discussion.
Expected %IU/dl was recalculated using the most frequent and highest dose recommended for individual products. Assessing the –SD value: 10 of 10 products achieved > 1% trough at 24 hrs; 8 of 10 at 48 hrs; 2 of 10 at 72 hrs; and 1 of 10 at 96 hrs. Note at 72hrs, only 2 of 10 products achieve minimal trough at –SD level when 4 of 10 claim q3day dosing.
Conclusions:
Initial dosing for routine prophylaxis relies on the mean half-life. The PK parameters are based on only 18 to 30 patients under well-controlled conditions. There is waste on both sides of the PK half-life distribution curve. Would it be reasonable to assure minimal breakthrough bleeds and less over-dosage by initiating treatment with a personal PK profile for each patient in order to identify the correct dose and frequency? This model could be used to find optimal dose and frequency with input from a personalized product half-life.
Characterization of Women and Girls with Hemophilia Treated in the US from A Claims Database
Updated results from a dose-escalation study in adults with severe or moderate-severe hemophilia B treated with AMT-060 (AAV5-hFIX) gene therapy: up to 1.5 years follow-up
Journey to Best Outcomes in Hemophilia Transition: Passage to Independence
Objectives:
To streamline and standardize the transition process of care through improved collaborations with staff and for persons with hemophilia transferring from a pediatric to an adult HTC. Processes were developed to provide patients with documented transition skills in order to foster medical independence in a complex healthcare environment.
Methods:
Continuous quality improvement tools were used to develop, implement and test a standardized transition tool for patients diagnosed with hemophilia A or B, ages fifteen to nineteen years of age. The transition tool was designed to assess the knowledge and skills of the adolescent in preparation for transition to adult care. Adherence to the administration of the tool in the pediatric HTC was the initial outcome measure. Key drivers included 1) improving communication between staff at the HTCs 2) transition tool development 3) educational resource content identification and 4) education of staff and families regarding the transition project. Communication was fostered through weekly team meetings to discuss and develop the transition tool in collaboration with the adult HTC. The adult HTC social worker would then attend the comprehensive clinic appointment at the pediatric HTC for those identified patients to assist in the preparation of transferring care. An excel spreadsheet, along with the ATHN database, was utilized to track patients to provide continuity of care during the transfer. Additionally, quarterly meetings were implemented with both HTC teams to discuss transferring patients and the continuum of the transition process. The transition tool was developed after review of available transition tools and was designed to provide systematic assessment of patient knowledge for transition readiness. The tool was refined through a series of PDSAs and implemented at each comprehensive clinic. The patient responses to the transition tool highlighted educational opportunities and led to the development of a resource cart to provide readily accessible targeted educational tools. Family and staff were educated about the value of transition readiness through team meetings, community outreach and during clinic visits.
Summary:
Use of the transition tool began in March 2016. Data was available through May 17, 2017. Thirty of 31 (97%) eligible patients completing the tool. Communication was improved between HTC teams. Educational tools were identified, obtained and provided to patients.
Conclusions:
We have successfully streamlined and standardized the transition process, identified educational opportunities and improved communication with staff at our HTCs utilizing established quality improvement techniques. Next steps include measurement of answered transition tool questions to further enhance patient/family knowledge and promote successful transition, as well as expansion to other age appropriate transition tools, to facilitate the journey to medical independence.
My Life Our Future Genotyping Days: On the Road Again to New Horizons
Pharmacokinetics and Prophylaxis Regimens and Relationship to Bleed Outcomes in Patients With Severe Hemophilia A Treated with BAY 81-8973
Background:
Factor VIII (FVIII) pharmacokinetic (PK) parameters and other patient characteristics may play a role in effectiveness of FVIII prophylaxis. Objective: A post-hoc analysis was conducted to assess the association between PK parameters and BAY 81-8973 (Kovaltry® ) prophylaxis regimen in patients with severe hemophilia A.
Methods:
LEOPOLD I was a randomized, open-label, phase 2/3 trial to investigate use of BAY 81-8973 routine prophylaxis for treatment of bleeds in adolescents and adults with severe hemophilia A. In the efficacy part of the trial (part B), patients received BAY 81-8973 at a dose of 20‒50 IU/kg 2x/week (n=18) or 3x/week (n=44) for 12 months. Prophylaxis regimens were determined by study investigators, independent of BAY 81-8973 PK data. A subset of 20 patients (2x/week, n=7; 3x/week, n=13) had data from an earlier PK study (part A), in which patients received a single 50-IU/kg injection of BAY 81-8973 and had blood samples collected over 48 hours to assess PK parameters, including area under the curve (AUC), half-life (t½), and clearance (CL). Estimates of PK parameters were also derived using a population PK model based on data from all patients. This analysis compared model-based PK parameters, baseline characteristics, total annual dosing, and bleed outcomes between 2x/week and 3x/week prophylaxis groups. A similar analysis was done among the subset of patients in part A who had PK data. Differences between 2x/week and 3x/week dosing were assessed using Wilcoxon rank sum tests for continuous variables, and chi-square or Fisher’s exact tests for categorical variables.
Results:
In the full trial population, model-based AUC [mean (SD): 1957.1 (648.3) vs 2139.1 (875.7), 2x/week and 3x/week, respectively], t½ [15.0 (3.7) vs 15.9 (6.6)], and CL [0.028 (0.009) vs 0.027 (0.011)] were comparable between groups. The proportion of patients with ≥3 bleeds at 12 months was 33% for 2x/week and 48% for 3x/week. The 3x/week group had more bleeds in the past 12 months, higher baseline Gilbert bleeding scores, were more likely to have target joints at baseline, and received higher doses of BAY 81-8973 during the 12- month treatment period. In the part A subset analysis, observed and model-based AUC, t½, and CL were similar in both groups. The proportion of patients with ≥3 bleeds at 12 months was 0% for 2x/week and 46.2% for 3x/week (P=0.052).
Conclusions:
The similarity of PK parameters in patients successfully treated with 2x/week and 3x/week regimens suggests that clinical factors other than t½, AUC, and CL are important in predicting success of prophylaxis with BAY 81-8973. Physician decisions, taking into consideration clinical factors to identify appropriate patients for specific prophylaxis regimens, appear to be an important factor for 2x/week or 3x/week dosing with BAY 81-8973. These factors should be considered when individualizing BAY 81-8973 prophylaxis frequency.
BAY 94-9027, a Site-Specifically PEGylated Recombinant Factor VIII: Preliminary Results From a Global Comparative Laboratory Field Study
Background:
Accurate measurement of factor VIII (FVIII) activity in patients with hemophilia A is important for patient monitoring and treatment decisions. Discrepancies in results using different assays or reagents to measure prolonged–half-life factor products have been recognized. BAY 94-9027 is a prolonged–half-life FVIII product site-specifically conjugated with a 60-kDa polyethylene glycol molecule (2×30 kDa branched).
Objective:
A global field study was conducted to assess the ability of clinical laboratories to measure BAY 94-9027 activity in spiked hemophilic plasma samples using their in-house or specific assays.
Design/Method:
In this 2-part study, laboratories received sample sets (3–4 per laboratory) of 26 blinded samples in randomized order for analysis. Each set consisted of triplicate test samples of BAY 94-9027 or a comparator (antihemophilic factor [recombinant] plasma/albumin-free method [rAHF-PFM (Advate® ); Shire]) spiked at low (<10 IU/dL), medium (10–50 IU/dL), and high (50–100 IU/dL) concentrations in pooled hemophilic plasma. Normal control plasma and unspiked hemophilic plasma in triplicate were positive and negative controls, respectively. Two additional blinded samples matching 2 of the other 24 samples in the set were included in each set to decrease the predictability of each set. Laboratories analyzed test samples using their in-house assays (one-stage, chromogenic, or both), reagents, and standards (part 1). In part 2, all laboratories tested 2 additional sample sets using 2 activated partial thromboplastin time kits (Pathromtin® and HemosIL® SynthASil) previously shown to accurately measure BAY 94-9027 and full-length FVIII. FVIII recovery and FVIII levels were primary and secondary endpoints, respectively.
Results:
Fifty-two laboratories in North America, Europe, and Israel participated in the study. In part 1, 49 laboratories tested samples using the one-stage assay, 16 used the chromogenic assay, and 13 used both assays. The reagents routinely used for measuring FVIII activity varied among participating laboratories. Mean FVIII recovery ranged from 75.1%‒103.2% for BAY 94-9027 and 94.6%‒114.7% for rAHF-PFM across all concentrations and reagents using the one-stage assay. As expected based on previously published data, the PTT-A and HemosIL® APTT-SP kits underestimated BAY 94-9027 at all concentrations. More accurate one-stage results were generated using the Pathromtin® and SynthASil kits, as shown in part 2 of the study. For the chromogenic assay, mean FVIII recovery ranged from 104.4%‒117.1% for BAY 94-9027 and 87.7%‒107.8% for rAHF-PFM across all concentrations.
Conclusion:
BAY 94-9027 can be accurately monitored using the chromogenic assay and select commonly used one-stage assay kits without need of a conversion factor.
Patient Reported Outcomes, Burdens and Experiences (PROBE) Study Data Visualization and Analysis
Addressing issues of women and girls with blood disorders (WGBD) through a collaborative obstetrics/gynecology, adult and pediatric hematology lifespan clinic: a pilot project
Attitudes and Perspectives on Ankle Function in People with Hemophilia: A Qualitative Study
Background:
For people with hemophilia, mild trauma can cause internal joint bleeding resulting in stiffness and pain, limited range of motion and irreversible bony changes. Ankle pain may occur early in life affecting the ability to participate in activities of daily living, work and leisure. The purpose of this study was to explore experiences and priorities of people with hemophilia regarding their foot and ankle function, activity and participation.
Methods:
Eleven participants with hemophilia A or B, twenty-one years and older with a history of ankle pain, were recruited. Semi structured interviews were recorded, transcribed and then analyzed for themes with NVivo 10 Software.
Results:
Themes: (1) "Pain impacts my daily life, but I still have to get things done." (2) "Management of ankle function is highly individualized." (3) "Self-advocacy is crucial." (4) "I want healthcare providers who listen to me and respect my knowledge."
Conclusions:
For our participants, ankle pain and dysfunction impact daily life. Expressed themes highlighted priorities for participation, health management and desired healthcare.
Discussion:
As health care moves from volume-based to value-based care delivery, the patient’s voice is increasingly important in prioritizing interventions. The participant-identified priorities and experiences from our study can begin to inform healthcare providers, allowing them to deliver more targeted care for their patients with hemophilia.
Factors influencing uptake of evaluation among hemophilia carriers and potential carriers
A Study of Ethical Issues in the Bleeding Disorders Community (BDC)
Survey Finds Interest in Bleeding Disorder Social Work Specialty Credentialing
Chronicles of Caring: Nursing Stories from the Heart of Hemophilia
Access to Dental Care for People with Bleeding Disorders: Survey Results of Hemophilia Treatment Centers in the United States
Aim:
The goal of this project was to gather data and identify factors affecting access to dental care for people with bleeding disorders in the United States.
Methods:
The Arizona School of Dentistry and Oral Health and the National Hemophilia Foundation conducted a joint survey. The survey was completed by 102 of the 147 hemophilia treatment centers (HTC) in the USA. This represents 69% of the HTC’s in the country. Each HTC provided specific information concerning the dental services and education provided for patients.
Summary:
Survey results revealed inconsistent levels of oral health services available to patients. Major factors limiting access to care include finances, patient anxiety and a lack of providers with the skills to treat this population.
Conclusion:
Improvement in oral health for persons with bleeding disorders requires appropriate education for providers, patients and families. Additionally, both public and private health funding must be re-evaluated as it relates to people with bleeding disorders.
Building ‘Zoris in the Sand’ – Best Practice for Bleeding Disorder Capacity Building in the Underserved US Commonwealth of the Northern Mariana Islands
Intervention of Mexicans children and teenagers with hemophilia for their social integration
Adopting and Piloting the Patient Safety and Clinical Pharmacy Services Collaborative (PSPC) breakthrough model to transform and significantly improve adherence, improve health status and bring patients under control in the Hemophilia Community
Assessment of motor proficiency in people with bleeding disorders using the Bruininks-Oseretsky Test of Motor Proficiency, Second Edition (BOT-2™)
Background:
Activity limiting joint disease has greatly decreased with the introduction of prophylactic treatment for people with severe bleeding disorders. Previous research using the Bruininks-Oseretsky Test of Motor Proficiency, Second Edition (BOT-2TM), a standardized, normative, age and sex matched test of motor development, suggested motor development of males aged 4-21 years with bleeding disorders may be lower than age-matched peers.
Objective:
The primary purpose of this study was to compare the gross motor proficiency of boys with hemophilia ages 4-21 years followed at the Hemophilia Center at Oregon Health & Science University utilizing the BOT-2. Secondarily, we examined the relationship between joint health and gross motor proficiency.
Methods:
-
a) Participants and setting: Thirty-four subjects with either hemophilia A or B were recruited
from our center. Data collection occurred during clinic visits or at the patients’ homes.
-
b) Design and Procedures: A prospective, cross-sectional study design was used. The Upper Limb Coordination, Bilateral Coordination, Balance, Running Speed and Agility, and Strength subtests of the BOT-2 were administered. Body composition, range of motion, presence of an inhibitor, and use of prophylaxis were collected at the time of testing or from chart review.
-
c) Analyses: Analysis of variance (ANOVA) modeling was used to compare BOT-2 scores of PWH with baseline BOT-2 scores estimated from the general population of comparable age.
Summary:
Mean Running Speed & Agility scores were greater among boys with hemophilia compared to the control population (p=0.0026). Agility scores were similar between boys with hemophilia A and B (p>0.60), and significantly greater compared to the control group (p=0.0153). No other significant differences were found comparing boys with hemophilia to the control group. Within the boys with hemophilia cohort, age-adjusted ANOVA found no significant differences in BOT-2 scores between subjects of different severities, treatment regimen (prophylaxis or episodic), or diagnoses (Hemophilia A or B).
Conclusion:
Boys with hemophilia have the same or better gross motor proficiency as age matched peers.
Effect of hemophilia treatment center monitoring on bleeding rates
HEMO-milestones tool increases assessment of self-management competency and plan for skill building in patients with hemophilia
Attainment of self-management skills, disease knowledge, and a documented transition plan are key elements for smooth transfer from pediatric to adult care for patients with chronic disease. Our hemophilia center had no standardized approach for assessing key patient competencies or independence readiness. In the last quarter of 2014, our team undertook a quality improvement project; we developed a transition tool with the goals of consistently assessing self-efficacy skills at a sustained rate of 90% during annual comprehensive visits; and developing patient-centric skill-building plans based on these assessments.
We previously reported that after four PDSA ramps of tool utilization, 31 patients, age 2-21 years, had been seen in hemophilia comprehensive clinic during the 3 month period studied. Utilizing the newly developed tool, 97% had a global assessment of competency with respect to their current age group; 93% had transition or progress plans documented. This demonstrated significant improvement compared with retrospective data from the three month period prior to implementation of the tool.
Since this previous report, we have continued to utilize the tool during hemophilia comprehensive clinic. In the proposed poster, we will demonstrate sustainability of the tool through analysis and presentation of Hemo-milestone data from the first half of 2015. Further, based on these results we will propose additional areas of study inspired by the transition tool, with the aim of continued improvement of the skill-building and transition process. These include improved utilization of educational materials based on identified learning needs; evaluation of documented, planned interventions; the development of collaborative educational events with our partner adult HTC; and retrospective, patient- centered evaluation of the transition process after the transition to adult hemophilia care.
Update on a phase 1/2 open-label trial of BAX 335, an adeno-associated virus 8 (AAV8) vector-based gene therapy program for hemophilia B
An Evaluation of the Switch from Standard Factor VIII Prophylaxis to Prophylaxis with an Extended Half-Life, Pegylated, Full-length Recombinant Factor VIII (BAX 855)
Increased physical activity levels and improvement in treatment outcomes in patients who switch from on-demand to prophylaxis with BAX 855
SPACE (Study of Prophylaxis ACtivity, and Effectiveness): An interim descriptive analysis of patient activity levels and participation
Objective:
Personalizing treatment to a patient’s lifestyle and promoting overall health and wellness in persons with hemophilia (PWH) is essential to optimizing outcomes. There is limited evidence that correlates how activity and infusions impact bleeding episodes and further data on this relationship is needed. The research objective of SPACE is to prospectively explore the association between activity level, timing of infusion, and occurrence of a bleeding episode in PWH using novel technology.
Methods:
This six-month prospective, observational study includes PWH A or B in the United States currently receiving ADVATE or RIXUBIS between the ages of 13 and 65 years. Enrolled PWH use a smartphone eDiary application to log information on activities, infusions, and bleeding episodes. As an additional measurement of activity, enrollees are given a FitBit, a consumer-based activity tracker that measures steps taken and calories burned. Activity types are assessed based on their level of perceived risk for collision, according to the NHF “Playing It Safe” brochure. We report here current study status and descriptive analysis of baseline data.
Results:
The interim analysis included 15 patients with a median age of 19 (Range: 13 to 47). At baseline, 87% of patients were on prophylaxis and 13% treated on-demand treatment. Fifty-three percent of patients had 0 target joints at baseline. Eighty-seven percent of patients indicated that they had discussed activity participation with their physician. Sixty-seven percent of patients considered themselves ‘very satisfied’ or ‘satisfied’ with their level of activity. Data collected from the FitBit indicated that patients in SPACE walked on average 7,367 (SD: 3250) steps per day and burned 979 (SD: 398) calories from their activity. For patients on prophylaxis, the mean number of days per week doing mild, moderate and strenuous activity were 3.57, 2.64, and 1.5 respectively. Of the data reported on bleeding episodes, 40% of patients reported no bleeds at the time of the interim analysis. Forty percent of patients did not report having a bleed at the time of the interim analysis. Of all bleeds reported, 34% were associated with physical activity.
Conclusions:
Current data from SPACE demonstrates that subjects are active and participating in various activities. Continued data will provide better understanding of the types of activities and infusion schedules that may be associated with risk as well as protective effects on bleeding episodes by infusing prior to activity. A personalized approach to treatment based on physical activity levels may minimize bleeding risk in PWH.
Target Joint Spontaneous annualized bleed rate (sABR) Reduction: results from a pivotal trial of once weekly BeneFIX (nonacog alfa) in Hemophilia B subjects
Hemophilia B Patients Who Switch From rFIX to Extended Half-Life rFIX-Fc: A Retrospective Analysis of Cost using US Specialty Pharmacy Dispensing Data
Background:
Until recently, the hemophilia B community has only had standard Factor IX (FIX-Fc) products as treatment choices. With the availability of additional standard rFIX products and extended half-life (EHL) rFIX-Fc, there has been much debate (1-4) regarding clinical and cost expectations, balanced with optimizing use in patients. To level the debate, an understanding of real-world prescription costs of patients who switch to EHL FIX-Fc products is warranted.
Objectives:
1) To characterize patients who switch from rFIX to EHL rFIX-Fc, and 2) to understand their change in factor costs.
Methods:
Retrospective analysis using national US specialty pharmacy dispensing databases from Jan 1, 2013-Apr 25, 2015 of Hemophilia B (ICD-9 code 286.1) individuals who switched from rFIX to rFIX-Fc. Descriptive statistics summarized patient characteristics. Patients who had at least 90 days of prescription coverage on both products were included. Utilization was averaged on a monthly basis pre and post switch. Costs were calculated by multiplying the units by the wholesale acquisition prices for each product (Red Book) and the percentage change in cost from rFIX to EHL-rFIX was compared. This analysis studied patients who switched but stayed on similar regimens. Cohorts were characterized as prophylaxis to prophylaxis (P to P), or on-demand to on-demand (OD to OD).
Results:
Sixteen switchers were included in the study. Hemophilia severity was reported as 62.5% severe, 12.5% moderate, 6.25% mild, and 18.75% of unknown severity. The P to P cohort comprised of the majority of patients at 87.5% (n=14) while OD to OD were smaller at 11% (n=2). The median ages for these cohorts were similar. The P to P median age was 15 (range: 5-51) and OD to OD was 14 (range: 13-15) years old. Median prescription costs increased in the P to P cohort by 40% (range: -56% to 181%), while OD to OD increased by 173% (range: 1% to 348%).
Conclusion:
While the availability of EHL rFIX-Fc allows hemophilia B patients an option with less frequent infusions, our findings demonstrate that for those who initially switched from prophylaxis to prophylaxis or from on-demand to on-demand regimens, costs increased for the majority of patients. This analysis provides initial real-world cost data for those who have switched to EHL and may be helpful in decision makers’ understanding of the value of EHL rFIX-Fc in hemophilia B.
My Life, Our Future: A “Genetics Day” to Facilitate Efficient Enrollment
Efficacy and safety of pdFX, a new high-purity factor X concentrate, in patients with mild to severe hereditary factor X deficiency undergoing surgery
Patient Preferences for FVIII and BAX 855: Results from the BAX 855 Pivotal Trial
Objective:
BAX 855 is an extended half-life recombinant Factor VIII based on ADVATE. It was developed as an option to further personalize care and improve outcomes in people with hemophilia A. As a new therapy, it is important to understand patient satisfaction and preferences relative to their prior treatment. The objective of this research was to describe patient satisfaction with and preference for their prior FVIII therapy and BAX 855 as reported in the pivotal trial.
Methods:
BAX 855 was studied in a 6-month, phase 2/3, multi-center, open label study. Subjects in Arm A received 45 ± 5 IU/kg of BAX 855 twice weekly while subjects in Arm B infused BAX 855 on-demand. Patient preference and satisfaction were included as exploratory endpoints. Patient satisfaction was captured with the following options describing their treatment: very satisfied, satisfied, neither satisfied nor dissatisfied, or very dissatisfied at baseline for their prior FVIII therapy and at follow-up for BAX 855. Patient preference for BAX 855 or their prior FVIII therapy was captured at follow-up. Ethics approval and informed consent were obtained. The percent of very satisfied and satisfied patients were combined for this analysis. Results were reported for all patients and by treatment arm.
Summary:
138 patients were in the intent to treat (ITT) sample. The majority of patients in the ITT sample completed the satisfaction questionnaire, however the number varied slightly by question and visit. While 79.4% (100 of 126) of patients were satisfied with their prior FVIII at baseline, satisfaction increased to 90.1% (109 of 121) with BAX 855 and 84.7% (100 of 118) preferred BAX 855 over their prior FVIII. Of the 85 patients in Arm A previously on prophylaxis, 18 patients in Arm A previously on-demand, and 15 patients in Arm B previously on-demand, 83.5%, 88.9% and 86.7% preferred BAX 855 over their prior FVIII, respectively.
Conclusion:
Notably, patients enrolled in the BAX 855 trial had a high degree of satisfaction with their prior FVIII therapy. Nonetheless, treatment with BAX 855 was successful in further improving patient satisfaction with the majority of patients preferring it over their prior FVIII product.
Pharmacokinetics, safety, and efficacy of pdFX, a new high-purity factor X concentrate: a phase 3 study in patients with moderate or severe hereditary factor X deficiency
Initial Observations From the Pain, Functional Impairment, and Quality of Life (P-FiQ) Study: Patient-Reported Outcome Assessments in US Adults With Hemophilia
Objective:
To assess pain and functional impairment through 5 patient-reported outcome (PRO) instruments in non-bleeding adults with hemophilia.
Methods:
Adult men with hemophilia (mild-severe) with history of joint pain/bleeding completed a hemophilia/pain history and 5 PROs (EQ-5D-5L with visual analog scale [VAS], Brief Pain Inventory v2 [BPI-SF], International Physical Activity Questionnaire [IPAQ-SF], SF- 36v2, and Hemophilia Activities List [HAL]) during routine visits. Initial patients were asked to complete the PROs again after their estimated 3-4 hour visit (retest population). PRO scores were calculated from published algorithms. Generally, higher scores indicate better health- related quality of life (HRQoL) and functional status, and greater pain severity/interference.
Summary:
381 Patients were enrolled between October 2013 and October 2014; 164 of the initial 187 completed the retest and are reported here. Median time for completion of the initial survey/PROs was 36.0 minutes and 21.0 minutes for the PRO retest. Most retest subjects had hemophilia A (74.4%) and were white-non-Hispanic (72.6%). Median (Q1, Q3) age was 33.9 (26.9, 46.0), 48.7% were married, 62.6% had some college or graduate education, 80.7% were employed, and 61.0% were overweight or obese. HCV (49.4%) was more common than HIV (16.5%); 61.0% self-reported arthritis/bone/joint problems. Median EQ-5D- 5L VAS was 80.0 (0-100 scale), and EQ-5D-5L health index 0.80 (-0.11-1 scale); 61.6% reported any problems with mobility (29.3% reported moderate/severe problems), 55.8% with usual activities (18.4% moderate/severe), and 22.0% with self-care (4.3% moderate/severe). 73.2% reported pain-discomfort (43.3% moderate/severe), and 41.1% anxiety-depression (14.7% moderate/severe). For BPI, median pain severity was 3.0 (0-10 scale) and pain interference 2.9 (0-10 scale); median worst pain was 6.0, least pain 2.0, average pain 3.0, and current pain 2.0. Pain most impacted general activity, mood, walking ability, and normal work, and least impacted relations with other people. Ankles were the most frequently reported site of pain. Median IPAQ total activity was 693.0 MET/min/week; 49.3% reported no activity in the prior week. Median SF-36v2 scores (0-100 scale) were lower for physical health (39.6) than for mental health (51.6). Median overall HAL score was 76.1 (0-100 scale); complex lower extremity activities were the most impacted activity domain.
Conclusions:
These 5 PRO instruments provide different levels of detail describing the impact of hemophilia on pain and function, and consequently, have varied burdens of administration. PRO data from the retest population demonstrate that most adults with hemophilia experience pain and functional impairment that impacts HRQoL, highlighting the importance of assessments and patient dialogue.
Development of a novel patient-centered outcome measure in hemophilia using Goal Attainment Scaling
Background and Objective:
Standard outcome measures in hemophilia, such as the annual bleeding rate, have inherent limitations in both clinical practice and in research, including lack of both sensitivity and personalization. Goal attainment scaling (GAS) is a method for evaluating outcome that is patient-centered and sensitive to change in individuals and populations; collaborative goal setting may also be viewed through the biopsychosocial lens as a potential resource to strengthen disease-related coping. GAS has been used successfully in several disease areas. However, feasibility can be an issue. To address this, standardized goal attainment menus can be developed that preserve individualization, while allowing greater ease of use by non-experts. We initiated a process to develop a novel outcome measure in hemophilia, the GAS-Hem, by creating a goal attainment menu for patients living with hemophilia.
Methods:
We conducted 2 multidisciplinary workshops with participants from medicine, nursing, social work, and physical therapy (n=12). During the first workshop, we developed a list of goal areas, each with an associated set of descriptors of attainment levels. At the second workshop, a second group of experts (n=8) critically reviewed each goal to evaluate its importance and relevance for inclusion in the GAS-Hem.
Summary:
28 goal areas with associated descriptors of attainment levels were developed, covering 3 broad categories: ability to manage hemophilia, ability to recognize and treat complications, and ability to cope with the impact of hemophilia on daily life. Descriptors incorporated several key parameters for each goal (eg. skill level, desire for change, utilization of available resources) (Table).
Conclusions:
We developed a novel, patient- centered outcome measure for patients with hemophilia. A comprehensive, preliminary list of goal areas and attainment levels was successfully created. The next step will be to incorporate feedback from patients and families, after which a feasibility study will be conducted to evaluate content and construct validity and overall ease of use.
Table. Examples of Goal Areas and Descriptors
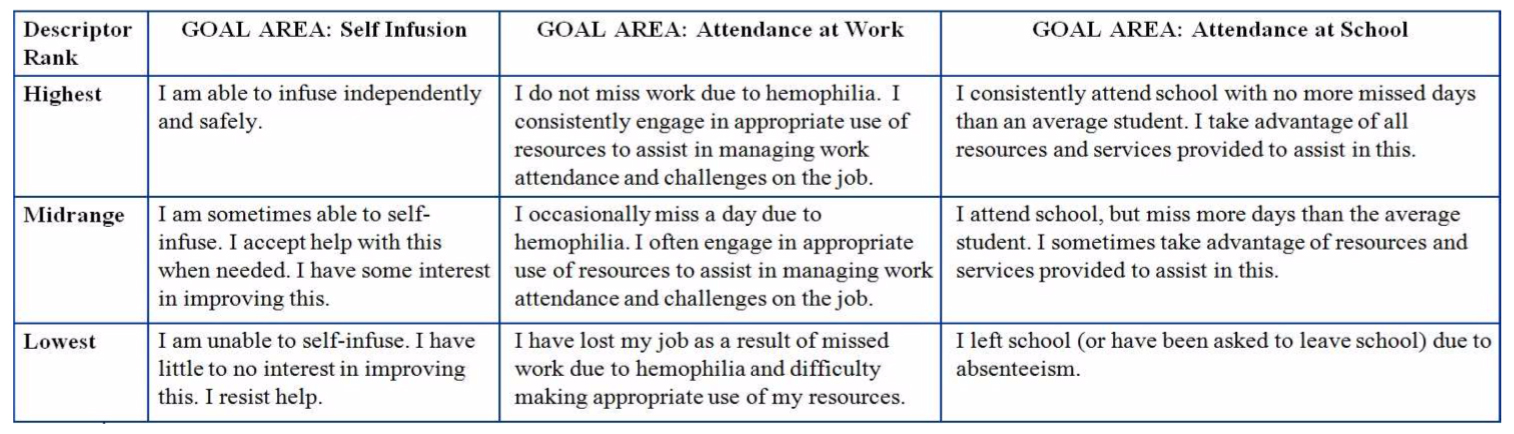
Leading an Active Lifestyle with Hemophilia B: Estimating the Bleeding Risk with Different FIX Treatment Regimens
A Novel Anti-Tissue Factor Pathway Inhibitor Antibody, BAY 1093884, Restores Hemostasis in Induced Hemophilia A Rabbits With Reduced Thrombogenic Potential
Background:
BAY 1093884 is a fully human monoclonal antibody against tissue factor pathway inhibitor developed as a bypass agent for hemophilia patients with inhibitors. It restores thrombin burst, leading to stable clot formation in hemophilic conditions in vitro and effectively stops bleeding in vivo.
Aims:
Efficacy and thrombogenic potential of BAY 1093884 were evaluated in bleeding models in acquired hemophilia A rabbits and the Wessler venous stasis model, respectively.
Methods:
The efficacy of BAY 1093884 was tested in rabbit bleeding models. Anti-factor VIII antibodies rendered rabbits hemophilic, increasing bleeding time by ~3-fold (median 120–390 seconds) in an ear bleeding model and >7-fold (median 240–1800 seconds) in a saphenous vein bleeding model. In the Wessler model, thrombosis was induced by complete ligation of the jugular vein after administration of BAY 1093884, recombinant FVIIa (rFVIIa), or activated prothrombin complex concentrate (aPCC) to measure in vivo hypercoagulability.
Results:
In both bleeding models, BAY 1093884 corrected bleeding time close to normal in a dose-dependent manner. A statistically significant reduction in saphenous vein bleeding was reached at 1 and 3 mg/kg BAY 1093884, reducing bleeding time to 330 and 240 seconds, respectively. In contrast, 1 mg/kg rFVIIa only slightly reduced bleeding time to a median of 1200 seconds. In the Wessler model, 1 mg/kg rFVIIa and 40 IU/kg aPCC resulted in thrombus formation, reaching full occlusion in rabbits with normal coagulation system. In contrast, BAY 1093884 up to 100 mg/kg did not fully occlude, and only showed a slight increase in localized clot formation over baseline, which could be completely prevented by an anticoagulant. In addition, no stasis-triggered thrombi were seen in hemophilia A rabbits treated with BAY 1093884, indicating reduced thrombogenicity risk for BAY 1093884 in patients with hemophilia.
Conclusions:
These studies showed that BAY 1093884 potently controls bleeding in hemophilic rabbits while showing reduced thrombogenic risk compared with the current standard of care.
Low Annualized Bleeding Rates in Phase 3 Studies of Recombinant Factor VIII Fc Fusion Protein (rFVIIIFc) in Subjects with Target Joints at Baseline
Introduction and Objectives:
The phase 3 A-LONG and Kids A-LONG studies demonstrated the safety and efficacy of rFVIIIFc for the control and prevention of bleeding episodes in subjects with severe hemophilia A. This subgroup analysis of A-LONG and Kids A-LONG examined bleeding frequency with rFVIIIFc prophylaxis in subjects with target joints at baseline.
Methods:
A-LONG (≥12 y) and Kids A-LONG (<12 y) subjects with a history of target joints (a major joint with ≥3 bleeding episodes in a 6-month period) who were previously treated with FVIII and received on-study rFVIIIFc prophylaxis were identified. Self-reported pre-study 12- month bleeding history and on-study annualized bleeding rate (ABR) for all bleeds were evaluated.
Results:
93/141 subjects in A-LONG and 13/69 subjects in Kids A-LONG had target joints at baseline. The most prevalent locations of pre-study target joints identified at baseline among subjects in A-LONG were the elbow (61.7%) and ankle (59.6%). Similarly, in Kids A-LONG, these were the ankle (69.2%) and elbow (30.8%). A total of 59.7% and 52.4% of subjects receiving individualized (Arm 1) and weekly (Arm 2) rFVIIIFc prophylaxis in A-LONG, respectively, and 53.8% of subjects receiving rFVIIIFc prophylaxis in Kids A-LONG with target joints at baseline had no target joint bleeding episodes on-study. Median on-study ABRs with rFVIIIFc prophylaxis tended to be lower than pre-study bleeding rates in subjects with target joints at baseline (Table).
Conclusions:
For subjects with severe hemophilia A who had target joints at baseline, rFVIIIFc prophylaxis lowered bleeding rates across age groups compared with pre-study FVIII treatment.
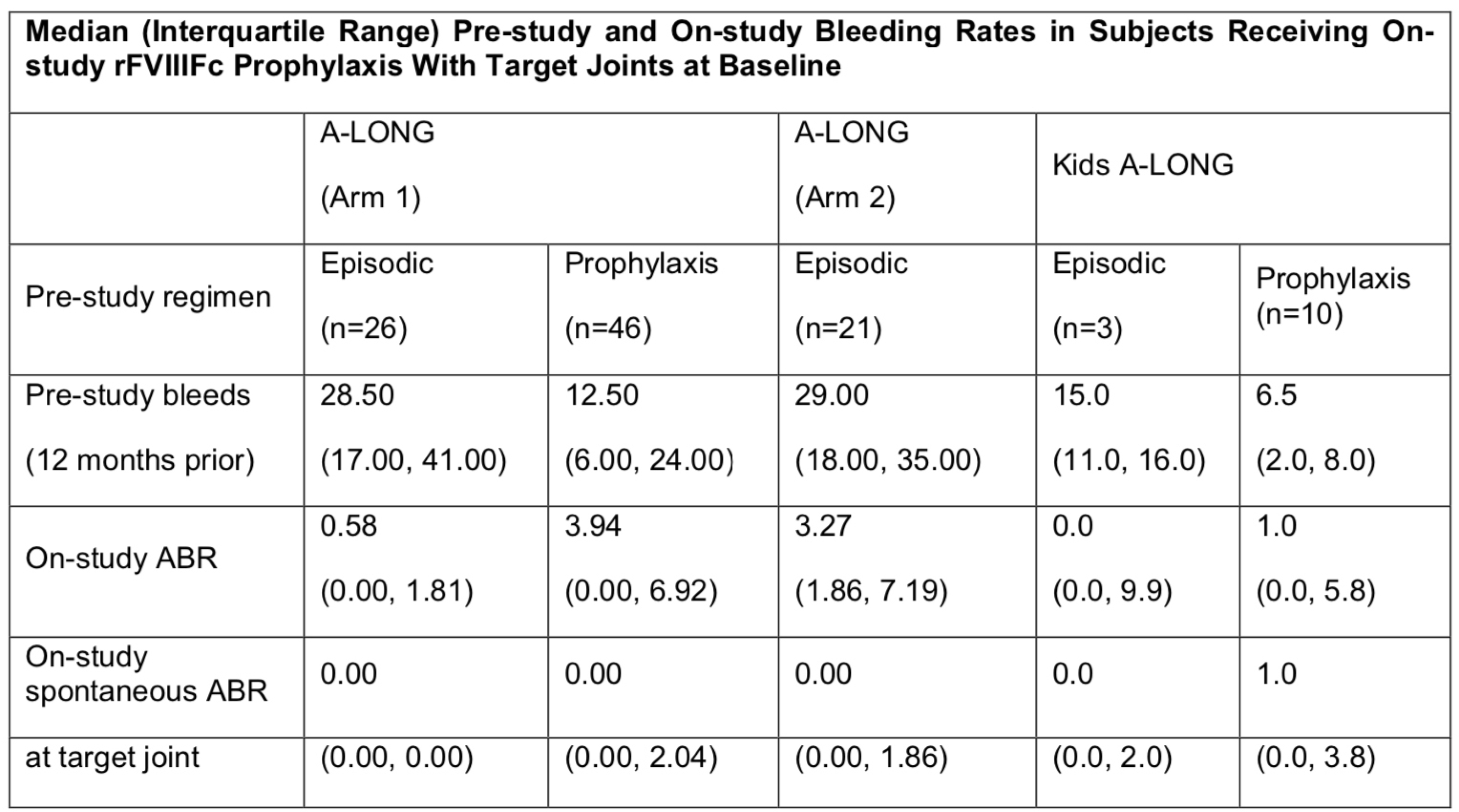
The impact of missing one or two infusions per month: a comparison of rFVIII and rFVIIIFc regimens
Hospitalization for Acute Bleeding Events among Individuals with von Willebrand Disease (VWD) in the United States
Description and Management of Pain and Functional Impairment in US Adults With Hemophilia: Initial Observations From the Pain, Functional Impairment, and Quality of Life (P-FiQ) Study
Objective:
Hemophilia is marked by frequent joint bleeding, resulting in acute and chronic pain and functional impairment. Surveys in US adults with hemophilia demonstrate suboptimal pain management and quality of life (QoL). The objective of P-FiQ was to methodologically assess QoL parameters, including functional impairment and pain, and pain management strategies.
Methods:
Adult men with mild-severe hemophilia with a history of joint pain and/or bleeding completed a hemophilia/pain history and various patient-reported outcome assessments (completed twice in “retest” population of initially enrolled patients).
Summary:
Of 164 adults with hemophilia (median age 34) in the “retest” population, more had hemophilia A (74%) than B (26%); 6% had inhibitors. Most had some college-or-above education (63%), 81% were employed, 61% were overweight/obese, and 61% self-reported arthritis/bone/joint problems. Current patient-reported treatment regimens were prophylaxis (42%), on-demand (39%), or mostly on-demand (19%; 25/31 using infusions ahead of activity). One-third (32.9%) reported unrestricted school/work/recreational activities in the prior 6 months; 6.2% reported needing assistance for school/work/self-care, or did not participate in recreational activities because of pain, loss of motion, and weakness. Some patients (31.3%) reported using a cane/crutches/walker (3.8% always) and 7.6% a wheelchair (1.3% always), and 47.0% reported a history of joint surgery (41 knee, 37 ankle, 29 elbow). Patients reported losing an average of 3.7 and 1.8 school/work days in the previous 6 months due to lower and upper extremity problems, respectively. Patients reported that during the prior 6 months they had experienced acute pain only (24%), chronic pain only (33%), or both (29%); 15% reported no pain. Acute pain was most frequently described as sharp (77%), aching (66%), shooting (57%), and throbbing (55%), and chronic pain as aching (74%), nagging (49%), throbbing (44%), and sharp (40%). Most common analgesics in the past 6 months for acute/chronic pain were acetaminophen (69%/58%), NSAIDs (40%/52%), hydrocodone-acetaminophen (29%/33%), oxycodone (12%/11%), and oxycodone- acetaminophen (9%/8%). Most common nonanalgesic treatment strategies reported for acute/chronic pain in the past 6 months were ice (73%/37%), rest (48%/34%), factor or bypassing agent (48%/24%), elevation (34%/28%), relaxation (31%/23%), compression (27%/21%), and heat (25%/15%); other reported strategies include medical marijuana (17%/9%), physical therapy (12%/9%), prayer (11%/8%), faith (9%/8%), alcohol (8%/7%), aquatherapy (5%/6%), illicit drugs (4%/2%), acupuncture (2%/1%), hypnosis (2%/1%), and biofeedback (1%/1%).
Conclusions:
Initial data corroborate the high prevalence of pain and functional disability in adults with hemophilia and highlight opportunities to address clinical assessment, patient dialogue, and management strategies to improve outcomes.
Cost of Treating Thrombotic Events in a US Population of Von Willebrand Disease Patients
Efficacy of a Recombinant Factor IX Investigational Product, IB1001 (trenonacog alfa) for Perioperative Management in Hemophilia B Patients
Objective:
To evaluate efficacy of IB1001 for perioperative management of bleeding under surgical circumstances in hemophilia B patients.
Methods:
The efficacy of IB1001 for perioperative management has been evaluated in a prospective, open-label, multicenter international study where 17 subjects (16 male PTPs and one female hemophilia B carrier) underwent 20 major surgical procedures. The PTPs were defined as patients with a minimum of 150 exposures to another factor IX preparation. The subjects had severe or moderately severe (factor IX level ≤ 2 IU/dL) hemophilia B without inhibitors, with exception of one mild hemophilia B female carrier. Efficacy of IB1001 was based on the surgeon’s assessment including estimation of blood loss as ‘less than expected’, ‘expected’, or ‘more than expected’ at the time of surgery and assessment of hemostasis at 12 and 24 hours post-surgery as ‘superior’, ‘adequate’, or ‘poorly controlled’. Transfusion requirements were also monitored.
Summary:
Thirteen major procedures in 12 male subjects were managed by bolus regimen, and 6 major procedures in 4 male subjects were managed by continuous infusion regimen. Mean loading dose for 13 major procedures managed by bolus regimen was 120 ± 11.4 IU/kg and mean maintenance bolus dose (given every 12 hours) was 59.7 ± 12.2 IU/kg. During the 24 hours following surgery, factor IX levels were successfully maintained over 60%, as intended. Factor IX levels at pre-infusion were 59.7% ± 15.9% at 12 hours after surgery and 54.4% ± 16.5% at 24 hours after surgery. For a major procedure in one female carrier, the bolus dose was 110 IU/kg, while the mean maintenance dose was 44.9 ± 7.0 IU/kg. Mean loading dose for 6 major procedures managed by continuous infusion regimen was 95.4 ± 14.5 IU/kg and the mean maintenance infusions were 7.1 ± 4.0 IU/kg/hr. In all major procedures, blood loss at the time of surgery was ‘expected’ or ‘less than expected’ as assessed by the surgeon. IB1001 was rated by the surgeon as ‘superior’ or ‘adequate’ in controlling hemostasis post-surgery, including 8 knee arthroplasties, two elbow arthroplasties, one knee amputation, one percutaneous Achilles tendon lengthening, one open inguinal hernia repair, one tibiotalar fusion, two arthroscopic synovectomies, three debridements and one total hysterectomy/bilateral oopherectomy. There were no transfusions required perioperatively.
Conclusions:
At the time of surgery, blood loss was expected or less than expected after IB1001 treatment, while post-surgery effective hemostasis control was achieved following IB1001 treatment in hemophilia B patients.
Efficacy of a Recombinant Factor IX Investigational Product, IB1001 (trenonacog alfa) in Previously Treated Patients with Hemophilia B
Objective:
To evaluate efficacy of IB1001 with respect to breakthrough bleeding during prophylaxis and with respect to control of hemorrhaging in prophylaxis and on-demand treatment regimens in previously treated patients (PTPs) with hemophilia B.
Methods:
The efficacy of IB1001 has been evaluated in a prospective, open-label, uncontrolled multicenter international study in which a total of 68 male PTPs between 7 and 64 years of age received IB1001 either as prophylaxis or on-demand treatment. All subjects had severe or moderately severe (factor IX level ≤ 2 IU/dL) hemophilia B without inhibitors. There were 61 subjects who received prophylaxis [predominantly secondary or tertiary prophylaxis as defined by World Federation of Hemophilia (WFH) guidelines] and 12 were treated on-demand during the study conduct. Both, the subjects and the study investigators rated the efficacy of IB1001 in management of bleeding episodes.
Summary:
The mean number of exposure days (ED) was 138.2 (median: 127.5 ED), including 55 subjects with ≥ 50 ED and 45 subjects with ≥ 100 ED. Subjects on prophylaxis received a mean intravenous dose of 55.0 ± 12.8 IU/kg IB1001 twice weekly, while subjects in the on- demand regimen received a mean dose of 60.0 ± 18.2 IU/kg IB1001. In the prophylaxis group, median annualized bleed rate (ABR) was 1.52, and in the on-demand group, median ABR was 16.39. There were 19/61 (31.1%) subjects on IB1001 prophylaxis who reported no bleeding episodes. A total of 508 bleeding episodes were treated with IB1001; 286 bleeds were recorded for patients on prophylaxis and 222 in the on-demand regimen. Majority of the bleeds (70.9%) resolved after a single IB1001 infusion, while 13% of the bleeds required two infusions. A minority of the bleeds (4.7%) required 5 or more infusions; these bleeds were predominantly related to trauma, target joints and/or muscle bleeds. Subjects rated efficacy of IB1001 as ‘excellent’ or ‘good’ in 84% of all treated bleeding episodes. Of 414 subject visits where the efficacy of IB1001 was evaluated by the study investigators, 92% were rated as ‘effective’ in prevention and treatment of bleeding by IB1001.
Conclusions:
IB1001 is effective for the treatment of hemophilia B either as secondary or tertiary prophylaxis or on an episodic (on-demand) basis. Based on clinical trial data, IB1001 is effective in controlling breakthrough or episodic bleeding episodes.
Personalization of Treatment Regimens for Physically Active Patients: A Comparison of Factor VIII and Extended Half-life Treatment Regimens
Changes in Child and Parent Ratings of Health-Related Quality of Life Among Children With Hemophilia A in the KIDS A-LONG study
Real-world dosing and patient characteristics of rFVIIIFc in hemophilia A patients
Objective:
To analyze real world rFVIIIFc patient characteristics and treatment interval patterns in patients with hemophilia A based on specialty pharmacy dispensing records.
Methods:
A retrospective analysis was conducted using aggregate Specialty Pharmacy Provider (SPP) records from July 2014 through Mar 2015. SPP data included 63 different attributes for each prescription, including trade name, NDC, quantity shipped, prescribed infusion dose, days supplied, and dose frequency. Patients were considered eligible for the analysis if they received at least one shipment of rFVIIIFc for a prophylactic treatment regimen. Patients were excluded from the analysis if they were being treated episodically, for immune tolerance induction, or pharmacy records did not specify a prescribed infusion dose. Patients were categorized according to their age and dosing interval.
Summary:
The analysis included 405 hemophilia A patients that received at least one shipment of rFVIIIFc with a median age of 23 (range: 2-68) and median weight of 70 kg (range: 10-154kg). Eight percent of dispensing records were for patients less than 6 years of age, 29% were between 6 and 17 years of age, and 63% were 18 years or greater. Pharmacy dispensing records represented 179 distinct prescribers across 40 states. Dosing frequency ranged from four times weekly to beyond once-weekly, with every four days as the most common dosing interval, representing 33% of patient records. The median infusion frequency was every four days. Fifty percent of all patients had a dosing frequency of four days or greater. Of the patients receiving rFVIIIFc that had previous rFVIII dispensing records, the most common rFVIII dosing frequency was three times per week. The majority of patients previously on prophylaxis regimen with a rFVIII dosing frequency of three times per week had a decreased number of prophylactic infusions per week on rFVIIIFc; 4% of patients reduced infusion frequency to every 3 days, 44% of patients reduced infusion frequency to twice weekly, 30% of patients reduced infusion frequency to every 4 days, 10% of patients reduced infusion frequency to every 5 days.
Conclusions:
Current SPP dispensing records demonstrate that rFVIIIFc is being used in a broad patient population based on age range and geographical distribution. Patients with hemophilia A in the US may experience reductions in FVIII infusion frequency when they switch to rFVIIIFc, with conversion to an infusion frequency every four days as the most common treatment regimen and the recommended prophylaxis starting dose according to the US Prescribing Information.
Quantitative research into people with hemophilia and caregiver perceptions of pre-filled solvent syringe (MixPro®)
Recombinant von Willebrand factor in severe von Willebrand disease: a prospective clinical trial
Objectives:
This trial evaluated the hemostatic efficacy, pharmacokinetics, and safety of a recombinant VWF (rVWF) in adults with severe von Willebrand disease (VWD) (type 3 [VWF:Ag ≤ 3 IU/dL], severe type 1 [VWF:RCo< 20 IU/dL], 2A [VWF:RCo< 20 IU/dL], 2N [FVIII:C<10% ], 2B, or 2M).
Methods:
Bleeds were to be treated initially with rVWF and rFVIII (1.3:1 ratio), followed by rVWF alone (as long as FVIII:C >40%). Hemostatic efficacy was evaluated using a pre- defined 4-point rating scale (none=4, moderate=3, good=2, excellent=1). PK parameters for rVWF vs. rVWF:rFVIII were assessed in a randomized crossover design, and a 6-month repeated PK evaluation for rVWF alone.
Summary:
Of 31 subjects assigned to bleed treatment, 22 (17 type 3, 4 type 2A and 1 type 2N) experienced 192 bleeding episodes (several in multiple locations) that were treated with rVWF: 106 occurred in mucosal tissue (including 32 menorrhagic, 42 nasopharyngeal, 26 mouth/oral bleeds), 59 joint bleeds, 6 gastrointestinal bleeds, and 37 in other locations. Treatment success (mean efficacy rating <2.5) was achieved in all 22 subjects (100%; Clopper-Pearson exact 90% confidence interval: 87.3 to 100.0). ‘Excellent’ ratings were given for 186/192 (96.9%) bleeds (119/122 minor, 59/61 moderate, 6/7 severe, 2/2 unknown severity) and the remaining 3.1% were ‘good’. One infusion was effective in 81.8% of bleeds (median [range]: 1 [1-4] overall; 2 [1-3] for severe bleeds). The subject’s own assessment of treatment efficacy (an exploratory endpoint), was ‘excellent’ within 8 hours after the first infusion for 125/134 (93.3%), ‘good’ for 8/134 (6.0%) and ‘moderate’ for 1/134 (0.7%) bleeding episodes. A substantial increase in FVIII:C and sustained stabilization (> 40% by 6 hours, rising to >80% 24 hours post-infusion) was observed after infusion with rVWF. PK parameters for VWF Ristocetin cofactor (VWF:RCo, a surrogate for the platelet-dependent function of rVWF) were similar when rVWF was infused alone (mean T1/2 21.9 h vs. 19.6 h with rVWF:rFVIII). Eight adverse events (tachycardia, infusion site paraesthesia, ECG t wave inversion, dysgeusia, generalized pruritis, hot flush, chest discomfort and increased heart rate) in 5 subjects were assessed as related to rVWF. No inhibitor or anti-VWF binding antibody development was observed, and there were no thrombotic events or severe allergic reactions.
Conclusions:
rVWF was safe, well-tolerated and effective in the treatment of a variety of bleeding presentations in severe VWD. The sustained stabilization in FVIII:C after the initial infusion enables subsequent infusion of rVWF without rFVIII, when multiple infusions are required.
Kids B-LONG: Safety, Efficacy, and Pharmacokinetics of Long-Acting Recombinant Factor IX Fc Fusion Protein (rFIXFc) in Previously Treated Children with Hemophilia B
Objective:
Kids B-LONG was an open-label phase 3 study that evaluated the safety, efficacy, and pharmacokinetics (PK) of rFIXFc in previously treated children (aged <12 years; ≥50 prior exposure days [EDs] to FIX) with severe hemophilia B (≤2 IU/dL endogenous FIX) and no history of FIX inhibitors.
Methods:
Participants initiated prophylactic treatment with 50–60 IU/kg rFIXFc once-weekly; dose and interval adjustments were based upon PK data and bleeding frequency. The primary endpoint was development of inhibitors (neutralizing antibodies). Key secondary outcomes included PK and annualized bleeding rate (ABR).
Summary:
The study enrolled 30 participants (<6 years of age, n=15; 6 to <12 years of age, n=15); 90% completed the study. Prestudy, all participants received FIX prophylaxis (23/30 were dosing ≥2x/week). The median time on study was 49.4 weeks; 24 participants had ≥50 rFIXFc EDs. No participant developed inhibitors to rFIXFc. The pattern of adverse events reported was typical of the population studied. There were no serious allergic reactions and no thrombotic events. No serious adverse events were assessed by the investigator as related to rFIXFc. The terminal half-life (geometric mean [95% CI]) of rFIXFc was 66.5 (55.9, 79.1) hours in the <6 years cohort (n=11) and 70.3 (61.0, 81.2) hours in the 6 to <12 years cohort (n=13). The geometric mean (95% CI) half-life of prestudy BeneFIX was 18.2 (15.5– 21.3) hours in the <6 years cohort (n=11) and 19.2 (17.6–20.9) hours in the 6 to <12 years cohort (n=9). Median (IQR) ABR was 1.97 (0.00, 3.13) overall, and 0.00 (0.00, 1.16) for spontaneous bleeds; 33.3% of participants reported no bleeds on study. At study end, 97% of participants were dosing once weekly. The median (IQR) total weekly prophylactic dose with rFIXFc was 59.4 (53.0, 64.8) IU/kg and 57.8 (51. 7, 65.0) IU/kg, in the <6 years and 6 to <12 years cohorts, respectively. The prestudy FIX median (IQR) total weekly prophylactic dose was 110.0 (58.0, 188.0) IU/kg and 100.0 (58.0, 120.0) IU/kg in the <6 years and 6 to <12 years cohorts, respectively. 75.0% of bleeding episodes were controlled with 1 infusion; 91.7% with 1 or 2 infusions (median average dose per infusion: 68.22 IU/kg).
Conclusions:
rFIXFc was safe and effective for the prevention and treatment of bleeding in children with severe hemophilia B. Study participants achieved low bleeding rates with extended-interval rFIXFc prophylaxis, while reducing their weekly prophylactic factor consumption compared with their prior FIX regimen.
The Effects of Hemophilia on Socialization
Global Knowledge and Confidence Assessment of Hemophilia Clinical Practice Approaches Among Pediatricians
Objective:
Clinical knowledge gaps of hemophilia can affect patient outcomes through delayed diagnosis/referral as well as improper monitoring and interventions. A study was undertaken to identify and characterize clinical practice gaps and confidence levels in the management of hemophilia specific to pediatricians.
Methods:
Building upon a previous assessment developed in 2014, an updated global, hemophilia-specific continuing medical education-accredited clinical practice assessment survey was developed utilizing current evidence-based consensus guidelines and best practices, including guidelines from the National Hemophilia Foundation and the World Federation of Hemophilia. The assessment included both knowledge- and case -based, multiple-choice questions that healthcare providers completed confidentially on-line between March 23, 2015 and April 9, 2015. Areas such as appropriate triggers for initiating prophylaxis and use of physical therapy were assessed. Responses from pediatric providers were de- identified and aggregated prior to analysis.
Summary:
660 pediatricians (30% of total respondents) completed the survey, from the following locales: North America (36%), Asia (23%), Europe (15%), Middle East (10%), Africa (7%), Central/South America (6%), and Australia (4%). Academic (31%), private practice (27%), community hospital (19%), community clinic (12%), and hemophilia treatment center (3%) practice settings were identified. Analysis of pediatricians who indicated professional interaction with hemophilia patients (87% of pediatrician respondents) demonstrated knowledge gaps including (% incorrect responses): classification of severity of hemophilia (37%); optimal use of prophylactic therapy, e.g., when to initiate (31%), at what dose (53%), prophylaxis in active patients (26%); likelihood of inhibitors (75%); using bypassing therapy (58%); comprehensive care model (61%); supporting overall joint health and quality of life (70%); and adherence (60%). A low level of confidence in the ability to identify when to use prophylaxis was reported among 31% of pediatricians. The top barriers to the administration of prophylaxis identified by the pediatric providers included lack of availability of FVIII or FIX concentrates, lack of venous access, and insurance coverage (29%, 22%, and 21% for respondents, respectively).
Conclusions:
This study demonstrated gaps in knowledge and confidence about the assessment and optimal care of hemophilia for pediatricians, suggesting that further education specific to the needs of these providers is warranted.
A Novel Anti-Tissue Factor Pathway Inhibitor Antibody, BAY 1093884, Prevents Bleeding in Hemophilia A Mice
Background:
BAY 1093884 is a fully human monoclonal antibody against tissue factor pathway inhibitor (TFPI) developed as a bypass agent for hemophilia patients with inhibitors. It restores thrombin burst for stable clot formation in hemophilic conditions in vitro.
Aims:
The goal of this study was to elucidate the in vivo prophylactic hemostatic potency of BAY 1093884.
Methods:
The hemophilia A mouse tail vein transection model was used for this study to mimic the venous bleeding characteristics of severe hemophilia. Male hemophilia A mice (n=12/group) were treated prophylactically with escalating doses of BAY 1093884. All treatments were administered as single intravenous (IV) bolus at 4 different time courses prior to injury (1, 3, 5, and 7 days). Bleeding was induced by a 1.2-mm deep incision across the left lateral vein. Following injury, the animals were monitored hourly for moribundity for up to 9 hours and after 24 hours followed by euthanasia.
Results:
Efficacious doses of BAY 1093884 providing 50% protection for survival (ED50) for up to 6 days after IV bolus ranged from 0.6–2 mg/kg. In comparison, only 25% protection was achieved 6 days after the treatment with a very high dose of 1000 IU/kg of recombinant factor VIII (rFVIII). The effect of a single IV dose of 18 mg/kg BAY 1093884 providing 80%–90% survival was maintained over 8 days (ED50 6 mg/kg), whereas a single IV dose of rFVIII failed to provide protection over an 8-day period.
Conclusions:
These studies demonstrate that BAY1093884 prevents bleeding and increases long-term survival to a greater degree than rFVIII in hemophilia A mice and may offer a convenient prophylactic treatment option for hemophilia patients with inhibitors.
Reduced Polyethylene Glycol–Conjugated B-Domain–Deleted Factor VIII (PEG-BDD-FVIII) Clearance: Selective PEG Steric Modulation Without Affecting Potency
Pegylated full-length recombinant factor VIII (BAX 855) for prophylaxis in previously treated adolescent and adult patients with severe hemophilia A
Objective:
To assess the pharmacokinetics (PK) and efficacy of prophylactic treatment with BAX 855 - a novel polyethylene glycol (peg)ylated full-length recombinant factor VIII, built on the rAHF-PFM (ADVATE) protein - by age group in previously-treated male patients with severe hemophilia A.
Methods:
Adolescent (12 to <18 years) and adult (18 to 65 years) subjects received 45 ± 5 IU/kg BAX 855 twice weekly as prophylaxis for approximately 6 months. PK was assessed in a subgroup (n=25 planned, including ≥6 adolescents) for one ADVATE infusion, then for BAX 855 at the initial infusion and after ≥50 exposure days (EDs). Efficacy was assessed in all subjects.
Summary:
Twenty-six subjects (8 adolescents, 18 adults) were included in the PK evaluation, and 121 (23 adolescents, 98 adults) were included in the efficacy analysis (all subjects assigned to treatment, i.e. full analysis set). The extended half-life (T1/2) and mean residence time (MRT) of BAX 855 (initial dose) compared to ADVATE were demonstrated by fold increases in the means of 1.4 and 1.5, respectively in both adolescents and in adults, using the one-stage clotting assay. The initial and repeat PK assessments of BAX 855 showed similar results. Consistent trends were observed when PK was determined using the chromogenic assay. The arithmetic mean (SD) annualized bleeding rate (ABR) during prophylaxis with BAX 855 was higher in adolescents than in adults (6.2 [6.1] versus 3.2 [4.2]). ABRs for injury-related bleeding episodes (BEs) were higher in adolescents (arithmetic mean [SD]:2.3 [3.2] versus 1.7 [3.1] in adults), thus contributing to the overall higher ABR in this group. Joint ABRs were lower in adolescents (arithmetic mean [SD]: 1.8 [2.5] versus 3.2 [8.8] in adults) with an inverse relationship for non-joint ABRs. Six adolescents (26.1%) and 39 adults (40.2%) had zero BEs. A total of 48 BEs occurred in adolescents (20 minor; 26 moderate; 2 severe); 182 occurred in adults (69 minor; 103 moderate; 10 severe). The hemostatic efficacy of BAX 855 was rated excellent or good at resolution for the majority of BEs in both age groups (93.8% in adolescents; 92.9% in adults).
Conclusions:
Although fewer adolescents than adults were included in the study, the data suggest that BAX 855 is efficacious in both age groups for twice-weekly prophylaxis and control of BEs. As expected, joint ABR was higher in the adult group.
Long-term safety and prophylactic efficacy of once-weekly subcutaneous administration of ACE910 in Japanese hemophilia A patients with and without FVIII inhibitors: Interim results of the extension study of a phase I study
Relative Health Status of Young Adults in the Hemophilia Utilization Group Studies (HUGS)
Associations Between Annual Bleeding Episodes and Financial Burden of Illness Among Persons with Hemophilia A and B in the United States
Objective:
To evaluate the effect of bleeding episodes on hemophilia patient burden of illness using observational data.
Methods:
Between 2005-2007 and 2009-2012, the Hemophilia Utilization Group Studies Va and Vb, respectively, recruited patients from ten Hemophilia Treatment Centers (HTCs) in eleven states. Adult patients or parents of children with hemophilia A or B completed an initial survey assessing socio-demographics, clinical characteristics, and treatment regimen. Work absenteeism, underemployment due to hemophilia, and unpaid hemophilia-related caregiver time were recorded at regular intervals over two years to estimate indirect costs using the human capital approach. Direct costs were estimated using healthcare services utilization and drug dispensing records. All costs were annualized and converted to 2014 US dollars. Annual mean bleeding episodes were calculated from patient-reported number of bleeds recorded in follow-up interviews, and used to stratify patients into bleeding categories of 0, 1-3, 4-6, 7-9, and 10+ bleeds. Associations between bleeding episodes and healthcare utilization, work productivity losses, and total costs were analyzed in patient subgroups based on both severity and treatment regimen.
Results:
Of 477 recruited patients, 352 with complete healthcare utilization and dispensing records and at least three months of follow-ups were included. A larger proportion of hemophilia A patients had severe disease and used prophylaxis compared to those with hemophilia B, but no socio-demographic variables differed significantly between the groups. Among severe patients, adults compared to children and episodic treatment compared to prophylaxis users had significantly more average annual bleeds [respective mean(standard deviation): 16.24(13.9) vs 5.54(9.47), p<0.0001 and 15.69(12.65) vs 8.39(11.1), p<0.0001]. Nearly two-thirds of the 82 severe patients using episodic treatment (63.4%) had 10 or more annual bleeds. Higher bleeding categories (more annual bleeds) were significantly associated with higher annual mean indirect costs for mild/moderate patients using episodic treatment [mean range across categories: $423-$21,434 (p=0.0003)] and severe patients on prophylaxis [$6,467-$14,890 (p=0.005]. Increased bleeding was also significantly associated with higher mean total costs in episodic treatment users with mild/moderate disease [$17,373- $136,552 (p<0.0001)] and severe disease [$83,957-$226,614 (p=0.008)]. Across all subgroups, increased bleeding was associated with more emergency room visits, outpatient procedures, and missed days of work, oftentimes reaching statistical significance.
Conclusions:
A larger proportion of severe hemophilia patients treating episodically have poor bleed management compared to those on prophylaxis. Overall, higher bleeding frequency is associated with both higher direct and indirect costs for individuals with hemophilia A and B across disease severity and treatment regimen.
Long-term safety and efficacy of recombinant factor VIII Fc (rFVIIIFc) for the treatment of severe hemophilia A: United States subgroup interim analysis of the ASPIRE study
Objective:
The ongoing rFVIIIFc extension study, ASPIRE (clinicaltrials.gov #NCT01454739), evaluates the long-term safety and efficacy of rFVIIIFc in adults, adolescents, and children with severe hemophilia A. Here we report interim outcomes for United States (US) subjects in ASPIRE.
Methods:
Eligible subjects could enroll in ASPIRE upon completing A-LONG or Kids A-LONG. There were 4 treatment groups: individualized prophylaxis (25-65 IU/kg every 3-5 days, or 20-65 IU/kg on D1, 40-65 IU/kg on D4 if twice weekly); weekly prophylaxis (65 IU/kg every 7 days); modified prophylaxis (to further personalize and optimize treatment when needed); or episodic treatment. Subjects could change treatment groups at any time. Subjects <12 yrs participated only in individualized and modified prophylaxis groups. Primary endpoint: development of inhibitors. Secondary outcomes included annualized bleeding rate (ABR) and rFVIIIFc exposure days (EDs).
Summary:
Sixty subjects (49 from A-LONG; 11 from Kids A-LONG) enrolled. As of the interim data cut (6 Jan 2014), the median time on ASPIRE was 80.37 (A-LONG) and 15.94 (Kids A-LONG) wks; 82% (A-LONG) and 27% (Kids A-LONG) of subjects had ≥100 cumulative rFVIIIFc EDs. 7/49 A-LONG subjects changed treatment groups upon enrollment into or during ASPIRE; 2 Kids A-LONG subjects switched from individualized to modified prophylaxis upon enrollment into ASPIRE. Median ABRs were low with rFVIIIFc prophylaxis (Table). Overall, most subjects treated prophylactically during the parent study did not experience changes to their total weekly prophylactic dose or dosing interval during ASPIRE. For subjects who enrolled from A-LONG and Kids A-LONG, 94% and 100% of all bleeding episodes during ASPIRE, respectively, were controlled with 1 infusion. In the overall study population, no inhibitors were observed during ASPIRE; adverse events were typical of the general adult and pediatric hemophilia A populations.
Conclusion:
Interim data from US subjects in ASPIRE are consistent with those of the phase 3 parent studies and the overall ASPIRE interim analysis. Results from ASPIRE confirm the longer-term safety of rFVIIIFc and the maintenance of a low ABR with extended interval prophylactic dosing in individuals with severe hemophilia A.
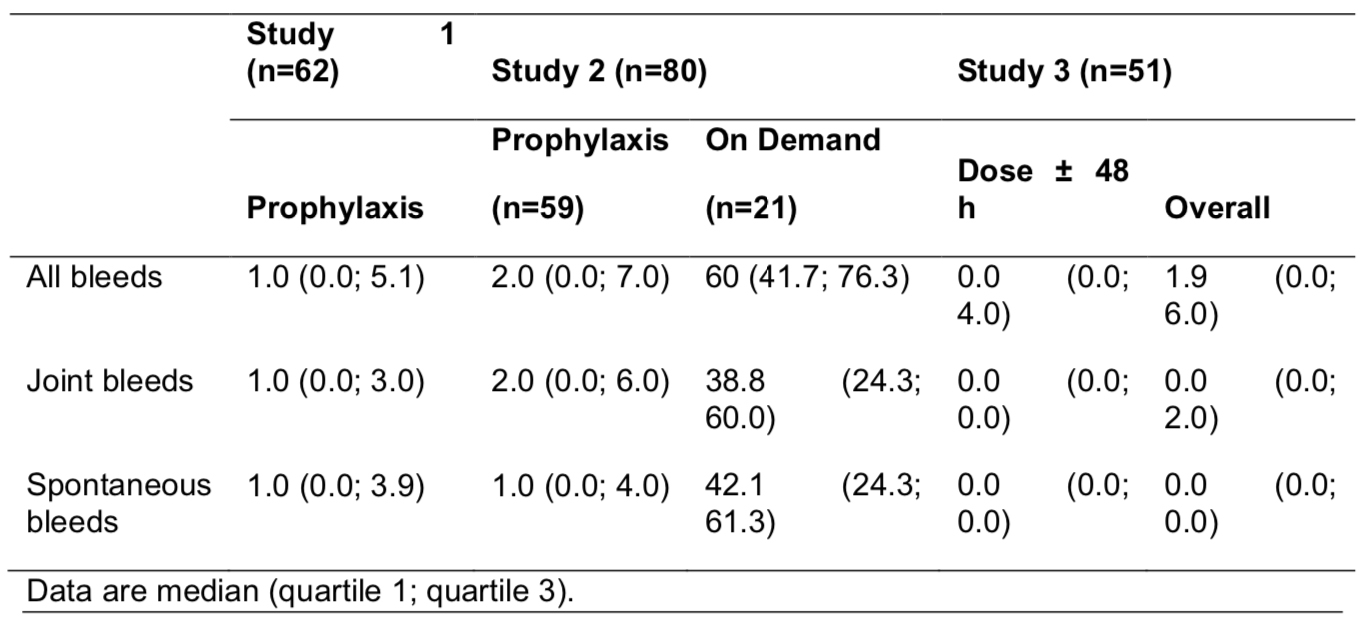
Increase or Maintenance of Physical Activity in Patients Treated with Recombinant Factor IX Fc Fusion Protein (rFIXFc) in the B-LONG and Kids B-LONG Studies
Patients Treated with Recombinant Factor VIII Fc Fusion Protein (rFVIIIFc) Reported Increased or Maintained Physical Activity in the A-LONG and Kids A-LONG Studies
Introduction and Objectives:
In the phase 3 A-LONG and Kids A-LONG studies, subjects with severe hemophilia A receiving rFVIIIFc prophylaxis 1-2 times/week had low annualized bleeding rates (ABRs), with comparable pre-study and on-study weekly factor consumption for subjects previously on FVIII prophylaxis. This report evaluated the effect of rFVIIIFc on subjects’ physical activity across a variety of age groups using a subject-reported assessment.
Methods:
Subjects eligible for A-LONG (≥12 y) and Kids A-LONG (<12 y) were previously treated males with severe hemophilia A (<1 IU/dL endogenous FVIII activity). Subjects in A- LONG were enrolled into 1 of 3 arms: Arm 1, individualized prophylaxis; Arm 2, weekly prophylaxis; or Arm 3, episodic treatment. All subjects in Kids A-LONG received rFVIIIFc prophylaxis. There were no restrictions regarding physical activity. Physical activity assessments were conducted at Weeks 7, 14, 28, 38, 52, and end of study (A-LONG) and Weeks 2, 7, 12, 17, 22, 26, and end of study (Kids A-LONG). At each visit after their first rFVIIIFc dose, subjects were asked to report any changes in their activity levels relative to their prior study visit as: more (or more intensive), fewer (or less intensive), or about the same amount of physical activities. To summarize each subject’s change in physical activity during the study compared to baseline, subjects’ reports were classified into four groups: less, the same, more, or undetermined.
Results:
A total of 165 and 71 subjects enrolled in A-LONG and Kids A-LONG, respectively. Overall, the majority of subjects in A-LONG reported more or the same amount of physical activity, and few subjects reported less physical activity during the study (less, the same, more, undetermined in Arm 1 [n=117], 8%, 36%, 51%, 5%; Arm 2 [n=23], 9%, 48%, 39%, 4%; Arm 3 [n=23], 9%, 52%, 26%, 13%, respectively). Results were generally similar for subjects in Kids A-LONG (for subjects aged <6 y [n=35], 3%, 26%, 66%, 6%; for subjects aged 6 to <12 y [n=34], 9%, 26%, 56%, 9%).
Conclusions:
The majority of subjects in A-LONG and Kids A-LONG reported similar or increased physical activity levels during the studies, while maintaining low ABRs. These self- reported data suggest that subjects across a variety of age groups with severe hemophilia A who are transitioning to rFVIIIFc may maintain or increase physical activity levels, while reducing infusion frequency and maintaining similar weekly factor consumption, without compromising efficacy.
Patient Satisfaction with US Hemophilia Treatment Centers 2015: National Results
Impact of FVIII CRM-positive status on the immunogenicity of FVIII in the hemophilia A mouse model
BAY 81-8973: Safety Observations in Clinical Trials in Adults, Adolescents, and Children With Severe Hemophilia A
Introduction:
BAY 81-8973 is Bayer’s new full-length recombinant factor VIII (FVIII) product in development for the treatment of hemophilia A. BAY 81-8973 has no human- or animal- derived raw materials added to the cell culture, purification, or formulation processes.
Objective:
To evaluate the safety profile of BAY 81-8973 as documented in clinical trials with BAY 81-8973.
Methods:
The safety of BAY 81-8973 for prevention and treatment of bleeds in patients with severe hemophilia A (<1% FVIII) was evaluated in 3 clinical studies: 2 in adolescent and adult previously treated patients (PTPs; aged 12–65 years with ≥150 previous exposure days [EDs]) and 1 in pediatric PTPs (aged ≤12 years with ≥50 previous EDs). A total of 193 PTPs (including 51 pediatric patients) were included to assess the frequency of adverse reactions in the 3 phase 3 studies. The immunogenicity of BAY 81-8973 was also evaluated in PTPs. Adverse reactions were collected and analyzed throughout the studies.
Results:
The frequency, type, and severity of adverse reactions in children were similar to those in adults. Adverse reactions in PTPs are listed in Table 1. In PTPs evaluated to date for immunogenicity, no inhibitors were detected.
Table 1. Adverse Reactions in Previously Treated Patients (N=193)

Conclusions:
The incidence of treatment-related adverse and serious adverse reactions in the BAY 81-8973 trials was low (<10%) and similar to those reported in the literature for other full-length FVIII products. No PTP developed a FVIII inhibitor.
A Half-Life Extended Fusion Peptide Inhibitor of TFPI Improves Hemostasis in Preclinical Models of Hemophilia
Prevalence of high BMI in school age children with hemophilia
BAY 81-8973: The Effects of Routine Prophylaxis on Total, Joint, and Spontaneous Bleeding in Adolescents, Adults, and Children With Severe Hemophilia A
Introduction:
BAY 81-8973 is Bayer’s new full-length recombinant factor VIII (FVIII) product for the treatment of hemophilia A, with no human- or animal-derived raw materials added to the cell culture, purification, or formulation process.
Methods:
The safety and efficacy of BAY 81-8973 for routine prophylaxis in patients with severe hemophilia A (<1% FVIII) was evaluated in 3 clinical studies: 2 in adolescent and adult previously treated patients (PTPs; aged 12–65 years [Study 1 and Study 2]) and 1 in children aged ≤12 years (PTPs: completed [Study 3]; previously untreated patients: ongoing), enrolling a total of 204 patients. In Study 1, the prophylaxis regimen was 20–50 IU/kg 2x–3x/wk as determined by the investigator. In Study 2, patients were randomized to prophylaxis (20–30 IU/kg 2x/wk or 30–40 IU/kg 3x/wk) or on-demand treatment. In both 12-month studies, the primary efficacy variable was the annualized bleeding rate (ABR). In Study 3, the prophylaxis regimen was 20–50 IU/kg 2x–3x/wk or every other day, as determined by the investigator, for ≥50 exposure days (approximately 6–8 months). The primary efficacy variable was the ABR for total bleeds occurring within 48 hours of previous prophylaxis treatment; ABR was also analyzed independent of time of injection.
Results:
A total of 193 patients treated with BAY 81-8973 were included in the analysis (Table 1). Sixteen (25.8%), 16 (27.1%), and 23 (45.1%) patients treated prophylactically had 0 bleeding episodes in Studies 1, 2, and 3, respectively.
Table 1. Annualized Bleeding Rates
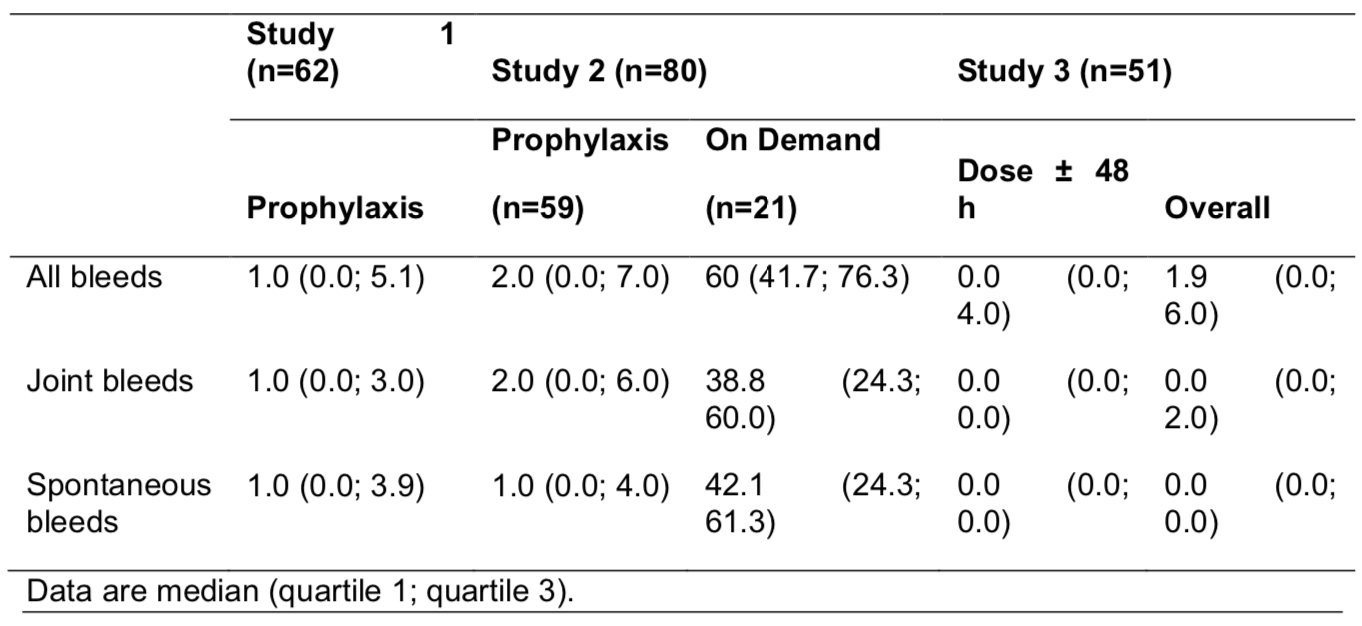
Conclusions:
Study 1 and Study 2 demonstrated the efficacy and safety of routine prophylaxis treatment with BAY 81-8973 in adolescents and adults. Study 2 also demonstrated the superiority of prophylaxis over on-demand treatment during a one-year treatment period. An additional study in children aged ≤12 years showed low annualized rates for all bleeds, joint bleeds, and spontaneous bleeds within 48 hours after injection or later during routine prophylaxis.
SPINART 3-Year Analyses: Patient- and Joint-Level Changes in Colorado Adult Joint Assessment Scale and Magnetic Resonance Imaging Scores With Bayer’s Sucrose-Formulated Recombinant Factor VIII (rFVIII-FS) in Adolescents and Adults
BAY 81-8973: Pharmacokinetic Parameters in Adolescents, Adults, and Children With Severe Hemophilia A
Introduction:
BAY 81-8973 is Bayer’s new full-length recombinant factor VIII product in development for the treatment of hemophilia A, with no human- or animal-derived raw materials added to the cell culture, purification, or formulation process. The pharmacokinetic (PK) properties of BAY 81-8973 were investigated in 3 studies in previously treated adults, adolescents, and children.
Methods:
For all PK evaluations, a single dose of 50 IU/kg BAY 81-8973 was injected. Serial blood samples were collected over 48 hours in adults and 24 hours in children <12 years of age. PK samples in adolescents and adults were analyzed using one-stage and chromogenic assays. Limited samples were collected in children and were analyzed using only the chromogenic assay. Ethnic subgroups included Chinese, Japanese, and non-Asian patients.
Results:
PK parameters using the chromogenic assay for children (aged <12 years), adolescents (aged 12–17 years), and adults (aged ≥18 years) are shown in Table 1.
Table 1. Pharmacokinetic Parameters Based on the Chromogenic Assay
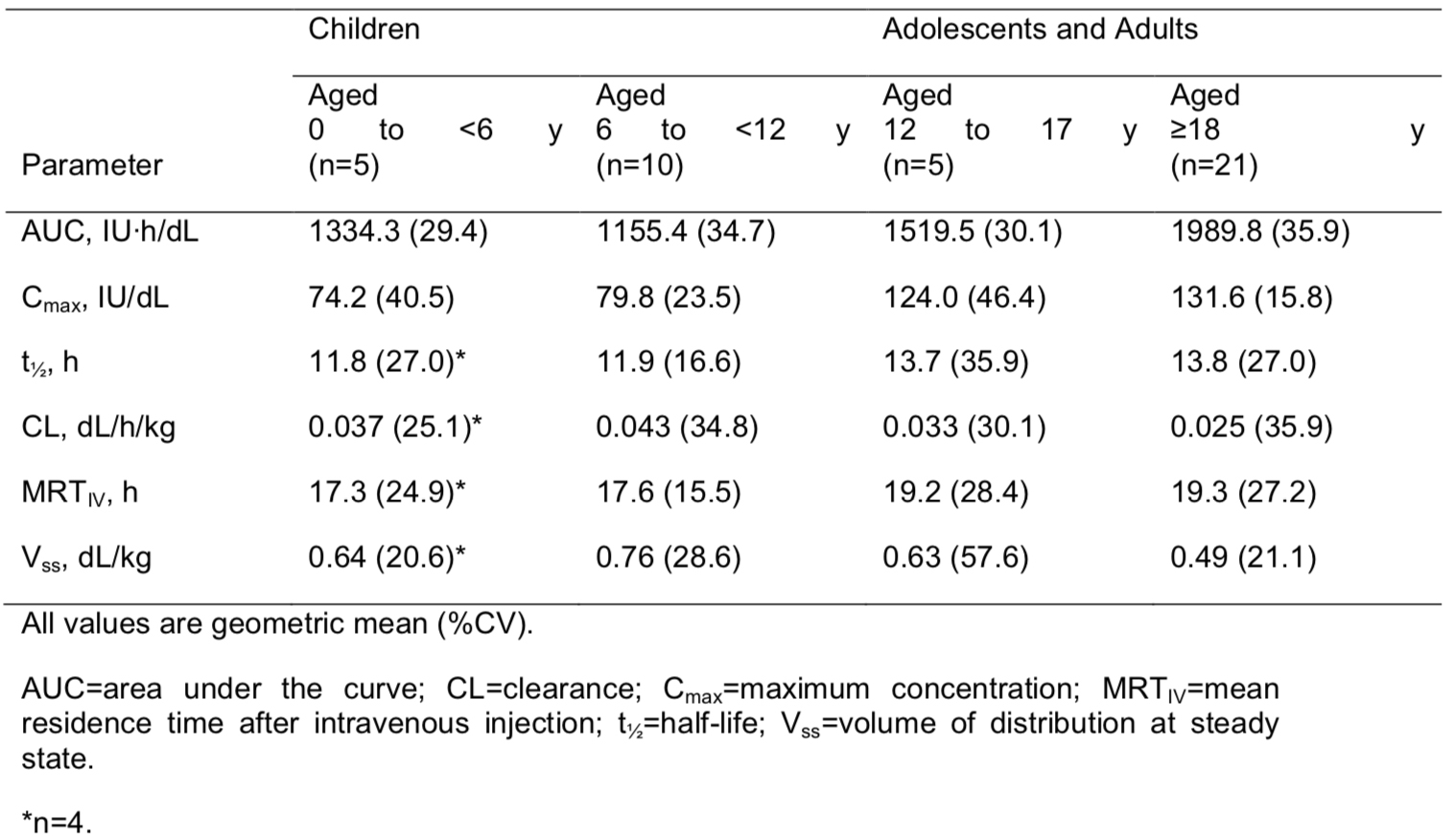
Conclusions:
Analysis of PK across the different age groups showed that the values for maximum concentration (Cmax) and area under the curve (AUC) for adolescents were within the range of those seen for adults. PK values were slightly lower in children than in adults. There were no significant differences among the ethnic groups studied.
Long-term safety and efficacy of recombinant factor IX Fc (rFIXFc) in adults and adolescents with severe hemophilia B: interim results of the B-YOND study
Objective:
The ongoing rFIXFc extension study, B-YOND (clinicaltrials.gov #NCT01425723), evaluates the long-term safety and efficacy of rFIXFc for the treatment of severe hemophilia B. Here we report interim safety and efficacy data for adults and adolescents enrolled in B- YOND.
Methods:
Upon completing B-LONG, eligible subjects could enroll in one of 4 treatment groups in B-YOND: weekly prophylaxis (20 to 100 IU/kg every 7 days), individualized prophylaxis (100 IU/kg every 8 to 16 days, or twice monthly), modified prophylaxis (a prophylaxis regimen different from weekly or individualized prophylaxis), or episodic treatment. Subjects could change treatment groups at any point in the study. The primary endpoint was development of inhibitors. Secondary outcomes included annualized bleeding rate (ABR) and rFIXFc exposure days (EDs).
Summary:
93/115 subjects (80.9%) who completed B-LONG enrolled in B-YOND. As of the interim data cut (17 October 2014), 18 subjects had completed, 7 had discontinued, and 68 remained on study (median time on study, 119.9 weeks). From the start of B-LONG to the B- YOND interim data cut, the median time on rFIXFc was 171.6 weeks and 68 subjects (73%) had ≥100 cumulative rFIXFc EDs. 29/90 subjects (32%) from Arms 1-3 of B-LONG changed treatment groups at the start of or during B-YOND, including 9/19 subjects who changed from episodic to prophylactic treatment (1 subject changed back to episodic treatment before the interim data cut). In the weekly prophylaxis group, the median (IQR) average weekly prophylactic dose was 49.5 (21.0, 105.6) IU/kg. The median (IQR) average dosing intervals in the individualized and modified prophylaxis groups were 13.7 (10.1, 14.0) days and 6.9 (4.9, 7.0) days, respectively (a reduction in annual infusions of ~75% and ~50%, respectively, compared with twice-weekly administration of a standard half-life FIX product). As of the B- YOND interim data cut, no inhibitors were observed, and there were no reports of anaphylaxis or serious hypersensitivity reactions associated with rFIXFc, and no thrombotic events. Adverse events were generally typical of the hemophilia B population; no subject discontinued due to an adverse event. Median ABRs were low with rFIXFc prophylaxis (Table). 97.2% of bleeding episodes were controlled with 1 or 2 infusions.
Conclusions:
Interim data from B- YOND confirm the long-term safety of rFIXFc and the maintenance of a low ABR with prophylactic dosing every 1 to 2 weeks.
Interim results of the B-YOND study evaluating long-term safety and efficacy of recombinant factor IX Fc (rFIXFc) in children with severe hemophilia B
Adherence influences annualized bleeding rate during prophylaxis with turoctocog alfa: results from the guardian™1 trial
Pediatric Venous Thrombosis Associated With Staphylococcal Infections: A Single Institutional Experience
Introduction:
Since the emergence of community-acquired methicillin-resistant S. aureus (MRSA), severe manifestations of infection are encountered more frequently. Venous thrombosis (VT) has been previously reported in children with S. aureus infections. This study reviews our institutional experience and outcomes of children with VT and staphylococcal infections.
Methods:
A retrospective analysis of 15 pediatric patients (≤ 18 years) treated for VT and staphylococcal infections at Children’s Memorial Hermann Hospital between January 1, 2010 and December 31, 2014 was performed.
Results:
Fifteen patients were included (10 males, 5 females) with a median age at diagnosis of 8 years (range 2 mo-17yrs). Underlying infections included: osteomyelitis (n=7), soft tissue infection (n=3), aortitis (n=1), meningitis (n=1), septic arthritis (n=1), central line infection (n=1), septicemia (n=1). Primary presentation included: swelling (n=12), pain (n=11), tenderness (n= 8), and color changes (n=3). One patient had prior history of VT. Family history was positive for VT in one patient. Isolated organisms included: MRSA (n=9), MSSA (n=3), polymicrobial (n=2), and Staphylococcus non- aureus (n=1). Eight patients had central venous catheters (CVC) and 5 of them had thrombosis at the CVC site. VT sites identified by Doppler US (DUS) included: upper extremity (n= 5), lower extremity (n= 8), and neck (n=2). All thrombophilia work up was negative. The median D-dimer at diagnosis was 3 ug/ml (range: 0.31- 6.54 ug/ml). Median time elapsed between infection and VT diagnosis was 5.5 days (range: 0-35 days). All patients received anticoagulation using LMWH (1mg/kg/dose) except one who had superficial thrombophlebitis and was managed conservatively. Median time to achieve therapeutic anti- factor Xa level was 2.5 days (range 1- 6 days). Median duration of anticoagulation was 3 months. Three out of 13 patients (23%) had resolution of thrombosis within one week of anticoagulation. Four other patients had thrombus resolution by DUS at 3-6 months. Five patients did not have DUS at 3-6 months. None of the 15 patients required hospital re- admission for bleeding or thrombotic complications.
Discussion:
Staphylococcal infections may increase the risk of VT in children. Nine out of 15 patients (60%) with VT had a documented MRSA infection, which appears to confer an even higher risk for development of VT. Over half of the patients responded favorably to anticoagulation with resolution of VT within 6 months.
Conclusions:
A high index of suspicion for VT is warranted in children with Staphyloccoccal infections (particularly MRSA) to promptly diagnose and treat. This approach may improve outcomes and minimize complications including septic emboli and post-thrombotic syndrome. Prophylactic anticoagulation in presence of MRSA infection could be considered in future studies.
Keywords: Children, staphylococcal infection, venous thrombosis
Pharmacist/Provider Collaboration needed to Optimize Dosing Regimens in Order to Reduce Bleed Rates in Hemophilia A Patients on Prophylaxis Regimens
Varying Regimens in Hemophilia A Patients Undergoing Immune Tolerance: Removing Barriers to Enhance Outcomes
Objective:
Promote awareness of and an opportunity for dialogue regarding variability among prescribed regimens to enhance care for hemophilia A patients with inhibitors undergoing immune tolerance.
Methods:
Retrospective chart review of all severe hemophilia A patients receiving interdisciplinary inhibitor management home infusion support between March of 2013 and March of 2015. Excluded mild patients developing inhibitors postoperatively and those for which insufficient titer information was provided/available from prescribers. 14 patients met the inclusion criteria. We attempted to further categorize these regimens in to low and high dosing regimens as outlined in the International Immune Tolerance Study.
Summary:
We identified 14 patients on 7 different ITI regimens, none of whom expressly followed the “low or high dose regimens” of 50 units per kg 3 times a week or 200 units per kg daily. They were on either recombinant FVIII products (11) or vWF containing products (3). The reviewed population was followed by 11 different HTC’s which included 13 different prescribers. We noted time to tolerization (when information available), bleed rate, as well as the interventions and support offered patients by the homecare inhibitor team. Although the sample was small, a notable increase in bleeds was seen in those patients on regimens below100 units/kg/day. Time to tolerization was unavailable for 3 of the 14. Of the remaining 11, time to tolerization ranged from 1-45 months and there was no significant difference seen amongst regimens.
Conclusions:
Seven different regimens for ITI were prescribed for 14 unique patients across the country. All achieved successful immune tolerance, but there was variability in the frequency of spontaneous bleeds and time to tolerance. Exact time to tolerance was limited by both the inability to obtain lab values (titers) from the prescriber or long periods between titer levels. Immune tolerance induction dosing regimens have been long debated and several studies continue to attempt to provide clarity and guidance. A missing component of the research published to date is the importance of patient adherence and the benefit of prescriber/pharmacist/payer collaboration. Economic influences further complicate this as many HTC’s perceive pharmacies as competitors, rather than collaborators in care. Additionally, payers may limit networks or implement other barriers to refills. The authors wish to work collaboratively to remove these barriers and enhance outcomes.
Swimming exercise ameliorates pain, swelling and bone quality in a blood-induced joint damage animal model
Background:
Arthropathy is one of the major complications in hemophilia, leading to functional limitations and poor quality of life. Exercise is recommended as being beneficial for people with hemophilia, yet a lack of exercise-related evidence using exercise and control groups exists in this population, compared to other joint pathologies (i.e. osteoarthritis).
Objective:
To evaluate effects of post-hemarthrosis exercise on: pain; swelling; bone density and joint damage, using an animal model.
Methods:
Twelve Wistar rats were divided into an Exercise Group (EG; n=6) and Control Group (CG; n=6). All rats received eight weekly intraarticular injections in their right knees with 0.1 mL autologous blood, and left knees with 0.1 mL saline solution. The EG performed an 8 wk swimming protocol (during the period of intraarticular injections) of 60 min (5x week) with 5% body weight attached on their tails. Pain status was assessed weekly with a static weight-bearing incapacitance test (SWIT). Joint diameters were measured post-protocol with a digital microcaliper. Bone mineral density was assessed using dual-energy X-ray absorptiometry (whole-body, tibia, femur and knee joint) pre and post-study. Following sacrifice, bilateral knee X-rays were taken and scored with Pettersson’s score. Resting heart rate and heart weight were registered post-study, as markers of physical training.
Results:
CG manifested decreased weight-bearing with SWIT, compared to EG (p<0.001). EG presented reduced joint swelling, compared to CG (p=0.034). Significant increase in bone mineral density variation (BMD∆) was observed at the tibia (p=0.016), and knee (p=0.018) of the EF compared to CG. The EG also showed a tendency of an increase in femoral BMD∆ compared to CG (p=0.09). Pettersson scores demonstrated increased bone destruction in the CG compared to EG (p=0.05).
Conclusion:
This is the first time that beneficial effects of a swimming exercise protocol on pain, swelling, bone density and radiologic changes have been shown in a blood-induced, joint damage animal model. Hopefully this data will provide clues for future study designs in this area.
Hemophilia Genotyping Results from the My Life, Our Future Project
Haemophilia A Carriers Experience Reduced Health-Related Quality of Life
Comorbidities among Adults with Hemophilia: Hemophilia Utilization Group Studies (HUGS V)
Objective:
To investigate the prevalence of comorbidities among adults with hemophilia in the Hemophilia Utilization Group Studies (HUGS)
Methods:
Standardized interviews were conducted for two prospective cohort studies HUGS- Va (hemophilia A) and HUGS-Vb (hemophilia B) at six and ten US Hemophilia Treatment Centers, respectively, between 2005 and 2011. Clinical records were reviewed. Information captured included self-reported comorbidities, sociodemographics, treatment patterns and other clinical characteristics. Overweight and obesity were defined as body mass index (BMI) 25-29 kg/m2 and BMI ≥30 kg/m2, respectively. The prevalence of comorbidities was calculated. The association of comorbidities with hemophilic severity, age and type of hemophilia were assessed using appropriate statistical methods for categorical or continuous variables.
Summary:
The analyses included a total of 213 adults (HUGS-Va: n=147, HUGS-Vb: n=66) aged 20 to 65 years (mean±standard deviation: 36.6±12.9). Approximately, 64% of hemophilia A and 44% of hemophilia B individuals had severe hemophilia. The five most prevalent self-reported comorbidities were liver disease/hepatitis (66%), overweight/obesity (60%), arthritis (51%), human immunodeficiency virus infection/acquired immunodeficiency syndrome (HIV/AIDS) (24%), and hypertension (23%). The individuals in the hemophilia A sample were more likely to report liver disease/hepatitis (71% vs. 53%) and HIV/AIDS (30% vs. 9%) than those in the hemophilia B sample (all p<0.009). The prevalence of overweight/obesity (59% vs. 60%), arthritis (55% vs. 42%), and hypertension (22% vs. 24%) were not significantly different between hemophilia A and hemophilia B samples (all p>0.05). Prevalence of comorbidities was greater among individuals with severe than mild/moderate hemophilia for most conditions: liver disease/hepatitis (79% vs. 48%), arthritis (61% vs. 38%), HIV/AIDS (37% vs. 6%), and stroke/brain haemorrhage (11% vs. 2%) (all p<0.02), the exception being overweight/obesity (52% vs. 70%, p=0.007). The individuals with hemophilia A had a significantly greater number of comorbidities than those with hemophilia B (mean±standard deviation: 2.5±1.9 vs. 1.0±1.1; p<0.0001); 85% of the hemophilia A sample reported having more than one comorbidity compared to 61% of those with hemophilia B (p<0.0001). The number of comorbidities increased significantly with advancing age (p<0.0001).
Conclusions:
As one of the largest prospective studies of persons with hemophilia, the HUGS sample is representative of the US hemophilia A and B populations. Except for overweight/obesity, the most prevalent comorbidities reported in HUGS related to their hemophilia complications, and were significantly associated with hemophilic severity. As the life expectancy of persons with hemophilia increases, the need for studies focusing on the health care needs of individuals with hemophilia and comorbid conditions will increase.
Does Quality of Life improve with successful immune tolerance induction? An illustrative case report.
A Comparison of US Prophylaxis Rates Shows Lower Prescribing for Severe Hemophilia B Patients than Severe Hemophilia A Patients
Changes in Healthcare Resource Utilization and Haemophilia Related Events in Patients Diagnosed with Haemophilia A
Objective:
To evaluate changes in healthcare resource utilization and haemophilia related events among patients diagnosed with haemophilia A between 2008 and 2012.
Methods:
This retrospective study analyzed data from the Humedica de-identified electronic medical record database between January 2008 and December 2012. Male patients diagnosed with hemophilia A (ICD-9-CM 286.0) receiving treatment with a clotting factor were eligible if they 1) were ≥18 years of age 2) did not receive Factor IX therapy and 3) did not have a diagnosis of Von Willebrand while receiving factor VIII therapy containing von Willebrand factor. All patient level resource utilization was converted to utilization per patient year. Resource utilization was then compared across time periods using repeated measures analysis of variance (ANOVA). The annualized number of haemophilia related events (haemophilic arthropathy or other joint related events) was calculated for each year. McNemar’s chi-square test was used to compare the frequencies across years.
Summary:
136 patients contributing 375 patient-years were included in this study. Office/clinic visits accounted for the majority of healthcare encounters annually; 7.5 all-cause visits per year and 2.2 haemophilia related visits per year. The number of annual all-cause office/clinic visits for Haemophilia A patients decreased significantly over time from 12.5 visits in 2008 to 5.9 visits in 2012 (p=0.0404), while haemophilia A-specific annual visits decreased from 4.0 to 1.5 (p=0.1991) during the same period. On average haemophilia A patients had less than 1 inpatient and emergency room visits per year, which did not change significantly over time (p=0.6371 and p=0.4845, respectively). Over the 5-year period, haemophilic events occurred in 30.93% of patient years, changed from 23.81% in 2009 to 34.09% in 2011 (p=0.6658).
Conclusions:
Office/clinic outpatient visits among patients diagnosed with haemophilia A has decreased overtime. However, the rate of haemophilia related arthropathies and other associated events have remained high. Further analysis is needed to understand how to best manage patients diagnosed with haemophilia A and reduce the proportion of patients who develop reduced joint mobility due to bleeding into joints.
Addressing patient concerns about product switching in hemophilia A: Evaluating turoctocog alfa data from the guardian™ clinical trial program
Treatment, Outcomes, and Access to Care Among Young Adults (ages 18-30) With Hemophilia in the US Hemophilia Experiences, Results and Opportunities (HERO) Study
Hemophilia impacts relationships and employment of young adults (ages 18-30) in the us hemophilia experiences, results and opportunities (hero) study
Objective:
To assess impact of hemophilia on relationships and employment during the transition to adulthood in young adult (YA) patients with hemophilia (PWH).
Methods:
Analysis of US YA-PWH respondents (aged 18-30 years) in the HERO study.
Summary:
Of 189 US adult PWH HERO respondents, 66 were aged 18-30 years (median: 26). Most lived in large cities (52%) or suburban areas (29%); 73% reported household income of <$60,000/year. Some (32%) were married or in long-term relationships; 20% lived alone. Negative impact on relationships was reported by 32%; 62% predicted an impact in the future, 62% cited difficulty understanding issues, and 52% worry about supporting a family. Only 9% had children; 77% wanted to have children. Only 45% received genetic counseling from the HTC; of those, 60% felt it was helpful. Most were very/quite satisfied with support of partners/spouses (95%), family (92%), and friends (86%). Negative reactions telling friends were reported by 41%; 59% reported most/all of their friends knew about their hemophilia. Most (78%) were employed with 57% reporting office work. One fifth (20%) reported being disabled, and 14% received disability benefits. The majority (74%) reported negative impact on employment; 39% reported moderate/very large impact. Many (43%) reported current treatment allows them to work in most situations, 37% selected a job based on their needs, 29% were helped to obtain a job, 24% had to leave a job, 20% were not hired, and 18% lost a job due to hemophilia. Only 36% received advice from the HTC on work/employment, mostly on what to do if a bleed occurs at work (71%), suitable jobs (67%), when (63%) and what (33%) to tell an employer, and workplace precautions (46%). Fifty-eight percent found the advice helpful. Only 37% reported most/all of their colleagues know about their hemophilia; 38% reported a select few or 1-2 and 26% none. Most were very/quite satisfied with the support of colleagues at work/school (82%). Most YA-PWH (62%) were members of an organization or online (48%) or other (35%) support group.
Conclusions:
During the transition to independent adults, YA-PWH are likely planning for family and careers. Many reported negative impacts on relationships and employment, highlighting a need for career counseling. YA-PWH are supported by friends and colleagues, but this may be a limited group. HTCs are an underutilized resource for addressing these issues, with perhaps online peer-support networks playing a larger role during this transition to adulthood and independence.
Pain and Arthropathy Impact Quality of Life of Young Adults With Hemophilia (ages 18-30) in the United States: Observations From the Hemophilia Experiences, Results and Opportunities (HERO) Study
Real World Utilization and Cost of aPCC versus rFVIIa Among Hemophilia Patients with Inhibitors in the US
Ongoing Prospective ADVATE Immune Tolerance Induction Registry (PAIR) Continues to Demonstrate Success Rates Consistent with Published Literature
Objectives:
PAIR is an ongoing, global, non-interventional, post-authorization safety surveillance designed to collect information on ADVATE safety and effectiveness in immune tolerance induction (ITI) therapy in routine practice.
Methods:
From July, 2007 to April, 2011, individuals with hemophilia A and inhibitors were enrolled in 10 countries. The primary objective is to assess the incidence of adverse events (AEs) related to ADVATE during ITI therapy. Secondary objectives are incidence of central venous access device (CVAD)-related complications, and success rates of ITI therapy. Maximum observation period for ITI is 33 months plus a 12 month follow-up.
Summary:
As of April 1, 2014, 36 of 44 subjects (81.8%) completed ITI therapy, 28 (63.6%) of which completed the 12 month follow-up. Six subjects withdrew prior to completing ITI therapy. Dosing regimens were: ≥200 IU/kg/day (n=4, 9.1%); 131-199 IU/kg/day (n=3, 6.8%); 90-130 IU/kg/day (n=26, 59.1%) and <90 IU/kg/day (n= 11, 25.0%). During the observation period, 337 bleeding episodes and 273 AEs were reported for all enrolled subjects (N=44). Of these AEs, 52 (19.0%) were serious and none were considered related, while 15 (5.5%) were non-serious and related. CVAD complications were common; 32 subjects experienced one or more CVAD-related AE such as hospitalization, line infection, line malfunction, line removal, and pain following port-a-cath bleed. Of the subjects that completed ITI, 21 achieved negative titer levels, two experienced a high to low titer conversion, seven failed to achieve negative titer, and six were un-assessable per protocol. After 18 months therapy, Kaplan Meier estimates of success for achievement of first negative titer was 65.4% (asymptotic 95% CI: 48.7-81.5%, n=36) for the completer group. Rates were higher for the per protocol analysis set (72.2%, CI: 54.6-87.5%, n=30), and slightly lower for the full analysis set (63.5%, CI: 48.0- 78.7%, n=44).
Conclusions:
These interim outcome results are consistent with previous reports from PAIR and other published data on ADVATE in ITI. No new ADVATE related safety issues have been seen. The last two participating subjects will end observation within the next year.
The use of combination therapy with plasma derived and recombinant factor VIII in patients with hemophilia A: a single institution experience
New insights from modeling FVIII Kinetics of Native vs. Extended Half-life FVIII Products - Comparing FVIII Coverage under Various Dosing Scenarios
Introduction:
Extended half-life factor VIII (FVIII) products currently in development require a careful evaluation of dosing options given that the pharmacokinetics of weekly FVIII levels , including FVIII peak and trough levels, have been shown to play a role in bleeding occurrence.
Methods:
Pharmacokinetic (PK) modeling was conducted using published literature and clinical trial data. Published population PK models for recombinant full length DNA, plasma and albumin free FVIII (octocog alfa, rAHF-PFM) and recombinant B-domain-deleted FVIII Fc fusion product (efraloctocog alfa, rFVIIIFc) were used. All calculations were performed assuming linear pharmacokinetics. Dosing frequencies assessed were every 2 days with rAHF-PFM and every 3, 4, 5 or 7 days with rFVIIIFc. Dosages assessed were 30 IU/kg of rAHF-PFM and 30, 40, 50 or 65 IU/kg of rFVIIIFc. Results were generated for a hypothetical patient aged 30 years, with 70 kg body weight, exhibiting a VWF level of 118 IU/dL and a hematocrit of 45%. Time spent below 1% and 3% per week and time spent above 10% and 20% per week were assessed and compared for each scenario.
Summary:
Compared to 30 IU/kg of rAHF-PFM dosed every other day, a patient on rFVIIIFc would spend more time per week below 1% FVIII level when dosed every 5th day up to 50 IU/kg and when dosed every 7th day at any dose within the range tested. Moreover, even when increasing the dosing frequency to 30 IU/kg of rFVIIIFc every 3rd day, or when dosing rFVIIIFc up to 65 IU/Kg every 4th, 5th and 7th day the patient’s plasma FVIII level will drop below 3%. When looking at time per week spent above higher FVIII levels, the model patient would spend more time above 10% when dosed with rAHF-PFM at 30 IU/kg every second day as compared to rFVIIIFc dosed every 3 days at 30IU/kg, and every 4, 5 and 7 days at any dose within the range tested (≤65 IU/kg). Likewise, this patient will also spend more time above 20% with rAHF-PFM every other day than with rFVIIIFc dosed every 4 days at 40 IU/kg and every 5 days at 50 IU/kg and every 7 days at 65 IU/Kg.
Conclusion:
These data indicate that choice and optimal dosing of FVIII products require a better understanding of individual pharmacokinetics in order to avoid that patients would spend extended time at levels insufficient to protect them from bleeding, particularly subjects with a more active lifestyle.
Using Oral History for Patient Education: The Gift of Experience II: Conversations with Parents about Hemophilia
The expression of codon-optimized blood coagulation factor VIII using a lentiviral system
The Impact of Reduced Treatment Frequency: Qualitative Evaluation of Adherence and Outcomes in Chronic Disease Management
Bleeding Risk for the Active Person with Hemophilia: A Comparison of Factor VIII Treatment Regimens
Background:
Bleeding risk during physical activities for persons with hemophilia has been shown to be reduced with adequate factor levels (Broderick 2012). With extended half-life Factor VIII and IX products offering opportunities for longer intervals between infusions, it is important to understand the bleeding risk for patients who have an active lifestyle.
Objective:
To evaluate the relative risk of bleeding between prophylaxis schedules using recombinant Factor VIII (rFVIII) vs. rFVIII-Fc among different patient physical activity profiles
Methods:
A mathematical model based on the literature was developed. Factor levels were estimated using a one-compartment pharmacokinetic model (Collins 2010). Half-life and incremental recovery values were taken from a crossover study of rFVIII and rFVIII-Fc (Mahlangu 2014). Five prophylaxis regimens were evaluated: two common to rFVIII (30IU/kg every other day (EOD); 35IU/kg 3x/week) and three studied in the rFVIII-Fc pivotal trial (2x/ week: 25IU/kg covering 3 days and 50IU/kg covering 4 days per week; 50IU/kg every 5 days; 65IU/kg 1x/week). Activities such as swimming, running and wrestling were classified as Type 1, Type 2, and Type 3 in Broderick 2012, derived from the NHF “Playing It Safe Brochure” (Anderson 2005). Risk of bleeding by activity category and factor level at time of activity was calculated using the odds ratio values from Broderick 2012. Three hypothetical patient activity profiles were evaluated: Consistently Active (M-Sun: Type 2 activities), Regular Exerciser (M-F: Type 2 activities, Sat-Sun: Type 1 activities), and Weekend Warrior (TThSun: Type 1 activities, MWF: Type 2 activities, Sat: Type 3 activities). For each regimen, the infusion schedule with the lowest bleeding risk for each patient activity profile was selected. The relative bleeding risk vs. the best prophylaxis regimen was estimated and compared for the activity profiles.
Results:
rFVIII 30IU/kg EOD and 35IU/kg 3x/week achieved the two lowest bleeding risk for all three patient activity profiles. Compared to rFVIII every other day, bleeding risk was increased by 20%, 25% and 46% for the Consistently Active patient prescribed rFVIII-Fc twice per week, every 5 days, and 1x/week, respectively. Compared to rFVIII 3x/week, bleeding risk was increased by 20%, 30% and 44% for the Regular Exerciser and by 21%, 32% and 45% for the Weekend Warrior prescribed rFVIII-Fc 2x/week, every 5 days, and 1x/ week, respectively.
Conclusion:
This model suggests that active patients characterized with the above profiles may have reduced bleeding risk with rFVIII compared to extended half-life FVIII dosing regimens evaluated in this analysis.
Safety and Effectiveness of Anti Inhibitor Coagulation Complex (AICC) in Routine Clinical Management: a Post-Authorization Safety Study (PASS)
Gingival Bleeding and Oral Hygiene in Women with von Willebrand Disease
Objectives:
To determine the relationship between von Willebrand disease (vWD), dental plaque, and gingival bleeding in women with vWD and to determine the oral hygiene habits and dental care utilization in women with vWD.
Methods:
Consenting adult women with vWD (n=40) will have been recruited for this study. A questionnaire was given with 34 items covering topics such as dental care utilization, oral health quality of life, and oral hygiene habits. A brief oral examination was performed on each subject to assess which surfaces of the six Ramfjord teeth presented with dental plaque, and which surfaces bled upon flossing. Information was also gathered about each subject’s medical history, including the type of vWD, severity, and last von Willebrand factor levels.
Summary:
Data is still being collected for this study. Data collection will be completed on June 30. The data gathered so far shows that the majority of women who participated in this study have a high plaque score, yet minimal bleeding with flossing, when a gentle c-wrap flossing technique was performed.
Conclusions:
Results of this study are expected to show that the bleeding disorder has minimal effect on the amount of gingival bleeding that occurs with a c-wrap flossing technique. It’s possible that conclusions may be made that correct flossing technique can be performed in a manner that does not, in itself, cause gingival bleeding. This can perhaps assist in increasing the amount of people with bleeding disorders that floss, diminishing the fear that many people with bleeding disorders have of causing excessive bleeding with flossing.
Study Design of a Phase 3, Open-Label Trial of the Safety and Efficacy of Recombinant Factor IX Fc Fusion Protein for the Prevention and Treatment of Bleeding Episodes in Previously Untreated Patients With Severe Hemophilia B
Study Design of a Phase 3, Open-Label Trial of the Safety and Efficacy of Recombinant Factor VIII Fc Fusion Protein for the Prevention and Treatment of Bleeding Episodes in Previously Untreated Patients With Severe Hemophilia A
A Study Evaluating the Impact of myCubixx, an Innovative Factor Inventory Management and Storage System with Selected Outcomes on People with Hemophilia A
Safety of BAX 855, a Polyethylene Glycol (PEG) Conjugated Full-Length Recombinant Factor VIII Product
Objectives:
Biopharmaceuticals are an emerging branch of therapeutic agents. Their short half-life, rapid elimination and ability to induce a specific immune response, however, may impair their applicability. These disadvantages have been overcome by chemical modification with polyethylene glycol (PEG), which has enhanced the PK and safety of several marketed proteins since the 1990s. PEGylation uses metabolically stable PEG polymers, often with a molecular size of 5-60 kDa.
PEGylated rFVIII candidates include PEG-protein-conjugates with a minimal amount of PEG attached to the protein. Baxter and Nektar are developing BAX 855, a PEGylated full-length recombinant (r) FVIII based on the FVIII molecule used for Baxter’s licensed rFVIII (ADVATE). Due to the high potency of FVIII, the absolute amount of conjugated PEG applied with PEG-FVIII is within the range of μg per kg body weight and week. PEGylation was optimized to retain functionality of the FVIII molecule and improve its pharmacokinetic properties.
PEGs ≤20 kDa are rapidly cleared mainly via the kidneys and excreted into urine. Over time, the protein portion of the PEG-FVIII conjugate is degraded by proteolysis leaving a PEG portion which is rapidly eliminated.
Methods:
Preclinical safety, toxicokinetics and formation of anti-product antibodies were assessed in rats dosed intravenously at 350 or 700U/kg BAX 855 every other day, and in macaques receiving 150, 350 or 700U/kg BAX 855 every five days, for 28 days.
Like other non- degradable entities, physiological clearance mechanisms of PEG may include liver macrophage uptake. Clearance by macrophages in mammals has been reported to cause vacuolization at high cumulative doses. Generally, vacuoles were shown to consistently resolve over time, with no cellular damage, inflammation at the vacuolization site or functional deficits of affected tissues, and are therefore regarded to not affect the safety of PEGylated therapeutics.
Summary:
No systemic adverse effects or vacuolizations were observed after 28-day intravenous administration with BAX 855. Therefore, 700 U/kg was considered the no observed adverse effect level in these studies.
Conclusions:
This favorable safety profile provides the basis for proceeding with human trials.
Extended-interval Dosing with rFIXFc Is Associated With Low Bleeding Rates and a Reduction in Weekly Factor Use
A Prospective Case-Control Study of Bleeding Phenotype in Hemophilia A Carriers
Dosing Routines and Bleeding Rates Before and Following Treatment with rFVIIIFc in the A-LONG Clinical Study
3-Year Results From SPINART: Prolonged Reduction of Bleeding With Prophylaxis Using Bayer’s Sucrose-Formulated Recombinant Factor VIII
Production and Characterization of BAX 855, PEGylated rFVIII with Extended Half-Life
Objective:
Baxter has developed BAX 855, a PEGylated form of Baxter’s recombinant full length FVIII (rFVIII) product based on the ADVATETM manufacturing process. Here we describe it ́s manufacturing and structural and functional characterization.
Methods:
A rFVIII intermediate of the ADVATETM manufacturing process is the starting material for the conjugation process for preparing BAX 855 by proprietary PEGylation technology from Nektar Therapeutics. Similar technology has been successfully employed for marketed and licensed PEGylated drug products and drugs in clinical use. The manufacturing process for BAX 855 comprises several steps, including controlled PEGylation followed by cation exchange chromatography. Final formulation uses the same excipients as ADVATETM. BAX 855 was characterized by a number of analytical methods, focusing on the elucidation of the primary structure, posttranslational modifications, PEGylation site distribution and three- dimensional structure. The overall hemostatic potency of BAX 855 in FVIII-deficient plasma was assessed by conventional FVIII 1-stage clotting and chromogenic assays and with a thrombin generation assay. The tenase cofactor activity of FVIII was determined by measuring the kinetics of FXa generation. Binding of BAX 855 in comparison to ADVATETM was determined to its ligands VWF and low-density lipoprotein-receptor-related protein (LRP).
Summary and Conclusions:
BAX 855 is a full-length rFVIII with extended half-life. PK studies in different animal species and humans with hemophilia A display longer survival of BAX 855 compared to ADVATETM. The product is based on the original, native FVIII protein utilized in the production of ADVATETM. Our analyses show that BAX 855 can be manufactured reproducibly without changes to the protein structure characteristic for a fully functional full-length rFVIII molecule. The process is suited to manufacture BAX 855 in large scale and showed a good batch to batch consistency, ensuring an equivalent product quality for each batch. BAX 855 has a specific activity similar to that of rFVIII in ADVATETM and PEGylation degrees in the range of 2 to 3 mol PEG / mol rFVIII. SDS-PAGE and Western blot analysis of BAX 855 confirm PEGylation and demonstrate an increase in the molecular weight of the various FVIII domains.
In comparison to ADVATETM the functional properties of BAX 855 were fully retained except for binding to LRP, indicating that PEGylation did not have an impact on the functional properties of rFVIII. The latter might explain why BAX 855 shows prolonged survival in the circulation of hemophilic species and patients with hemophilia A than ADVATETM.
SPINART Trial 3-Year Results With Bayer’s Sucrose-Formulated Recombinant Factor VIII: Improved Joint Function and Health-Related Quality of Life in Adults Using Prophylaxis
Joint Outcomes by Magnetic Resonance Imaging After Treatment With Bayer’s Sucrose-Formulated Recombinant Factor VIII in the SPINART Study: Results at the 3-year Evaluation Timepoint
Dosing Flexibility in Prophylaxis Regimens With Bayer’s Sucrose-Formulated Recombinant Factor VIII: Experience From Postmarketing Surveillance Studies
Objectives:
Factor VIII (FVIII) prophylaxis regimens for severe hemophilia A that allow more flexible dosing than the standard 3-times-weekly regimen while maintaining efficacy may improve adherence. This analysis compared the clinical efficacy of once- or twice-weekly versus ≥3-times-weekly prophylaxis dosing of Bayer’s sucrose-formulated recombinant FVIII (rFVIII-FS) in patients with severe hemophilia A.
Methods:
Data from 3 postmarketing studies were pooled. Patients with severe hemophilia A and no history of inhibitors who were receiving ≥1 prophylaxis infusion/wk of rFVIII-FS for ≥80% of a prophylaxis observation period (≥5 months) were included. Patients were categorized based on age (<18 and ≥18 years) and physician-assigned treatment regimens of 1–2 prophylaxis injections/wk (n=63) or ≥3 prophylaxis injections/wk (n=76). Descriptive statistics were determined for annualized bleeding rates (ABRs) by dosing group and age subgroups.
Summary:
Median (quartile 1; quartile 3) ABR for all bleeds was 2.0 (0; 4.0) in the group receiving 1–2 prophylaxis injections/wk and 3.9 (1.5; 9.3) in the group with ≥3 prophylaxis injections/wk. Similarly, median ABRs for joint, spontaneous, and trauma-related bleeds were numerically lower in the group receiving 1–2 prophylaxis injections/wk. The trend toward lower ABRs in the group with 1–2 prophylaxis injections/wk was observed in both age subgroups, although ABRs were somewhat higher in patients ≥18 vs <18 years. Zero annualized bleeds were reported by 30% and 7% of patients in the groups with 1–2 prophylaxis injections/wk and ≥3 prophylaxis injections/wk, respectively.
Conclusions:
These data demonstrate that bleeding control can be achieved in some patients with severe hemophilia A using a <3-times-weekly prophylaxis dosing regimen and that physicians’ judgment based on bleeding phenotype can successfully direct the frequency of prophylactic dosing.
Retrospective Database Analysis of the Prevalence of Cardiovascular Comorbidities in a US Patient Population with Hemophilia A: Confirmation of Findings
Global Assessment of Knowledge and Practices in the Diagnosis, Classification, and Management of Hemophilia among Pediatric Providers
Real-world Dosing Patterns of Factor in Hemophilia B Patients
Objective:
To analyse real-world FIX dosing and treatment interval patterns. A secondary objective was to compare the observed dosing patterns with the dosing regimens for FIX products evaluated in clinical studies.
Methods:
A retrospective analysis was conducted using aggregate Specialty Pharmacy Provider (SPP) records from 2012 through Q12014. SPP data included 63 different attributes for each prescription, including trade name, NDC, quantity shipped, prescribed infusion dose, days supplied, and dose frequency. Patients were considered eligible for the analysis if they received a shipment of any FIX product. Patients were excluded from the analysis if they were being treated episodically, for immune tolerance induction, or their pharmacy records did not specify a prescribed infusion dose. Patients with missing or extremely abnormal weights were also excluded. The patient’s weekly consumption was calculated for each shipment record by multiplying the prescribed infusion dose by the dose frequency and dividing the product by the patient’s weight, resulting in the patient’s average weekly prescribed dose (IU/kg/week). Patients were also categorized according to dosing interval.
Summary:
The analysis included 118 hemophilia B patients with a median age of 20 (range: 2-63) and median weight of 55.4 kg (range: 9.5-129 kg). Pharmacy dispensing records represented 78 distinct prescribers across 29 states. FIX therapies evaluated included Benefix®, Alphanine®, Mononine®, and Rixubis®. The average weekly consumption across all therapies was 139.0 IU/kg/week (95% CI, 128.7-150.3). Dosing frequency ranged from every other day to once weekly. Twice weekly was the most common dosing interval, representing 56.8% of patient records. According to clinical trial data and FDA labelled dosing for FIX therapies, lower weekly consumption may be expected. For BeneFIX the mean weekly consumption was 80.6 IU/kg, 40.3 IU/kg administered twice-weekly. For Rixubis the mean weekly consumption was 88.9 IU/kg, 49.4 IU/kg administered 1.8 times/week and a US dosing recommendation of 40-60 IU/kg dosed twice-weekly. Two prophylactic regimens have been evaluated for AlprolixTM. In the last 3 months of B-LONG, in the weekly prophylaxis arm, the overall median dose on study was 40.5 IU/kg. The individualized interval prophylaxis arm had a median weekly dose of 50.0 IU/kg, 100 IU/kg administered every 14 days. No real world dosing is available for Alprolix due to its recent approval.
Conclusions:
Dosing regimens evaluated in the real world for conventional FIX products indicate greater consumption than reported in clinical trials. This may result in unpredictability for payers who are responsible for healthcare budgets.
Real-world dosing of factor in hemophilia A patients
Mobilizing Patients Towards Positive Health Management Through Motivational Interviewing
Identification of Previously Unreported F8 and F9 Gene Mutations in Hemophilia Subjects From the Phase 3 Clinical Trials of rFVIIIFc and rFIXFc
Objective:
Hemophilia A and B are X-linked bleeding disorders caused by the deficiency of clotting factor VIII or IX, respectively. Mutations in the F8 gene can result in hemophilia A while mutations in the F9 gene can lead to hemophilia B. The objective of this analysis was to evaluate the F8 and F9 genotypes of subjects screened for enrollment in the phase III clinical trials of rFVIIIFc in hemophilia A (A-LONG) and of rFIXFc in hemophilia B (B-LONG).
Methods:
The F8 and F9 genotypes of 170 subjects with severe hemophilia A and 114 subjects with severe hemophilia B, respectively, were compared with several genotype databases (HAMSTeRS, [Hemophilia A Mutation, Structure, Test and Resource Site], CHAMP [CDC Hemophilia A Mutation Project], and King’s College London Hemophilia B database), as well as with the NCBI human F8 and F9 sequences.
Summary:
Among 170 subjects with hemophilia A, inversions in intron 22 (Int22inv) were identified in 36%, nucleotide substitutions in 39%, frameshift mutations in 21%, Int1inv in 3%, and an in-frame duplication in 1 subject. Previously unreported mutations (frameshift, missense, nonsense, and splice site changes) were found in 24 subjects, with 2 unrelated subjects having the same mutation, resulting in 23 novel mutations being identified. Among 114 subjects with hemophilia B, the majority (86%) had some form of substitution mutation (missense, nonsense, splice-site change), consistent with previous reports. Thirteen previously unreported mutations were identified, including 10 substitutions (7 missense, 2 nonsense and 1 splice-site change), 1 deletion, and 2 insertions.
Conclusions:
In this analysis, 23 previously unreported mutations in the F8 gene of subjects with severe hemophilia A and 13 in the F9 gene of subjects with severe hemophilia B were identified. Identifying mutations allows for prenatal diagnosis and identification of carrier status. These results will lead to further enhancement of databases for hemophilia A and B mutations and may assist in clarifying the relationship between genotype, phenotype, and pathophysiology in individuals with hemophilia.
The effects of FXIa on clot formation and lysis in the thrombin generation assay
An Initiative to Implement Quality Improvement Measures for Hemophilia Treatment Centers
Safety and efficacy of a recombinant factor IX (BAX326*) in pediatric previously-treated patients with hemophilia B
Objectives:
This prospective clinical trial was conducted to assess the safety, hemostatic efficacy and pharmacokinetic (PK) profile of a recently developed recombinant factor IX (BAX326*) in pediatric previously-treated patients (PTPs) with severe or moderately severe hemophilia B.
Methods:
PTPs aged <12 years with severe (FIX level < 1%) or moderately severe (FIX level ≤ 2%) hemophilia B were eligible for enrollment. BAX326 was administered as prophylaxis twice a week over 6-months, and on demand for treatment of bleeds. Efficacy was evaluated by treatment response rating (excellent, good, fair, none) and annualized bleeding rate (ABR). PK assessments after one 75 ± 5 IU/kg infusion of BAX326 were assessed using a non-linear mixed model (population PK) approach. IR was measured as part of the PK evaluation 30 minutes after the initial PK infusion and at 5, 13 and 26 weeks after the initial infusion.
Summary:
Nine subjects (39.1%) had no bleeds during the study. A total of 26 bleeds occurred (mean ABR 2.7 ±3.14, median 2.0), of which 2 were spontaneous. Fewer bleeds occurred in joints than in non-joint sites (19 non joint vs. 7 joint bleeds). Hemostatic efficacy was excellent or good in >96% of bleeds, and the majority (88.5%) resolved after 1-2 infusions. The median IR (IU/dl)/(IU/kg) at the initial PK assessment was 0.685 (range: 0.31- 1.00). As expected, a higher IR was observed in association with increased patient age; IR was slightly lower in subjects < 6 years (median 0.591; range: 0.31-0.75), than in subjects aged 6 to <12 years (median 0.714; range: 0.51-1.00). IR was consistent over time. There were no adverse reactions, no thrombotic events and no hypersensitivity reactions. None of the subjects treated (N=23) developed inhibitory or specific binding antibodies against FIX.
Conclusions:
BAX326 is efficacious and safe as prophylactic treatment as well as for bleed control in pediatric hemophilia B patients.
*Licensed in the USA and Australia (Rixubis®; Baxter Healthcare Corp., USA).
Point-of-care musculoskeletal ultrasound is critical for the diagnosis of hemarthroses and soft tissue inflammation in adult patients with painful hemophilic arthropathy
Systematic review of clinical trials results assessing health-related quality of life in hemophilia patients receiving prophylaxis
Introduction and Objective:
Prospective clinical trials have demonstrated the efficacy of prophylaxis in reducing bleeding episodes in hemophilia A and B patients and those with inhibitors. However, data, predominantly from observational studies, have suggested more equivocal effects on health-related quality of life (HRQoL) [Buchbinder 2013]. The present review examined the impact of prophylaxis on HRQoL as measured during prospective trials.
Methods:
We conducted a systematic literature review of prospective studies evaluating the efficacy of prophylaxis in hemophilia using factor VIII, factor IX, or bypassing agents. Applying the inclusion criteria, we selected studies which evaluated HRQoL via validated instruments and summarized key data.
Results:
A total of 12 studies (hemophilia A [n=7]; hemophilia B [n=2]; inhibitors [n=3]) met all inclusion criteria and were reviewed. Of these studies, the investigational products were Advate (n=2), Kogenate (n=2), NovoEight (n=2), Eloctate (n=1), Rixubis (n=1), Aprolix (n=1), Feiba (n=2), and NovoSeven (n=1). HRQoL was assessed using one or a combination of the following instruments: SF-36 (n=3), EQ-5D (n=5), Haemo-QoL (n=2), Haem-A-QoL (n=3), Haemo-QOL-A (n=2) and general pain VAS (n=1). Seven of the 12 studies reported significant improvement in ≥1 HRQoL measure following prophylaxis. Advate, Rixubis and Feiba prophylaxis (among good responders with ≥ 50% bleed reduction) demonstrated statistically significant and clinically meaningful improvement in the physical component and certain domain(s) scores of the SF-36 (Valentino 2012; Windyga 2013; Gringeri 2013). Additionally, prophylaxis with Feiba showed clinically meaningful and/or statistically significant improvements in HRQoL (EQ-5D, Haemo-A-QoL), general health status (EQ-VAS) and general pain scores (VAS) (Antunes 2014; Stasyshyn 2014). Although, a previous Kogenate study indicated non-significant change in HRQoL measures (Collins 2003), recently published results from the SPINART trial demonstrated statistically significant and clinically meaningful improvement in several domains of the Haemo-QoL-A (Hong 2014). Prophylaxis with Eloctate and Alprolix resulted in non-significant change in the HRQoL measures used in their respective trials (Wyrwich 2013). Statistical and clinical significance were not reported for prophylaxis treatment with NovoEight (Santagostino 2014). Prophylaxis with NovoSeven showed a non-significant trend towards improvement in all dimensions of the EQ-5D but statistical improvement in general health status (EQ-VAS) (Hoots 2008).
Conclusion:
Results from Advate, Kogenate, Rixubis and Feiba trials offer robust evidence of clinically and statistically significant improvement in HRQoL in hemophilia patients treated with prophylaxis.
Modelling the transfer of rFVIIa procoagulant signal from tissue factor to platelets
Hemophilia Inhibitor PUP Study (HIPS)
The Medical Home Neighbor: The Intermountain Hemophilia and Thrombosis Center’s Experience with Quality Improvement via the Children’s Health Improvement Collaborative Medical Home Demonstration Project in Utah
Objective:
Patient-centered medical home (PCMH), a team-based model of practice involving patients, families, providers and care team members, focuses on high quality, efficient, and patient-centered care. The purpose of this project was to implement appropriate elements of the PCMH model in the care delivered by the Intermountain Hemophilia and Thrombosis Center (IHTC).
Methods:
The IHTC, as a medical home neighbor, participated in a 3 1⁄2 year (5/2011- 11/2014) Children’s Health Improvement Collaborative Medical Home Demonstration (MHD) that involved 3 specialty and 9 primary care practices in Utah. IHTC focused on both MHD- wide and practice-specific quality improvement (QI) goals. Our QI team included the IHTC core multidisciplinary team and a parent partner and medical home coordinator (MHC) who were funded by the MHD. The MHD led four sequential, 8-12 month projects: “Improving Collaboration Among Pediatric Generalists and Specialists,” “Implementing Care and Self- Care Plans for Children with Chronic Conditions,” “Improving Healthcare Transitions for Children with Special Health Care Needs,” and “Sustainability.” Practice-specific projects targeted goals established via a needs assessment and parent partner input. Plan-Do-Study- Act (PDSA) cycles were facilitated by the MHC and a practice coach from the MHD. Continuing education and peer support were provided via learning sessions, webinars, and ongoing mentorship.
Summary:
IHTC met all MHD-wide and practice-specific goals. Selected MHD-wide improvements included: completion of the patient history prior to new consultation (improved from ~15% to 95%); patient self-care plans (0% to 97%); and youth with an up-to-date transition tool (0% to 100%). To address “sustainability,” IHTC will continue using QI, implementing the PCMH model, and will maintain the MHC as a member of the care team. Practice-specific strategies resulted in improved efficiency and family-centered approach to the annual comprehensive clinic visit (31⁄2 hour visit decreased to 2 hours with reduced redundancy), reduced no-show/cancellations (~33% to 10%), established means for continuous individual patient/family feedback, and a formalized IHTC-specific emergency preparedness plan (currently in progress).
Conclusions:
Via participation in the MHD, the IHTC learned that QI is both realistic and rewarding. Essential components for ongoing improvement include: specific, defined and measurable goals; a QI leader; parent/consumer input; and participation by all clinical team members. The PCMH model provides a framework for meaningful change for patients, families, and clinical practice.
Acknowledgments:
Funded in part by a CHIPRA Quality Demonstrations grant: CFDA 93.767 from the U.S. Department of Health and Human Services, Centers for Medicare & Medicaid Services.
Depression in children with severe Hemophilia - a pilot study
Children with severe Hemophilia (CWH) suffer pain and inconvenience due to the required IV factor concentrates' injections and mostly exhibit poor quality of life. These children don't have any hope that their life will improve in the future. Most of them keep the Hemophilia a secret; therefore they are unable to get any emotional support from their peer group. These parameters are known stressors and triggers leading to depression, especially among children and adolescents (due to their lack of mature psychological defense mechanisms). The consequences of depression might be hazardous, since such children may neglect their medical treatment, leading to further deterioration of their medical state.
Objective:
We compared the depression level of children with Hemophilia to healthy children of the same age and background.
Methods:
Depression was evaluated using a standard validated questionnaire of depression that was developed by A.Beck - the CDI. We compared 20 children with severe Hemophilia with 25 non-Hemophiliac children.
Summary:
The calculated score for degree of depression was 7.6 for CWH vs age matched controls with a score of 12.32. The mean of normal populations is around 9. Parametrical T test for Equality of Means = 0.013.
This is the first reported study objectively addressing the issue of depression in CWH.
Conclusions:
We found the opposite of what we had expected: The children with Hemophilia were rated significantly much less depressed then the children without Hemophilia. This finding merits further validation in future larger studies and
must be examined very carefully, due to the complexity of the psychological defense mechanisms.
Kids A-LONG: Safety, Efficacy, and Pharmacokinetics of Long-Acting Recombinant Factor VIII Fc Fusion Protein (rFVIIIFc) in Previously Treated Children with Hemophilia A
Home infusion/specialty pharmacy inhibitor management program leads to patient/provider collaboration to facilitate enhanced program and patient outcomes
Objective:
To evaluate the impact of interventions in a focused home infusion/specialty pharmacy based inhibitor management program on patient outcomes including, adherence, retention, prescriber communication.
Methods:
From our bleeding disorders patients’ databases between April 1, 2013 and March 31, 2014 (12 month period), prescriptions and assessment records were analyzed for patients with hemophilia A or B with inhibitors and those with acquired inhibitors. 49 unique patient records were reviewed, care managers were interviewed, and interventions were highlighted.
Results:
44 patients had Hemophilia A with an inhibitor, 2 had an inhibitor to Hemophilia B and 3 had acquired hemophilia. Hemophilia A patients had a diagnosis of severe hemophilia A (39), Moderate (2), Mild (2). 1 Hemophilia B patient was severe and the other moderate. Total number of active inhibitor patients on service (with a detectable Bethesda Unit titre) increased from 22 to 30 over this same period. 8 patients left service during the year due to insurance changes (4), and transfer to HTC’s 340B program (4). At the beginning of the study period, a multidisciplinary inhibitor management team (HTC experienced RN’s, RPh, SW, PT, Patient Advocate, and Hispanic Coordinator) was assembled and goals and processes for patient review and intervention were established. Monthly and ad hoc patient review meetings were implemented. Multiple barriers to adherence were encountered including immigration issues, language barriers, transportation issues, potential for caregiver burnout, storage and security of product concerns, relocations, product allergies, and needle phobia. Team worked collaboratively with HTC and other prescribers to intervene successfully in these issues. Interventions included long term twice daily nursing, securing an immigration attorney, hemophilia experienced translators, social worker, and physical therapist interventions, links to foundations for financial support, product bridge for insurance lapses, lab monitoring and reporting to HTC for remote patients, obtaining equipment (locking refrigerator) and protective supplies/ cooling compression cuffs. 30 patients received immune tolerance therapy during the study period. Of these, 25 were treated by an HTC and 5 were not. 10 patients on ITI achieved an “undetectable” inhibitor status during the study period. 8 were followed by an HTC and 2 were not. No patients were lost to therapy/management. All patients maintained positive interactions with prescribers and follow up visits.
Conclusion:
Inhibitor patients can be very challenging to manage. A focused, multidisciplinary inhibitor management team can extend the comprehensive model to the home, promote patient/prescriber/treatment team collaboration, and target interventions that enhance outcomes.
Hemophilia of Georgia's Preventive Dental Program
Rates of falls, injurious falls, and activity restriction due to fear of falling in adults with hemophilia
Don’t Push Your Luck! Educational Board (not Bored) Game for Families Living with Hemophilia
Objective:
This poster outlines evaluation of an educational family board game, “Don’t Push Your Luck!” designed to inspire discussion about hemophilia, and help school-age children learn about decision-making. Since children with hemophilia have a life-long disorder, this game provides a resource to help them learn how to make decisions for transitions to self- care.
Methods:
This game was developed based on recommendations by school-age children from previous research on partnership roles in hemophilia care. In the game, each player takes on the role of a child with hemophilia, exploring choices and consequences in everyday experiences. A multi-site, mixed method research project was coordinated by Mount Royal University, with sub-sites in Canada and United States. In phase I, the board game prototype and questionnaires from boys (n=3) and parents (n=5) living with hemophilia and boys (n=3) and parents (n=5) living with cystic fibrosis was refined. In phase II, we evaluated the revised version of the board game with children who were living with hemophilia and their household family members over age 8 years. The primary objective was to explore how playing an educational board game affected school age children’s engagement in decision-making for self-care. Children and parent perspectives were compared in the way the board game affects engagement in decision-making for children’s hemophilia self-care. Recommendations for future board game development were solicited. Purposive sampling was used to recruit household family members (n=50), including at least one parent/guardian (n= 22) and children aged 8-12 years living with hemophilia (n= 16). Two researchers visited homes to play the game, interview families, observe their responses to the game, and provide pre and post-game questionnaires on decision-making and Haemo-Quol Index© quality of life, and post-game enjoyment. Audio recordings and field notes were documented to record participant observation. Questionnaire items on decision making, quality of life observations, and game enjoyment were analyzed using descriptive statistics. Qualitative analysis of written, verbal and observed behaviours was summarized in thematic categories provided further evaluation of the board game intervention.
Summary:
Comparisons between children and adults were analyzed. Findings indicate that this game is an enjoyable and effective resource for school-age families to engage in discussions relevant to hemophilia self-care skills and decision-making.
Conclusion:
This board game is an interactive, developmentally appropriate resource for families with school-age children who are living with hemophilia to facilitate engagement and conversation about everyday life experiences in preparation for their transition to adult self- care.
This project was generously funded by an unrestricted grant from Bayer Healthcare.
Prevalence of Depression in US Patients with Hemophilia A Compared with a General Medical Population: A Retrospective Database Analysis
Objective:
Prevalence of clinical depression in persons with hemophilia (PWH) has been reported to be from 0%–50%. Most papers studied fairly small numbers of PWH; many had no controls and used instruments not validated for depression. One recent paper, using the Patient Health Questionnaire-9, a validated instrument for depression, reported a prevalence of 37% in 41 adult PWH. Our objective was to determine the prevalence of depression in PWH in the United States.
Methods:
Using the MarketScan® Commercial and Medical Research databases, we compared depression prevalence in 2506 PWH and 7518 controls. Male patients with hemophilia A were identified using an International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) code (ICD-9-CM 286.0) and were matched (based on age, eligibility months in the study, region, and health plan type) in a 1:3 ratio with controls (no hemophilia diagnosis). Evidence of depression was determined using ICD-9-CM 296.20– 296.26, 296.30–296.36, and 311 codes. Chi-square tests were used to compare frequencies.
Summary:
PWH had a statistically significant increase in depression prevalence overall and in age groups 50–59 years and 60–69 years and a numerical increase in all other age groups except 18–29 years (Table). The delta between PWH and controls steadily increased between ages 30 and 69 years.
Table. Patients with Major Depressive Disorder
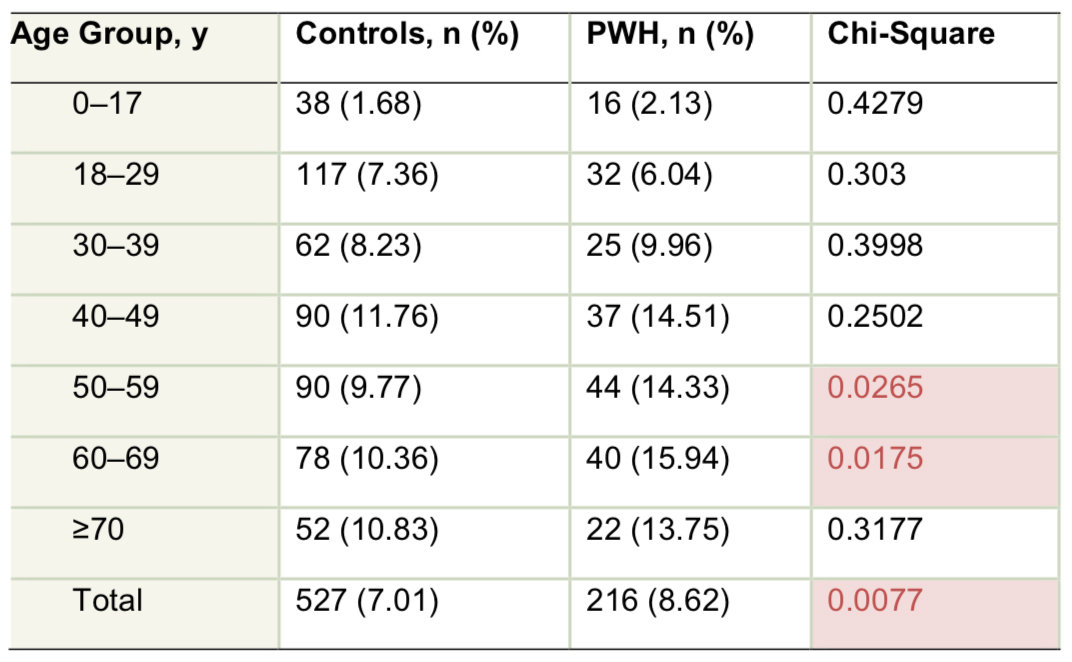
Conclusions:
Commercial claims databases have limitations, including coding errors and inability to verify accuracy of diagnoses. However, in PWH versus a 3:1 control group in a US commercially insured sample that may not be representative of the overall population, depression prevalence was greater in PWH, reaching a peak of 16% in those aged 60–69 years. Awareness of this comorbidity is important as the hemophilia population ages in an era of declining healthcare-delivery resources.
Pediatric Hemophilia and Post Trauma Stress Disorder (PTSD)
Patient, Caregiver, and Nurse Satisfaction with BAXJECT III, a Next-Generation Reconstitution System for AHF-rFVIII (ADVATE®)
FEIBA PROOF: A Prospective, open-label, randomized, parallel study with FEIBA NF to evaluate the efficacy and safety of prophylactic versus on-demand treatment in haemophilia A or B subjects with inhibitors
The PROLONG-ATE Study: A Phase 2/3 Study to Evaluate Efficacy and Safety of BAX 855,A Longer-Acting PEGylated Full-Length Recombinant Factor VIII (PEG-rFVIII),for Prophylaxis and Treatment of Bleeding in Severe Haemophilia A
A Phase I Study of Safety and Pharmacokinetics of BAX 855, a Longer Acting PEGylated Full-Length Recombinant Factor VIII (PEG-rFVIII), in Patients with Severe Hemophilia A
Outcomes of Total Knee and Hip Arthroplasty for Hemophilic Arthropathy
Objective:
To assess subjective and objective outcomes of total joint arthroplasty (TJA) as a treatment for hemophilic arthropathy, and to assess the safety and efficacy of perioperative pharmacologic thromboprophylaxis as a means to prevent venous thromboembolism in this population.
Methods:
We performed a retrospective chart review to identify patients with congenital bleeding disorders who underwent TJA between 1987 and 2012. We collected data on range of motion (ROM) and pain before and after surgery and on early and late complications (bleeding, infection, thrombosis). Data are presented descriptively using median values and ranges where appropriate.
Summary:
We identified 38 procedures (29 knees (TKA) and 9 hips (THA) in 28 patients (26 male, 2 female) with hemophilia A (n = 21), hemophilia B (n = 4), factor 11 deficiency (n = 1) and von Willebrand disease (n = 2). Median age at operation was 42 years (range, 17 – 74) for TKA and 45 years (range, 18 – 71) for THA. Inhibitors were present in one patient with hemophilia A (1.5 B.U.) and one patient with factor 11 deficiency (0.5 B.U.). All patients were treated with hemostatic agents appropriate to their disorders for up to 4 to 6 weeks post- operatively. Complete data at 2 months post-operatively are available for 27 TKA patients, of whom, 7 (23%), demonstrated improvement in ROM (median 15 degrees, range 5 - 25). At 1.5 years post-operatively, 17/29 (59%) TKA patients showed improvement in ROM (median 15 degrees, range 4 - 58) and 100% reported decreased knee pain. All 9 THA patients demonstrated improved ROM at 2 months post-operatively. Eight (89%) demonstrated gains in internal rotation (median, 45 degrees, range 15 – 45), 9 (100%) in external rotation (median 30 degrees, range 15 – 45), 5 (56%) in flexion (median 35 degrees, range 27 – 55), 7 (78%) in extension (median 15 degrees, range 3 – 95), and 7 (78%) in abduction (median 15 degrees, range 10 – 25).
We were able to contact 22 of 28 study subjects (79%), accounting for 31 of 38 (82%) procedures. Patients who underwent 25 of the 29 TKAs (86%) and 6 of the 9 THAs (67%) agreed to provide answers to yes/no questions about their experience with TJA. 25 of 25 (100%) TKA subjects reported improvement in pain and stated that if given the opportunity to go back and revisit their decision, would make the same decision to have the surgery. 24 of 25 (96%) TKA subjects reported improvement in their joint function after the surgery. 6 of the 6 THA subjects we contacted stated that they experienced improvement in joint pain and function as a result of the surgery, and 5 of 6 (83%) stated that they would choose to have the surgery if they had to choose again.
Low molecular weight heparin was administered post-operatively in 29 of 38 procedures (76%). Thromboprophylaxis was discontinued in 3 patients for non-joint bleeding (one hematuria, two cases of hypotension and anemia). There were no symptomatic VTE. Early complications included 5 cases of cellulitis and 2 hemarthroses in patients not receiving thromboprophylaxis. Late complications included two patients with aseptic loosening in prosthetic knees leading to TKA revisions, one with a subsequent joint infection requiring surgical debridement and one patient with a worsening flexion contracture requiring TKA revision.
Conclusions:
While there are risks associated with TJA in patients with bleeding disorders, our data suggest they are outweighed by the benefits manifesting as decreased pain and improved function. Pharmacologic thromboprophylaxis appears safe in this population; whether it is necessary is unknown and should be a subject of future trials.
Relationship of quality of life, pain, and self-reported arthritis with age, employment, bleed rate, and utilization of hemophilia treatment center and healthcare provider services: US results from adult patients with hemophilia in the HERO study
Objective:
Examine potential relationships between health-related quality of life (QoL), pain interference and self-reported arthritis and age, employment, activity, bleed frequency, and hemophilia treatment center (HTC) and healthcare professional utilization within the HERO psychosocial assessment study.
Methods:
In HERO, adults with hemophilia (≥18 years) from 10 countries completed a 5-point Likert scale on pain interference over the prior 4 weeks, EQ-5D-3L (mobility, usual activities, self-care, pain/discomfort, anxiety/depression) and EQ-5D health-related visual analog scale (VAS, 0-100, coded as an 11-point categorical response). US responses are considered below.
Summary:
Of 675 adults, 189 (90 with self-reported arthritis) respondents were from the US. Adults with arthritis were older; median age also increased with progressive disability and worsening pain. The percentages reporting full-time, part-time, or self-employment and the percentage reporting “good” EQ-5D VAS scores of 80-90-100 declined with increasing disability and pain interference. Median number of annual bleeds increased with increasing disability, pain interference, and arthritis. There was little difference in the median number of HTC visits per year in those reporting pain or arthritis. The percentage of adults reporting a lot/extreme pain interference was higher in those with more disability and with arthritis. Adults with increasing pain interference and arthritis were more likely to report social worker and nurse involvement. Physiotherapist utilization decreased with increasing disability and arthritis.
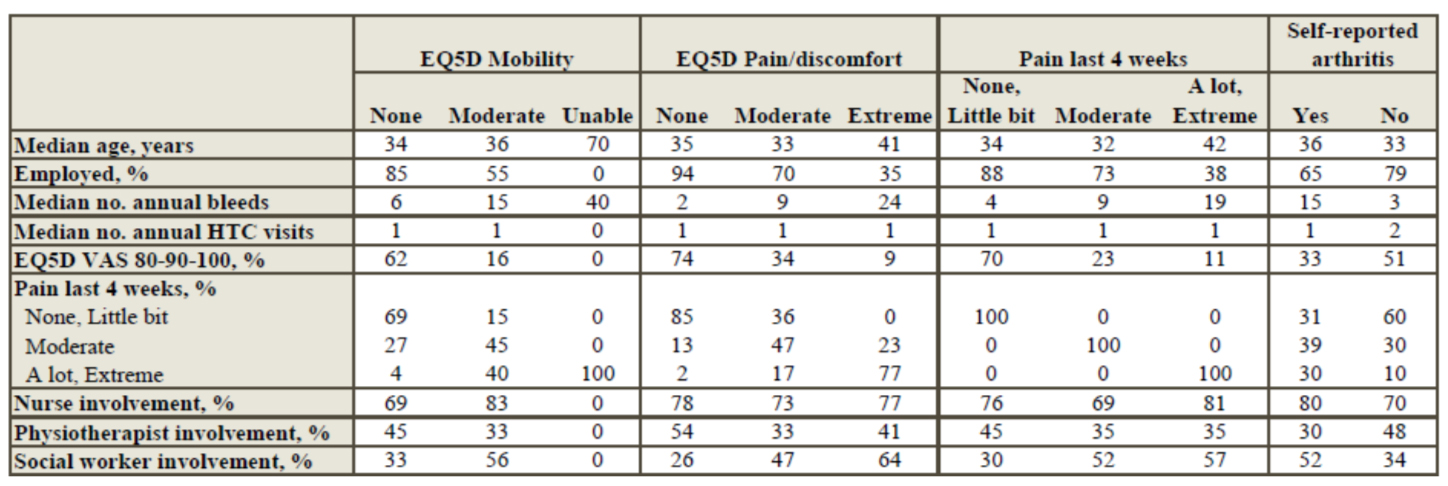
Conclusions:
In the US, increased disability and pain were associated with increased age, lower employment, higher reported bleed frequency, and lower QoL. Adults who reported experiencing more pain were more likely to report suffering from arthritis and more issues with mobility.
Atrial Fibrillation in People with Hemophilia: a Cross-Sectional Evaluation in Europe by the ADVANCE Working Group
Objective:
With increasing life expectancy of people with hemophilia (PWH) in developed countries, the number of PWH affected with age-related diseases is also increasing. Atrial fibrillation is a common health problem in the general population, but in PWH, evidence-based guidelines for the management of AF are lacking.
The aim of this cross-sectional pan-European study is to analyze the prevalence of AF and risk factors for stroke in our adult hemophilia population and to document current anticoagulation practice.
Methods:
The ADVANCE Working Group consists of members from 14 European hemophilia centers. Each center retrieved data on the number of PWH with AF in their hemophilia population, as well as their total number of adult PWH. For each person with AF, a case report form was completed.
Summary:
In total, 29 PWH with AF were documented. The mean age was 68.2 years (IQR 62-75.5). Hemophilia was severe in 6 (20.6%), moderate in 6 (20.6%) and mild in 17 (58.6%) patients. The prevalence in the total studied hemophilia population was 0.94% (29/3094) and increased with age; in patients >40 years it was 1.7% (29/1723) and in patients >60 years 3.6% (23/635). The mean CHA2DS2-Vasc score was 1.3 (IQR 0-2). Hypertension was reported in 12 patients (41.4%), diabetes in 3 (10.3%), previous stroke or TIA in 1 (3.4%), peripheral vascular disease in 4 (13.8%). In 11 patients (37.9%), anticoagulation was started of whom 9 low dose aspirin and 2 vitamin K antagonists. Of these 11 patients, 9 had mild hemophilia, 1 moderate and 1 severe with FVIII prophylaxis. During follow-up after diagnosis (mean follow-up 52.9 months), there were no thrombotic events reported, nor increases in bleeding severity.
Conclusions:
In this largest cohort of PWH with AF so far, the prevalence of AF in hemophilia increases with age and is predominantly present in mild hemophilia. Based on the population based CHA2DS2-Vasc risk scores, PWH have a low stroke risk that might be even lower considering the hypocoagulable state. Hemophilia doctors prescribe anticoagulation therapy approximately in half of their mild hemophilia patients and very few in moderate and severe.
Cardiovascular management in hemophilia: acute coronary syndromes – an assessment by the ADVANCE Working group on applicability of the ESC Guidelines
Objective:
Age is a major risk factor for cardiovascular disease. Comprehensive care and the improved safety of factor replacement therapy and therapeutic approaches, such as prophylaxis, have increased life expectancy for people with hemophilia people with hemophilia (PwH).
PwH may acquire cardiovascular risk factors (such as diabetes, hypertension, hyperlipidemia, obesity and renal disease) as a consequence of advancing age, lifestyle and hemophilia- related conditions, yet little information is available on cardiovascular risk assessment among PWH.
The ADVANCE Working Group, an expert panel of European hemophilia centers supported by an educational grant from Bayer Healthcare, convened to raise awareness of age-related comorbidities among PwH. There are currently no evidence-based guidelines for antithrombotic management in PwH presenting with acute coronary syndrome (ACS). ADVANCE met to perform a review of the current European Society of Cardiology guidelines, and to consider how best they should be adapted for PWH.
Methods:
Structured communication techniques based on a Delphi-like methodology were used to achieve expert consensus on key aspects of clinical management.
Summary:
The main final statements are: a) ACS and myocardial revascularization should be managed promptly by a multidisciplinary team that includes a hemophilia expert; b) Each comprehensive care center for adult PwH should have a link to a cardiology centre with an emergency unit and 24 hour availability of PCI; c) PCI should be performed as soon as possible under adequate clotting factor protection; d) Bare metal stents are preferred to drug eluting stents; e) Anticoagulants should only be used in PwH after replacement therapy; f) Minimum trough levels should not fall below 5-15% in PwH on dual antiplatelet therapy; g) The duration of dual antiplatelet therapy after ACS and PCI should be limited to a minimum; h) PwH receiving antiplatelet therapy should be offered gastric protection; i) The use of GPIIb- IIIa inhibitors is not recommended in PwH other than in exceptional circumstances; j) The use of fibrinolysis may be justified in PwH when primary PCI (within 90 minutes) is not available ideally under adequate clotting factor management.
Conclusion:
It is hoped that the results of this initiative will help to guide optimal management of ACS in PwH.
Prospective Study of Plasma-Derived Factor VIII/VWF in Immune Tolerance Induction Therapy: The Spirit Registry
Factor for Felons; Management of Incarcerated Hemophiliacs
Burden of Bleeding Episodes Among Persons With Hemophilia B
Objective:
To characterize the one-year bleeding pattern and assess the burden of bleeding among persons with hemophilia B, a subgroup of individuals with hemophilia that is frequently under-represented in population-based and health services surveillance.
Methods:
Hemophilia Utilization Group Study Part Vb (HUGS-Vb) collected prospective information about bleeding episodes, healthcare utilization, and burden of illness among persons with hemophilia B who obtained comprehensive care at one of ten hemophilia treatment centers in eleven geographically diverse states. Participants completed an initial interview and quarterly follow-up surveys. This analysis reports on baseline and one-year follow-up data from 93 participants. Wilcoxon-Mann-Whitney tests were used to determine the differences among subgroups. Spearman correlation coefficients (rho) were used to assess the relationship between bleeding episodes and utilization of health-related care.
Results:
Of the 93 participants, 50 (54%) were children. Forty-four (47%) participants (24 children) had severe hemophilia B, of whom 26 (59%) (16 children) reported using prophylactic therapy at initial interview. Fifty-eight (62%) participants reported having at least one bleeding episode within the one-year follow-up period. Mean number of bleeding episodes among participants with mild, moderate or severe hemophilia were 2.11±2.75, 3.47±6.25 and 6.53±7.66, respectively. Participants with severe hemophilia on prophylactic therapy had significantly (p=0.0203) fewer bleeding episodes (4.67±6.31), compared to those on episodic therapy (9.22±8.76). Significantly fewer outpatient visits (rho=0.2831, p=0.0060) and emergency room visits (rho=0.2810, p=0.0064) were reported among participants with fewer bleeding episodes. Mean absent days from work/school due to hemophilia among all participants was 2.24±4.58, and it was 0.98±2.16 days among parents of children under 18 years. Increased bleeding episodes were positively associated with increased days absent from work/school among both participants (rho=0.4598, p<0.0001) and parents of children (rho=0.3433, p=0.0147). Moreover, Positive relationship were found between increased bleeding episodes and increased time spent on the telephone with hemophilia centers (rho=0.5399, p<0.0001), pharmacists (rho=0.3373, p=0.0009) and employer/school personnel (rho=0.2230, p=0.0317).
Conclusions:
The HUGS Vb study documents the considerable burden of illness imposed by frequent bleeding episodes on persons with hemophilia B. These episodes increase patient and caregiver absenteeism from work/school, due to increased healthcare utilization and work/school coordination. As participants on prophylactic therapy have significantly fewer bleeding episodes than those on episodic therapy, prophylaxis should be encouraged among persons with hemophilia.
The use of Low Molecular Weight Heparins in Pregnancy: A single center experience 2002-2012
Prevalence and Predictors of Food Insecurity in Children with Hemophilia
Objectives:
The purpose of this pilot study was to quantify prevalence of food insecurity and determinants among households including children with hemophilia. Food insecurity, the limited or uncertain availability of nutritionally adequate and safe food, negatively affects children’s development and health. Households including people with hemophilia may be at increased risk for food insecurity due to hemophilia-related medical expenses and employment limitations.
Methods:
Food insecurity and health status, as assessed at annual comprehensive visits from May 2012-January 2013 were obtained by chart review. A two-question, validated screening tool was used to assess food insecurity status Descriptive statistics were applied to summarize participant characteristics. This study was approved by the Oregon Health & Science University Institutional Review Board.
Summary:
Data were available for forty-two male participants, aged 0-18 years, 42.9% had mild or moderate hemophilia and 57.1% severe. Prevalence of food insecurity overall was 16.7% (95% CI 5.4-28.0%), similar to national averages; food insecurity was rare among those with mild and moderate disease (5.6%) and concentrated among those with severe disease (25.0%; 95% CI 7.7-42.3%). Additionally, children who were older, taller, heavier, had higher body mass index (BMI) status, or were identified as a minority race or ethnicity were at increased risk for food insecurity (all P>0.05).
Conclusions:
This study provides pilot data showing the need for food insecurity screening and linkage to resources as a routine part of care, and the need for improved understanding of the determinants of food insecurity in this population.
B-LONG: Phase 3 Study of Long-Lasting Recombinant Factor IX Fc Fusion Protein (rFIXFc) in Hemophilia B
A-LONG: Phase 3 Study of Long-Lasting Recombinant Factor VIII Fc Fusion Protein (rFVIIIFc) in Hemophilia A
Preclinical Research with Recombinant Factor VIIa Fusion Proteins with Enhanced In vitro Activity and Improved Half-Life in Mice
H. Pylori as a cause of iron deficiency in children with bleeding disorders
Understanding the psychosocial undercurrents in spontaneous bleeds in severe hemophilia A to facilitate collaboration and customized/personalized regimens: a case study
Objective:
To examine the causes for spontaneous bleeds in severe hemophilia A patients on prophylaxis in an effort to increase care collaboration, decrease these incidents and optimize care.
Methods:
From our company’s bleeding disorders patients’ prescriptions and assessment records databases between April 1,2011 and March 31, 2012 (12 months period), prescriptions and assessment records were analyzed for prophylaxis (factor VIII) patients with severe hemophilia A who did not have inhibitors and who had at least one bleed (self- reported) requiring extra factor in the last 12 months. Due to limitations related to data retrieval from different databases, we eliminated all the patients where accuracy of match was doubtful resulting in a reduced “N”. Out of the 52 patients, 17 were identified with at least one spontaneous bleed. To identify details of psychosocial environment that might have contributed to the bleeds, chart reviews were done on five among them who reported two or more spontaneous bleeds.
Results:
Patients with four bleeds:
Patient A developed a left ankle target joint. Patient has not consistently reported bleeds to HTC. Specialty RPh notified HTC for evaluation of dose and to report bleed pattern.
Patient B frequently missed doses resulting in spontaneous bleeds.
Patients with three bleeds:
Patient C communicated well with home care and HTC. This collaboration contributed to care plan personalization and an increase in dose and frequency resulting in cessation of spontaneous bleeds.
Patients with two bleeds:
Patient D is a toddler making it difficult to identify spontaneous versus trauma induced bleeding. Homecare nurse communicated frequently with HTC leading to dosing adjustments resulting in rare spontaneous bleeds.
Patient E is a non-compliant teen and at times does not adhere to his prophy regimen.
Conclusion:
The goal of prophylaxis should be zero spontaneous bleeds. There are a variety of factors that might contribute to the bleeds such as lack of compliance, development of target joints, age, growth spurts, or improper dosing frequency and amount. Collaborating with a home infusion company or specialty pharmacy can afford an opportunity to identify and address some of these factors to take corrective actions wherever possible and hence to optimize outcomes.
Accommodating cultural needs and crossing language barriers in bleeding disorder patients: Results from a provider-patient survey by home infusion provider
Objective:
To demonstrate that providing homecare services to bleeding disorder patients with limited English proficiency in a culturally sensitive manner and in their native language can improve quality and outcomes of care.
Methods:
Provider and patient surveys were developed to measure the perceived value of interventions. Respondents were asked to rank dimensions of clarity of translated information, cultural sensitivity, satisfaction, and patient outcomes following homecare interventions on a scale of 1-9. Four patients from different ethnic backgrounds as well as their respective medical providers responded to a survey developed by the authors.
Results:
Our survey and its results, despite small numbers, demonstrate that patients as well as providers see value and improved outcomes when bilingual/bicultural professionals, interpreters and/or qualified translators were provided. Out of a total possible score of 9, an average score of 8.75 to 9 was obtained on the patient surveys. Patient respondents agreed that the information related to their treatment or care was provided in a language that was easy to understand and agreed the homecare service providers accommodated their cultural, religious or spiritual belief practices. Among the surveyed providers, services were ranked at an average of 8.75/9. The providers agreed that the language needs and cultural barriers of their patients were well addressed. It was reported that they “strongly believed” their patients “received accurate instruction on the prescribed treatment plan in a language that was easy for the patient/caregiver to understand” and addressing those needs “exceeded their expectations”.
Conclusion:
According to the Hemophilia Data Set (HDS) there has been a 236% increase in the Hispanic population and a 71% increase for other ethnic populations from 1990 to 2010. Our program has also seen a significant rise in the number of non-English speaking patients on service. This changing demographic landscape necessitates reform in the service delivery in terms of accommodating the needs of cultural diversity and overcoming language barriers. In order to do so, a continuous channel of communication is of high value to monitor and modulate changes in the population. Our survey and its results, demonstrate our success towards those ends, and hopefully will contribute to the continuous quality improvement (CQI) process in the provision of culturally competent care.
Specialty Pharmacy Educational Program, BE EMPOWERED, decreased joint bleeds in adults and increases RICE utilization among caregivers
Long-Term Follow-Up of Arteriovenous Fistulae in Bleeding Disorders
Objective:
Treatment of bleeding disorders consists of factor replacement on-demand in response to acute bleeding or prophylactically to prevent bleeding. Venous access is a critical aspect of hemophilia care. Placement of Arteriovenous Fistulae (AVF) has previously been reported (Urgo, J et al 2008). We are reporting the long-term use of the AVF and an additional 3 patients.
Methods:
All patients who received an AVF had their records reviewed and they were evaluated for their AVF usage patterns, perceived appearance, longevity of use, and overall satisfaction. The insertion protocol has been previously reported. Each patient’s AVF was assessed routinely at each clinic visit.
Summary:
There were 17 AVF insertions in 16 patients: two von Willebrand disease, 12 Hemophilia A (3 inhibitor), and 3 Hemophilia B (1 inhibitor). Mean follow-up was 5 years (1- 13 years). 15 patients had excellent results with adequate flow and patients/caregivers were able to easily access the AVF for treatment. 1 patient, who underwent 2 procedures, had a poor surgical result with inadequate blood flow to the AVF. No patients had bleeding complications from AVF creation. No patients have an AVF related infection over 5 years. Patients have not experienced any difficulty accessing the AVF for administration of factor. Four patients have reported dissatisfaction with the appearance of the AVF. All report embarrassment over appearance, self-consciousness, wear clothing to hide the AVF, and limited participation in activities where others may question the AVF. All report the AVF works well, no issues with access, and increased confidence in self-infusion. These patients all had enlarged AVF with increased blood flow as demonstrated by fistulogram. 1 patient had revision with banding that had excellent results as well as improved appearance and continued excellent intravenous (IV) access. Two more patients are scheduled for revision. The 4th patient had removal of the AVF due to increased availability of peripheral access.
Conclusion:
AVF continues to be a viable option in patients who do not have IV access and have had repeated complications with other methods of IV access. The complication rate for insertion is 3/17 (18%). Excellent IV access was achieved in 15/16 (94%) patients. Overall satisfaction is good with 9/13 (69%) patients reporting excellent function, ease of access, and satisfaction of the cosmetic appearance of the AVF.
Prolonged factor IX expression after AAV-mediated gene transfer in adults with severe hemophilia B
Examining The Impact Of Parent Demographics, Child Characteristics And Child Hemophilia Related History On Parental Stress And Parent Support
Psychosocial Intervention To Improve Compliance with Comprehensive Care Visits
Objective:
Patients with hemophilia have been receiving comprehensive care at Hemophilia Treatment Centers (HTC) for the past 30 years which includes: annual medical and psychosocial assessments, training for home infusions of factor, physical therapy evaluations and treatment.
As many of our patients with Hemophilia now self -infuse, they require less visits to the HTC, Emergency Department and hospitalizations. We have noted over the past few years that many patients are delinquent in keeping yearly comprehensive visits. In an effort to increase attendance at comprehensive clinic, and introduce and assess the success of psychosocial interventions, on increasing compliance with appointments, a standard assessment form was created to identify reasons for non-attendance at comprehensive care visits.
Methods:
Data collection included characteristics of the household such as marital status and family size, last comprehensive re-evaluation and ages of patients (< 18 or >18 years). Assessment of barriers included: drug addiction, school related issues relating to academic success, value of comprehensive clinic during a non-emergent time, DYFS involvement, major psychosocial issues, transportation and work related issues such as financial concerns and concern for using days for non-emergent reasons.
Each identified patient was contacted by the social worker to assess reasons for non- compliance utilizing the standard assessment form. Following the assessment, the social worker created and implemented an individually tailored plan developing interventions and employing education, counseling and supportive services.
Summary:
A total of 26 patients were identified as delinquent in comprehensive care visits. Eight were pediatric patients and eighteen were adult patients. All identified patients were called and assessed for barriers to compliance with clinic visits. The major barrier identified was both parents’ and patients’ value of comprehensive clinic during a non-emergent time (46 % of patients). The second major barrier was work related issues (31% of noncompliance). Major psychosocial issues (15 %) and school issues (8 %) accounted for the remaining causes of non-compliance with clinic appointments. Individual plans to address barriers were made and implemented. Following the psychosocial intervention 46 % of the previously non- compliant patients made comprehensive clinic visits.
Conclusions:
Identification of psychosocial reasons for non-compliance to comprehensive care clinic with the development of individual plans to address needs lead to improved compliance with visits.
Biodistribution of rVIIa-FP, a Recombinant Fusion Protein Linking Coagulation Factor VIIa With Albumin, in Rats
Objective:
The recombinant fusion protein linking the human coagulation factor VIIa to human albumin, rVIIa-FP (CSL Behring GmbH), is currently undergoing clinical investigation in the clinical trial program PROLONG-7P. The present study has been conducted to evaluate and better understand the biodistribution of rVIIa-FP.
Methods:
[3H]-rVIIa-FP, [3H]-rFVIIa, or [3H]-albumin were administered intravenously to male rats at a single radioactive dose of 300-400 μCi/kg. Using whole-body autoradiography, tissue radioactivity was determined up to 24 ([3H]-rFVIIa) or 240 ([3H]-rVIIa-FP, [3H]-albumin) hours. In addition to full body sections, the hind limbs were separately subjected to autoradiography to obtain more detailed information on the product distribution within the bone marrow, articular capsule, and synovial region of the knee joints. In parallel, plasma, urine, and feces were collected at pre-dose and at several sampling points throughout the 240-h study period to calculate excretion balance and assess physiological elimination pathways.
Summary:
Overall, both [3H]-rVIIa-FP and [3H]-rFVIIa were distributed predominantly into well-perfused tissues and organs and were rapidly present in synovial and mineralized regions of knee joint sections and seem to mostly localize to the zone of calcified cartilage within the growth plate regions of long bones. The longest retention time was observed in the bone marrow and endosteum of long bones. While [3H]-rVIIa-FP–associated radioactivity was well detectable at 72 h, comparable [3H]-rFVIIa–derived signals could only be observed up to 24 h after administration. The major route of elimination was urinary excretion. At 240 h, 74% and 18% of radioactivity was recovered in urine and feces, respectively. Plasma profiling showed that up to 8 h, 100% of the radioactivity could be assigned to unchanged [3H]-rVIIa- FP.
Conclusions:
Consequently, this study shows that rVIIa-FP exhibits biodistribution characteristics comparable to competitor products,1 but clearly distinguishes itself by its extended tissue half-life, potentially allowing a reduction in dosing frequency leading to increased convenience and compliance in hemophilia patients with inhibitors.
1. Nakatomi Y, et al. Thromb Res. 2012;129:62-67.
Great Plains Regional Girls with Bleeding and Clotting Disorders Camp
Effect of Albumin Fusion on the Biodistribution of Recombinant Factor IX-FP
Objective:
The present study has been conducted to explore the biodistribution of rIX-FP, a recombinant fusion protein linking the human coagulation factor IX to human albumin (CSL Behring GmbH), which is currently being investigated in clinical phase II/III trials (PROLONG- 9FP) for prophylaxis and on-demand treatment of bleeding in hemophilia B patients.
Methods:
Therefore, [3H]-rIX-FP, [3H]-rFIX, or [3H]-albumin were administered intravenously to male rats at a single radioactive dose of 320-420 μCi/kg. Using whole-body autoradiography, tissue radioactivity was determined up to 240 and 24 h following [3H]-rIX-FP and [3H]-albumin, and [3H]-rFIX administration, respectively. In addition to full body sections, the hind limbs were analyzed separately and plasma, urine, and feces were collected to calculate excretion balance and assess physiological elimination pathways.
Summary:
Overall, the tissue distribution of [3H]-rIX-FP and [3H]-rFIX was comparable; both penetrated predominantly into well-perfused tissues, were rapidly present in synovial and mineralized regions of knee joint sections, and seemed to mostly localize to the zone of calcified cartilage within the growth plate regions of long bones, with the longest retention time observed in the bone marrow and endosteum of long bones. Intriguingly, [3H]-rIX-FP signal was detectable over 72 h, whereas comparable [3H]-rFIX signal could only be detected until 24 h post-dosing. Elimination occurred primarily via the urinary route. For [3H]-rIX-FP, after 240 h, 73% of radioactivity was recovered in urine, ≤5% of radioactivity was eliminated in feces, and approximately 20% of radioactivity was present in tissues.
Conclusions:
The study shows that rIX-FP exhibits equal biodistribution compared to other marketed recombinant FIX products, but clearly distinguishes itself from rFIX (BeneFIX®) by its extended plasma half-life, allowing a reduction in dosing frequency leading to increased therapeutic convenience and compliance.
The National Hemophilia Program Coordinating Center - Assessing Support Needs of HTC Staff
Dosing intervals and bleeding rates before and following treatment with recombinant Factor IX Fc fusion protein in patients with severe hemophilia B: Experience from the B-LONG study
Adherence and outcomes in hemophilia
Objective:
Adherence to treatment has an important impact on health outcomes in chronic conditions, but the relationship between adherence to prophylactic infusions and outcomes in hemophilia is not well documented. This study was conducted to assess the relationship between adherence to prophylaxis and outcomes, including patient-reported health status and bleeding.
Methods:
Adults with hemophilia and parents of minors with hemophilia were identified through a panel of patients originally recruited from hemophilia treatment centres and associations. Panelists reporting moderate or severe hemophilia completed an on-line questionnaire, which included the Validated Hemophilia Regimen Treatment Adherence Scale-Prophylaxis (VERITAS-Pro) for adherence to prophylactic treatment, a measure of health status (adults: SF-12v2 questionnaire; parents of pediatric patients: SF-10) and items assessing the number of times they experienced clinical outcomes, such as breakthrough bleeds, ER visits, hospital admissions, and missed days from work/school due to bleeding episodes. All measures were through self- or parent-report. Generalized linear models were used to assess the relationship between adherence and outcomes, adjusting for age (adults only). The protocol and questionnaire were approved by an institutional review board and all respondents provided informed consent.
Summary (of results obtained):
A total of 53 adults with hemophilia A (n=43) or B (n=10) treated with prophylaxis completed the survey and provided age information. In analyses combining these groups, lower adherence was associated with more days of work or school missed due to bleeding episodes in the past year (p<0.05), as well as the number of bleeding episodes requiring administration of replacement factor in the past year (p<0.001). The relationship between adherence and bleeding episodes was also significant in analyses separating A and B patients (p<0.01 and p<0.05, respectively), as was the link between adherence and days missed in hemophilia A (p<0.05). Adherence was not significantly associated with physical health status (p=0.91) among adults. Among pediatric patients treated with prophylaxis (n=56), the relationship between adherence and number of bleeding episodes in the past year was not significant (p= 0.95). Adherence was associated with clinical outcomes related to bleeding episodes over the past year, such as infection at the injection site (p<0.05), hospital stay due to bleeding episodes (p<0.001), and missed days from work/school due to bleeding episodes (p<0.01). Furthermore, physical health status was better among more-adherent pediatric patients (p<0.01).
Conclusions:
Though sample sizes were limited, greater adherence to prophylaxis was associated with better self-reported clinical outcomes among both adult and pediatric hemophilia patients.
Relative importance of treatment characteristics to patients and parents of children with hemophilia
Preclinical Characteristics of a Recombinant Fusion Protein Linking Activated Coagulation Factor VII With Albumin (rVIIa-FP)
Preclinical PK/PD Characteristics of rVIII-SingleChain, a Novel Recombinant Single-Chain FVIII
Objective:
A novel recombinant coagulation factor VIII, rVIII-SingleChain, produced without added animal- or human-derived materials, is currently in a clinical phase I/III program (AFFINITY). The present non-clinical studies were conducted to investigate the pharmacokinetic (PK) profile of rVIII-SingleChain in animals to support assessment of its PK/pharmacodynamic properties for future clinical use.
Methods:
The PK behavior of rVIII-SingleChain was explored in hemophilia A mice, rats, and monkeys. Intravenous doses of 50-250 IU/kg for rVIII-SingleChain or a marketed full-length rFVIII concentrate were given. Systemic FVIII activity or antigen levels were recorded in plasma samples after injection. A thrombin generation assay was conducted to assess coagulation parameters ex vivo after treatment of hemophilia A mice with 250 IU/kg of rVIII- SingleChain or full-length rFVIII.
Summary:
In all animal species, treatment resulted in improved PK properties for rVIII- SingleChain compared to full-length rFVIII. Increased systemic availability and mean residence time were observed for rVIII-SingleChain. Correspondingly, the clearance rate was decreased and the terminal half-life was enhanced in comparison with full-length rFVIII. In vivo recovery and volume of distribution of rVIII-SingleChain were equivalent to full-length rFVIII. Consistent with the PK characteristics, rVIII-SingleChain showed a more favorable thrombin generation potential compared to full-length rFVIII between 2-6 days after treatment of FVIII-deficient mice. Results obtained showed that thrombin peak levels were kept between 50-250 nM for an increased period of time by rVIII-SingleChain compared to full-length rFVIII, with an average extension of 20 hours.
Conclusions:
The current investigations demonstrated favorable PK properties of rVIII- SingleChain in animal species. The presented results support the evidence necessary for conducting human trials to explore whether such favorable non-clinical PK characteristics may translate into clinical benefit.
Characterization of the Binding of a Novel Recombinant Single-Chain FVIII to von Willebrand Factor
Objective:
The binding behavior of rVIII-SingleChain to plasma-derived von Willebrand Factor (pdVWF) was assessed in surface plasmon resonance (SPR) studies.
Methods:
The purification of VWF from human plasma–yielded pdVWF free of factor VIII (FVIII). Subsequently, isolated pdVWF was immobilized on a SPR gold chip using monoclonal antibodies (MAbs). Thereafter, the binding behavior of rVIII-SingleChain and full-length rFVIII molecules were studied and binding kinetics were calculated. Regeneration of pd-VWF was performed with calcium chloride, while regeneration of the covalently coupled anti-VWF MAbs was achieved in the presence of an acidic pH.
Summary:
The affinity of CSL Behring’s rVIII-SingleChain to pdVWF was significantly higher than those of commercially available rFVIII full-length molecules. The higher affinity was derived from a higher association rate constant, while the dissociation rate constants were comparable. Intriguingly, the higher affinity had no influence on other functional characteristics of rVIII-SingleChain (eg, the binding to phospholipids, thrombin generation capacity, and FVIII enzymatic activity were comparable to full-length rFVIII). The results obtained from the SPR studies in vitro appear consistent with the observation of improved pharmacokinetic characteristics for rVIII-SingleChain in comparison to full-length rFVIII. After treatment of hemophilia A mice with single doses of rVIII-SingleChain or full-length rFVIII, the systemic availability and mean residence time were found to be increased for rVIII- SingleChain compared to full-length rFVIII. In addition, a decreased clearance rate and an enhanced terminal half-life were observed for rVIII-SingleChain, while in vivo recovery and volume of distribution of rVIII-SingleChain were comparable to full-length rFVIII.
Conclusion:
Overall, it seems conceivable that the higher affinity of CSLB’s rVIII-SingleChain to pdVWF may have a positive effect on its systemic availability by a delayed elimination from plasma.
Motivational Interviewing and Health Behavior Change: An Educational Intervention for Healthcare Professionals
The influence of co-morbidities on annualized bleeding rates in patients with severe hemophilia A: experiences from the pivotal turoctocog alfa prophylaxis trial (guardianTM1)
Overview of a global clinical trial program with turoctocog alfa, a new recombinant factor VIII: the guardian™ program
Turoctocog alfa, a new B-domain truncated, recombinant factor VIII (rFVIII) developed by Novo Nordisk for the prevention and treatment of bleeding episodes in hemophilia A patients
Objective:
Hemophilia A patients in the US benefit from safe, efficacious, and reliable factor VIII (FVIII) treatments. Novo Nordisk (Bagsværd, Denmark) has developed turoctocog alfa, the first new recombinant (r) FVIII in over a decade.
Methods & Summary:
Turoctocog alfa is a state-of-the-art, B-domain truncated rFVIII product manufactured without the use of human or animal proteins. Truncation of the B- domain was chosen as this domain does not have any function with respect to FVIII clotting activity. Once activated by thrombin, the turoctocog alfa truncated B-domain is cleaved, leaving an active FVIII molecule similar to the endogenous form. Turoctocog alfa is produced in Chinese hamster ovary cells, a reliable, well-established cell line used for the production of recombinant proteins for medicinal purposes. To ensure a homogenous product, isolation of turoctocog alfa uses a five-step purification process; detergent inactivation and concentration, immunoaffinity chromatography, anionic exchange chromatography, nanofiltration (20 nM filter), and gel filtration. The turoctocog alfa production method, together with its molecular structure ensures that all six FVIII tyrosines are fully sulfated. Tyrosine sulfation is important for physiologic binding of FVIII to its co-factor von Willebrand Factor, essential for FVIII stability when in circulation. Turoctocog alfa plasma concentration can be measured using standard one-stage clotting or chromogenic assays without the need for an external standard. Turoctocog alfa has been clinically tested in the guardianTM trials, one of the largest pivotal trial programs undertaken in hemophilia A with over 200 previously treated patients (PTPs) dosed. The safety and efficacy of turoctocog alfa was tested in adults and adolescents (guardianTM1) and children (guardianTM3). For adults and adolescents, turoctocog alfa had a success rate of 85% in management of bleeding episodes, and 89% of bleeds were successfully treated with 1-2 doses. For children, the success rate was 94%, and 95% of bleeds were treated with 1-2 doses. When used for prophylaxis, the median annualized bleeding rate for adults and adolescents was 3.7, while for children it was 3.0. In all surgical procedures performed in guardianTM1 and 3, the success rate was 100% with no safety concern. For both trials, no turoctocog alfa inhibitors were reported, and no safety concerns were observed. A clinical trial in pediatric previously untreated patients (guardianTM4) is ongoing.
Conclusions:
Turoctocog alfa is the new rFVIII treatment from Novo Nordisk, offering an alternative treatment option for patients with hemophilia A.
Prospective Clinical Trial of a Novel Recombinant Factor IX in Previously Treated Patients
Objective:
This prospective clinical trial was conducted to assess the safety, efficacy and PK of BAX326 (a novel recombinant FIX [rFIX] manufactured without the addition of any materials of human or animal origin, and with two viral inactivation steps [solvent/detergent treatment and nanofiltration]) in previously-treated patients aged 12 to 65 with severe (FIX level < 1%) or moderately severe (FIX level ≤ 2%) hemophilia B.
Methods:
Hemostatic efficacy after twice weekly prophylaxis with BAX326 was determined in terms of annualized bleeding rate (ABR) compared with a historical control group treated on- demand. PK equivalence was assessed between BAX326 and a commercial rFIX in a crossover design. Safety was evaluated by the occurrence of adverse events.
Summary:
In subjects on twice weekly prophylaxis with BAX326 over at least 3 months (N=56), 24 (43%) did not bleed throughout the study observation period, and the ABR was substantially lower when compared with a historical control group (79% reduction, p<0.001). Joint bleeds (major joints: wrist, elbow, shoulder, hip, knee, ankle) occurred at a mean ABR of 2.85 ± 4.25 compared with 1.41 ± 2.87 in non-joint bleed sites. Of the 32/56 subjects with bleeds, 90.6% (29/32) had arthropathy at screening and only 28.1% (9/32) did not have target joints, as compared to subjects without bleeds, of whom 79.2% (19/24) had arthropathy and 50% (12/24) did not have target joints at screening. Higher mean ABRs were observed in subjects with arthropathy (N=46) versus without arthropathy (N=8) (4.54 vs. 2.57 for all bleeds, 3.16 vs. 1.02 for joint bleeds, and 1.97 vs. 0.25 for spontaneous bleeds). A similar pattern was observed for the ABRs of joint bleeds and spontaneous bleeds in subjects with target joints (N=35) (mean ABR: 2.41 ± 3.79) and those with no target joints (N=21) (mean ABR: 0.58 ± 1.63). Most bleeds were controlled with 1-2 infusions of BAX326 and with an efficacy rating of “excellent.” BAX326 was equivalent to the comparator rFIX in terms of AUC 0 72 h /dose. BAX326 is safe and well tolerated in hemophilia B patients, with no signs of immunogenicity or thrombotic events.
Conclusions:
BAX326 has a positive safety profile and is efficacious in treating bleeds and in routine prophylaxis in PTPs aged ≥12 years with hemophilia B. The results also demonstrate that subjects with target joints and hemophilic arthropathy receiving secondary prophylaxis tend to have higher ABRs as compared to those without these underlying conditions.
Efficacy and safety of a novel rFIX (BAX326): phase III study in previously treated patients with severe or moderately severe hemophilia B undergoing surgical or other invasive procedures
Reduced Joint Range of Motion in Females with FVIII Deficiency
Objective:
Our hypothesis was that females with FVIII deficiency enrolled in the Universal Data Collection (UDC) project have reduced mean joint range of motion (ROM) compared to historic controls from the Normal Joint Study.
Methods:
We employed a cross-sectional study design utilizing the UDC dataset. The overall joint ROM was the sum of the ROM measurements of the five joints for the females with FVIII deficiency and the normal females. Results were displayed as mean overall joint ROM by age group and factor deficiency with differences between groups assessed using the Wilcoxon- rank-sum test.
Summary:
A total of 513 females were identified with FVIII deficiency; 144 females were removed because of a lack of verification for factor deficiency, one female lacking recorded range of motion data. Of the 368 females, the median age was 26 years (range 0-78). Final analysis was performed on 247 females with FVIII deficiency between the ages of 2-69 (excluding very obese females) for comparison to the control group. The mean overall joint ROM worsened with decreasing FVIII activity and in most cases was lower than that of the controls (see table 1).
Conclusions:
Females with FVIII deficiency enrolled in the UDC project had reduced mean ROM compared to normal females without deficiency.
Table 1. Mean overall joint ROM in females with FVIII deficiency by age and factor activity.
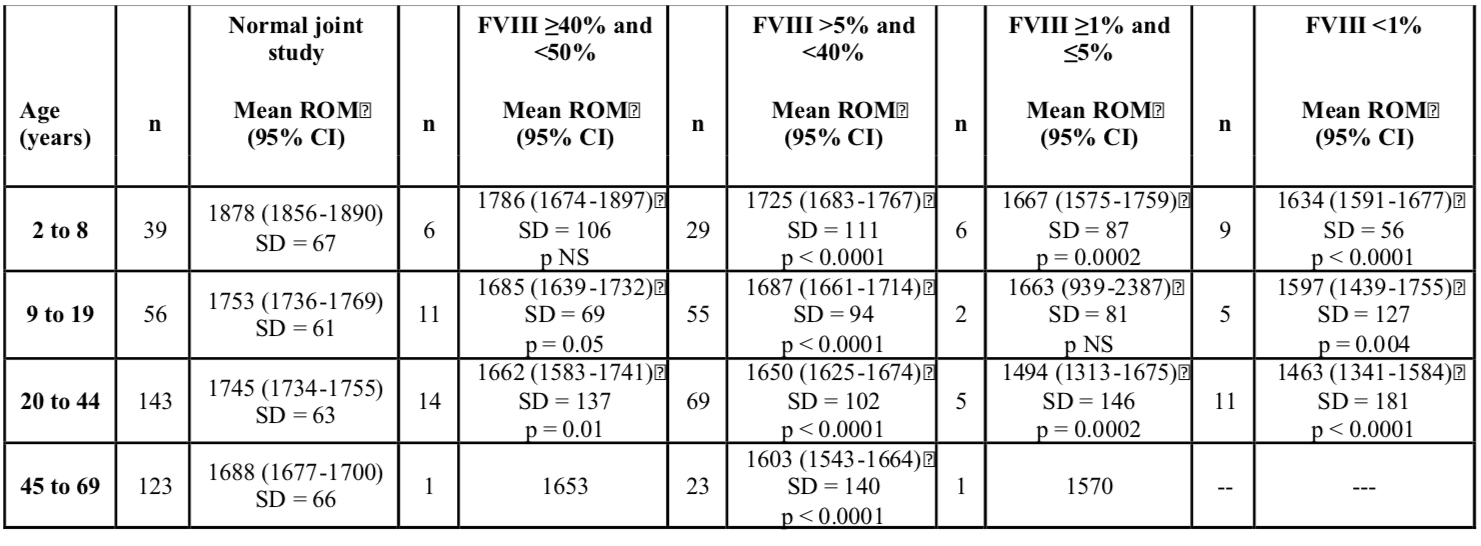
Collaborative partnership helps resolve cultural barriers in patient receiving continuous infusion of factor (ACAT Protocol) in the home setting
Objective:
When a surgical procedure is required in a patient with hemophilia, continuous infusion of factor (CIF) is a safe and effective alternative to bolus dosing.1-5 However, when cultural values collide with best practices, a patient-centered collaborative care plan is necessary to help ensure a positive outcome while respecting the core values of the patient.
Method:
Our team collaborated with our local Hemophilia Treatment Center (HTC) physicians and nurses to plan CIF for an Amish patient who required a total knee replacement. After interviewing the patient, the care team recognized when the patient transitioned on CIF to the home, we would need to respect the cultural beliefs of the patient without compromising the care.
The HTC physician and nurse ordered continuous infusion of factor for the patient with goal factor levels to remain between 70-100% on post-op days 1-7 and between 50-70% on post- op days 8-14. The home infusion team collaborated with the family and HTC team to finalize the care plan. The patient-centered decision prompted the use of a battery powered ambulatory infusion pump and the use of pre-approved sliding scale factor orders (ACAT protocol) with daily factor levels. Our biggest barrier was in respect to communication with the patient. In our local Amish community, telephones are not accessible. Our brainstorming lead to our COO suggesting we include a battery operated phone that would be attached to the pump and only be used for pump emergencies.
Summary:
The patient was discharged to home post-op day three with recombinant factor VIII running at 2.5 units/kg/hour via a battery operated ambulatory pump. Levels on post-op day four were below 70, prompting the nurse to call on the “pump phone” and return for a visit that night to increase the rate to 2.7 units/kg/hour. The rate remained the same throughout the remainder of the therapy and the levels stayed within the range designated by the physician.
Conclusion:
The patient-centered multidisciplinary care plan allowed for a positive outcome while respecting the patient’s culture.
Better Adherence to Prescribed Treatment Regimen is Associated with Less Chronic Pain among Adolescent and Young Adults with Moderate or Severe Hemophilia
Background/Aim:
Little data exist, especially for adolescents and young adults (AYAs), about the relationship between adherence to prescribed hemophilia treatment regimens and chronic pain (CP).
Methods:
A convenience sample of hemophiliacs aged 13-25 completed an IRB-approved, online survey addressing regimen-specific adherence and CP between April through December of 2012. Adherence was assessed for prophylactic (VERITAS-Pro) and on- demand (VERITAS-PRN) participants. VERITAS scores range from 24 (most adherent) to 120 (least adherent). CP was measured using the revised Faces Pain Scale (FPS-R). CP was dichotomized as high (‘moderate’ to ‘worst pain possible,’ i.e., ≥4) or low (‘mild’ or ‘no pain,’ (i.e., <4). Multivariable, parsimonious logistic regression models assessed factors associated with high vs low CP levels. Separate models were constructed to evaluate a combined VERITAS score among prophylactic and on-demand patients and the VERITAS- Pro score among prophylactic patients only. Small sample size precluded analysis of on- demand (only) participants.
Results:
Ninety-three AYAs participated. Mild patients (n=13) were excluded. Of the remaining 80 participants (79 male), 91% had severe disease, 86% infused prophylactically, and 91% had Hemophilia A. Fifty-one percent were aged 13-17, most were white (76%), non- Hispanic (88%), and never married (93%). The majority (94%) had some type of health insurance.
Mean VERITAS-Pro (n=69) and PRN (n=11) scores were 49.6 ±12.9 (range 25-78) and 51.0 ±11.6 (range 35-74), respectively. CP was reported as high for 35% of respondents (36% for prophylactic vs 27% for on-demand, p=.74). Mean VERITAS-Pro scores for those with high and low CP were 53.6 ±12.3 vs 47.4 ±12.9, p=.05. VERITAS-PRN scores were similar across CP status. Logistic regression analysis revealed that for each 10-point reduction (increase in adherence) in the combined VERITAS score (Pro and PRN) there was a 35% (OR=0.65; 95%CI=0.44, 0.96; p=.03) reduction in the odds of having high CP. Among prophylactic respondents: for each 10-point reduction in the VERITAS-Pro score there was a 39% (OR=0.61; 95%CI=0.39, 0.96; p=.03) reduction in the odds of having high CP and compared to whites, non-whites were 4.42 (95%CI: 1.21, 16.1; p=.02) times as likely to report high CP.
Oral Care for People with Von Willebrand Disease: An Unmet Need
Objective:
To assess the dental experiences of patients with von Willebrand disease for the purpose of developing guidelines for screening and dental management.
Methods:
A 13-question survey was administered to individuals at the National Outreach von Willebrand Conference, held in Phoenix, AZ in February 2012. A total of 55 respondents answered questions regarding oral hygiene habits, frequency and types of prior dental visits, dentists’ attitudes and knowledge of the disease, adverse bleeding events and quality of communication between dentist and haematologist.
Results:
Eighteen percent of respondents reported being refused dental treatment upon disclosure of von Willebrand disease history, while 81% of respondents reported that their dentist did not consult their haematologist prior to rendering treatment. More than half of those surveyed (56%) reported adverse bleeding events following dental procedures. Finally, 37 respondents reported gingival bleeding and 21 had not visited a dentist in the past six months.
Conclusions:
The results of this pilot study indicate that there is a need to educate the dental profession about von Willebrand Disease, especially its oral manifestations. Simultaneously, patients with von Willebrand Disease need to be educated as to the importance of maintaining oral health. Much more research needs to be done on the effects of poor oral health on the severity of bleeding disorders.
Speaking Frankly to Young Adults with Hemophilia
Objective:
Few online resources are available for teens and young adults living with hemophilia. Frankly.net aspires to serve as a candid, trusted resource on real issues of concern for this age group. Our online forum provides news, tips and information to help young adults with hemophilia live the lives they choose.
Method:
In 2008, an editorial board was established to guide the creation of Frankly.net, an online magazine targeting young men with hemophilia. Board members include experts in the areas of healthcare, social work and advocacy, and two are young men living with hemophilia.
Frankly.net content is controlled exclusively by the editorial board and sponsored by Bayer Healthcare with the goal of casting light on often taboo subjects within the community, such as sexuality, drugs and depression. An editorial calendar is maintained to ensure fresh content is published regularly.
Frankly.net is mobile-optimized and includes rich video content. Users are encouraged to keep up with the latest content by following @FranklyNet on Twitter.
Summary:
Since its inception, the editorial board has guided the creation of more than 80 stories. Articles include topics that resonate with young adults such as travel, entertainment, relationships and sex. Engaging video stories are also available in English and Spanish.
To date, Frankly.net has seen nearly 7,500 visitors from more than 125 countries across the globe, including the US, India, Germany, Canada.
In 2013, Frankly.net underwent a site makeover, re-launching with a new look and feel. Plans to further engage with an international audience are also underway. Korea launched a fully translated site in early 2013 under the guidance of a Korean editorial board. A Latin American version is in development and content from the site has been repurposed and translated in a dozen countries.
Conclusions:
Frankly.net is a unique resource for teenagers and young adults with hemophilia around the world. It continues to push boundaries as a way to help young men navigate the ups and downs of living with hemophilia.
Determining the impact of instrument variation and automated software algorithms on the thrombin generation test under hemophilia treatment conditions
Predicting PK/PD advantages of modified activated coagulation factor VII molecules
Stepping Up and Reaching Out to the Community
Objective:
Who better to understand and help advocate for the bleeding disorders community than those who live it every day? Step Up Reach Out (SURO) was created to help foster the next generation of leaders in the bleeding disorders community.
Method:
SURO is an international leadership program designed to help build tomorrow's leaders in the bleeding disorders community. The program was created by the University of Texas Health Science Center, Gulf States Hemophilia and Thrombophilia Center (UTHS) in 2007 with support from Bayer HealthCare.
SURO brings together young people (18-24 years old) from around the world for learning, personal growth and collaboration. This one-year program consists of two sessions of leadership training, activities focused on developing communications skills, and individual and group projects. During the time between the two sessions, participants are asked to identify an area in their communities for which they would like to step up and define an action plan, putting into practice the skills they have acquired. They continue to learn from and support one another through the SURO Alumni Network.
Summary:
To date, the SURO program has trained nearly 100 individuals from more than 20 countries, ranging from the US to Mexico, India and beyond. SURO alumni have come out of the program ready to put their action plans to the test, developing programming to support their local community needs.
Conclusions:
SURO helps participants build self-esteem, develop concrete thinking abilities and make decisions that will help make them become leaders in their own right.
Translation and Validation of an Instrument to Measure the Care-related Quality of Life of Informal Caregivers in English for the United States
Efficacy of a strength training program for improving elbow joint range of motion and function in adults with hemophilia
Objective:
To investigate the effect of modified pull-up exercises on elbow range of motion (ROM) and function for people with arthropathy secondary to hemophilia and recurrent bleeding
Methods:
- A) Participants: Men above age 18 years with hemophilia, and greater than 5 degrees ROM loss due to arthropathy from recurrent joint bleeding
-
B) Design and procedure: This pilot study was a prospective case series. Subjects were asked to perform a home exercise program consisting of modified pull ups three times per week for 8 weeks. Data was collected prior to start of program, at 3-5 weeks and at 8-10 weeks. Outcome measures included elbow ROM, pain, upper arm girth and activities of daily living (ADL) related reaching tasks. Information on how often the exercises were being performed, as well as episodes of bleeding was collected each week.
-
C) Analyses: A paired t-test was used to compare pre and post intervention measurements.
Summary:
Ten subjects have been recruited with ages ranging from 26-45 years. All have severe hemophilia A. Six subjects have completed the 8 week program to date. Those who completed the program demonstrated a mean increase of 5.3° of elbow flexion ROM (p=0.007). There was a trend toward increase in supination ROM between the two time points (p=0.058). No significant difference was seen in pre and post measurements for extension and pronation ROM. The only ADL related reaching task that demonstrated change between the two time points was palm of hand to occiput. Distance between the palm of hand to the occiput decreased 2.2 inches (p=0.009). Only 2 of the 6 subjects reported having pain in their elbow prior to the start of the program and it was unchanged during the course of the study. Arm girth data only existed for 5 of the 6 subjects and was measured at 3-5 week and 8-10 weeks. There was a trend toward increase in upper arm girth (p=0.078). There have been three reported episodes of elbow bleeding; only one was attributed to the exercises according to subject’s report.
Conclusion:
Preliminary results suggest that an eight week exercise program consisting of modified pull- ups may increase elbow flexion ROM in men with elbow joint contracture due to recurrent joint bleeding from hemophilia. Continuation of this study as well as development of others is needed to further determine if strength training is beneficial to improving elbow movement and function in those with hemophilic arthropathy.
Successful buffalo hump removal using liposuction in two men with severe hemophilia and HIV
Objective:
To describe successful buffalo hump removal in 2 patients with Hemophilia and HIV.
Methods/Case Description:
Development of a dorsocervical fat pad, or buffalo hump, occurs in 2-13 % of HIV infected individuals, the etiology is unclear. Two HIV positive patients, one with severe Hemophilia B and the other with severe Hemophilia A had symptomatic buffalo humps for 12 and 7 years respectively. Symptoms included headaches, sleep disturbances, neck stiffness and pain. These symptoms adversely affected activities of daily living. Their HIV disease was well controlled and stable. Both patients had been offered an open excision over liposuction, but the former carries a higher risk of bleeding and pain, as well as poor cosmetic result. The decision was made by a second plastic surgeon to attempt removal through liposuction. For hemostasis they received:

Both patients underwent similar liposuction procedures. 400-600cc of a tumescent solution containing epinephrine and Xylocaine was infiltrated into the subcutaneous tissue in order to minimize bleeding. Traditional liposuction was then performed through 4-5 stab incisions using a variety of cannulas with multiple passes through the fatty tissue attempting to aspirate and break up the fibrous septum connecting the buffalo hump to the cervical fascia. The deeper peri-fascial fat was removed first with later passes aimed at removing the more superficial fatty tissue. Lastly, multiple passes were made in a fan-like distribution to create an even and level contour of the back. Before closing the incisions, the remaining tumescent solution was infiltrated into the remaining cavity to prevent post-operative bleeding and to help with pain control. Both patients tolerated the procedures well with no peri-operative complications and were done in a same day surgery suite, admission was not required.
Results:
Both patients had complete resolution of pre-surgical symptoms and achieved a cosmetically pleasing outcome. Neither patient experienced any recurrence of the fat accumulation 8 and 5 months after the procedures.
Conclusions:
Liposuction is a minimally invasive modality that can be used to successfully remove buffalo humps in people with severe hemophilia, allowing for resolution of pain and improved quality of life.
Canadian social worker and nurse perspectives on the Hemophilia Experiences, Results and Opportunities (HERO) study results from Canada
Clinical Study to Investigate the Immunogenicity, Efficacy and Safety of Treatment with Human-cl rhFVIII in Previously Untreated Patients with Severe Haemophilia A
Results of a prospective, non-interventional clinical study in 170 VWD patients with a new generation of VWF/FVIII concentrate in Germany
Clinical Study in Children with Severe Haemophilia A Investigating Efficacy, Immunogenicity, Pharmacokinetics, and Safety of Human-cl rhFVIII
Use of a double virally inactivated FVIII/VWF in 30 children and young people with von Willebrands disease - a single centre experience
Global Development Plan for a Double Virus Inactivated Fibrinogen Concentrate for the Treatment of Congenital Fibrinogen Deficiency
Patients’ and Caregivers’ Perspectives on Stability of Factor VIII Products for Hemophilia A: A Web-Based Study in the United States and Canada
Pathogen Safety of Plasma-Derived Clotting Factor Concentrates Demonstrated by Validation of Inactivation and Removal Steps in the Manufacturing Process
Objective:
To assure plasma-derived clotting factor concentrates are pathogen safe, virus inactivation and removal methods are applied to the manufacturing process. The effectiveness of currently used methods and procedures was demonstrated.
Methods:
The virus reduction capacity of the manufacturing process for a specific product was quantitated for selected steps of the manufacturing process. A prerequisite of such virus validation studies is a valid downscaling of the manufacturing process to a laboratory scale. After spiking product intermediates with a panel of viruses the same as or similar to known and emerging pathogens, the manufacturing steps were performed according to the defined procedures and the reduction factors for the different viruses measured. Prion reduction was also studied, employing both a microsomal preparation (membrane-associated prions) and purified prion protein. Methods in the manufacturing process solely for the purpose of pathogen reduction, such as pasteurization, solvent-detergent treatment, dry heat treatment of the final container, and virus (nano) filtration, as well as manufacturing steps for purification and concentration of the desired protein(s) such as partitioning by precipitation or chromatography, were studied. The residual risk of transmitting pathogens was assessed based on the results of these studies and the potential virus load in the plasma pool used to produce these products.
Summary:
For a pasteurized FVIII/vWF concentrate, the overall log reduction factors were ≥10.2 for HIV, ≥11.7 for BVDV, 10.2 for PRV, 7.8 for HAV, and 6.0 for CPV, as well as 6.4 and 7.9 for the 2 prion preparations, respectively. For a FVIII/vWF concentrate using solvent- detergent and dry heat, values were ≥12.5 for HIV, ≥13.1 for BVDV, ≥11.4 for PRV, ≥12.2 for HAV, and 6.7 for parvoviruses, as well as 4.0 and 4.9 for the 2 prion preparations. For a FIX concentrate using affinity chromatography and virus filtration, values were ≥10.2 for HIV, ≥12.6 for BVDV, ≥12.8 for WNV, ≥16.1 for PRV, ≥6.7 for HAV, and ≥14.8 for parvoviruses. For a FXIII concentrate using adsorption, chromatography, heat treatment, and virus filtration, values were ≥10.2 for HIV, ≥12.6 for BVDV, ≥12.8 for WNV, ≥16.1 for PRV, ≥6.7 for HAV, and ≥14.8 for parvoviruses, as well as 9.6 and 9.7 for the 2 prion preparations.
Conclusions:
Pathogen safety is based on the overall pathogen reduction factor considerably exceeding the amount of pathogen potentially entering the manufacturing pool. Validation studies verify that safety is attained for currently manufactured plasma-derived clotting factor concentrates.
Assuring the Virus Safety of Source Plasma for Further Manufacturing by Establishing Epidemiological Limits in Donor Centers
Low inhibitor incidence in previously untreated patients with severe haemophilia A treated with octanate® - Update from the PUP-GCP clinical trial
Characteristics and Treatment Patterns of Medicaid Patients with Hemophilia A Receiving Prophylaxis vs. On-Demand Factor VIII Therapy
Objective:
Few data sources contain sufficient patient count to study the real-world efficacy of factor VIII (FVIII) therapy in patients with hemophilia A (HA). Health insurance claims databases offer that opportunity. This study sought to identify patients with HA receiving prophylaxis versus on-demand FVIII regimens and describe their characteristics and treatment patterns utilizing data from Medicaid programs.
Methods:
Healthcare claims from Florida, Iowa, Kansas, New Jersey, and Missouri Medicaid programs covering 1996 to 2011 were used. Patients with ≥2 HA diagnoses (ICD-9 286.0), ≥2 dispensings for FVIII after age 2, continuous Medicaid eligibility ≥6 months before (baseline period) and ≥12 months after the first FVIII fill, and no evidence of bypassing agents or desmopressin use were included. Prophylaxis and on-demand regimens were identified using an algorithm calibrated to distinguish the regimens within five age groups based on the annual total units of FVIII dispensed regardless of the severity level: 16,480 IU (2–7 years old); 32,770 IU (8–12); 66,920 IU (12–16); 98,349 IU (17–21); 204,552 IU (≥22). The algorithm was developed using prescription records of 1,311 patients from three U.S. specialty pharmacy databases. FVIII treatment patterns and patient demographic and clinical characteristics were examined.
Summary:
From the approximately 14.8 million covered lives encompassed in the five Medicaid databases, a total of 2,408 (0.016%) patients with ≥2 diagnoses for HA were identified; 448 met the study eligibility criteria with 229 (51.1%) patients receiving prophylaxis FVIII and 219 (48.9%) patients receiving on-demand FVIII. Younger patients were more likely to receive prophylaxis therapy (percent by age group: 61% [2–7]; 70% [8–12]; 63% [12–16]; 51% [17–21]; 20% [22+], mean (SD) age, prophylaxis: 10.8 (9.8) years old; on-demand: 18.7 (14.4) years old). Mean (range) observation period was 5.6 (1.0-15.2) years for prophylaxis patients and 5.6 (1.0-15.2) years for on-demand patients.
Conclusions:
In a population of 448 Medicaid patients with HA, this study found that 51.1% and 48.9% patients received prophylaxis and on-demand FVIII regimens, respectively. With the average observation of patients for more than 5.5 years, this database offers the potential for long-term follow-up to assess the relative efficacy of prophylaxis versus on-demand FVIII regimens in decreasing the incidence of bleeding events. These evidences from claims databases are critical to payers and decision makers as they reflect real-world clinical practice and patterns of FVIII use. Information on HA severity level was not available in the databases used.
Association of Overweight and Obesity with the Use of Self and Home-Based Infusion Therapy
The cytokine storm which follows joint bleeding: relationship of plasma cytokine concentrations and joint tissue gene expression in acute stage of hemarthrosis
The cytokine storm which follows joint bleeding: identifying a plasma signature of joint bleeding
Development of a plasminogen activator 1 (PAI-1) variant to modulate bleeding
Objective:
PAI-1 is a serine protease inhibitor (SERPIN) whose function in vivo is to downregulate fibrinolysis by inhibiting urokinase- and tissue-type plasminogen activators (uPA and tPA). The goal of this research is to use PAI-1 as a prototypical SERPIN scaffold from which to develop “designer” PAI-1 variants with altered specificity and inhibitory kinetics.
Methods:
PAI-1 variants of interest were identified using a molecular evolution approach in which traditional phage-display technology was coupled with next generation high throughput DNA sequencing. Filamentous phage displaying PAI-1 fused to the p3 coat protein were randomly mutagenized through error prone PCR, reacted with uPA, and selected with an anti-uPA polyclonal antibody in two sets of complimentary experiments to (1) determine the stable half-life of PAI-1 or (2) the rate at which PAI-1 inhibits uPA. At selected time points in each of the described experiments, the PAI-1 encoding portion of the phage genome was analyzed by next- generation sequencing. Evaluation of 7-10 million sequencing reads per time point facilitated massively parallel kinetic analyses to determine the effect of amino acid substitutions at every residue in PAI-1 simultaneously with respect to both half-life and rate of uPA inhibition.
Summary of Results:
This analysis generated data for 74% of all possible single amino acid substitutions, identifying 492 mutations that extended the functional half-life of PAI-1, as well as 1509 destabilizing mutations. These results were validated for representative single amino acid substitutions expressed as individual, purified recombinant proteins.
Conclusions:
These data provide a useful resource for interpreting human mutations identified by future large scale clinical human genome sequencing. In addition, these findings provide new insight into structure-function relationships in PAI-1. Finally, these tools lay the groundwork for future studies aimed at developing novel SERPINs based on the PAI-1 scaffold with altered target protease specific potentially applicable to treatment for a number of disorders of hemostasis and thrombosis, as well as various other SERPIN disorders (eg alpha-1-antitrypsin and C1-inhibitor deficiency).

