Bone and joint health markers in persons with hemophilia A treated with emicizumab in the HAVEN 3 clinical trial
A systematic review of mortality statistics and causes of death in people with congenital hemophilia A (PwcHA)
Objectives:
While publications have reported on mortality in PwcHA, a contemporary evidence-based understanding of mortality in congenital hemophilia A (HA) is absent. This systematic review aims to establish a benchmark of mortality rate and causes of death in PwcHA to enable comparisons and monitoring of mortality in a rapidly evolving treatment landscape.
Methods:
We conducted a systematic literature review of observational studies by searching Medline, Embase, and clinical trials registries for articles published January 2010 through March 2020, using the search terms: HA, mortality, cause of death. Interventional studies, studies not reporting fatalities, and those reporting only on hemophilia B, acquired HA, or mixed other coagulopathies were excluded. References of the included studies and literature reviews were checked.
Summary:
Overall, 7,818 unique records were identified; 1,144 manuscripts passed screening and 20 were included (Figure). In these 20 records, 6 reported mortality rates, 5 reported mortality ratios, and 16 reported cause of death. All studies reporting mortality rates and ratios were population-based; their data collection periods spanned 1961–2018, and most focused on the developed world.
Only four reports provided crude mortality rates (unadjusted for age) in the overall HA population, ranging from 0.38–0.75/100 person-years; two reported age-specific mortality rates. Age-adjusted mortality ratios generally decreased over time as life expectancies of PwcHA approached the general population. Mortality was strongly correlated with age and increased hemophilia severity. Comparisons of the risk of death in PwcHA to that of the general male population (standardized mortality or hazard ratios, adjusted for differing age distributions) ranged from 1.1–2.2 in the overall HA population (five articles) and from 2.4–6.6 in the severe HA population (three articles), indicating a raised mortality risk, particularly in severe HA. Two articles provided inconsistent mortality rates by factor VIII inhibitor status. HIV/HCV infection and liver disease were risk factors for mortality. Studies describing mortality from 1980–2000 reported a higher proportion of deaths from human immunodeficiency virus (HIV)/ hepatitis C virus (HCV).
Causes of death among PwcHA varied across populations, countries, and time in the 16 identified studies; however, underreporting of long-term outcomes limits evidence on mortality in PwcHA. Hemorrhage, HIV, HCV, and cancer were leading causes, with prevalence of cancer similar to the general population.
Conclusions:
Decreasing mortality ratios in PwcHA were observed across several decades, likely from advancements in detection, treatment and supportive care for hemophilia and related complications. Risk factors such as age and comorbidities should be considered when comparing mortality rates. Reporting of cause of death was highly heterogeneous, limiting practical categorization, hypothesis generation and actionable conclusions. A unified approach to reporting mortality and cause of death is needed to understand mortality in PwcHA and to monitor changes as treatments continue to advance.
Final Results of PUPs B-LONG Study: Evaluating Safety and Efficacy of rFIXFc in Previously Untreated Patients With Hemophilia B
Final Results of PUPs A-LONG Study: Evaluating Safety and Efficacy of rFVIIIFc in Previously Untreated Patients With Hemophilia A
Objective:
PUPs A-LONG aimed to evaluate the safety, including inhibitor development, and efficacy of extended half-life (EHL) recombinant factor VIII Fc fusion protein (rFVIIIFc) in previously untreated patients (PUPs) with severe hemophilia A.
Methods:
This open-label, multicenter, Phase 3 study (NCT02234323) enrolled male PUPs aged <6 years with severe hemophilia A (<1 IU/dL endogenous FVIII) to receive rFVIIIFc. Primary endpoint was inhibitor development (incidence rate=number of patients with inhibitors/number of patients reaching ≥10 exposure days [ED] milestone or with inhibitors). A secondary endpoint was annualized bleed rate.
Summary:
Of 103 patients receiving ≥1 dose, 80 (77.7%) were <1 year old, 20 (19.4%) had a family history of inhibitors, and 82 (79.6%) had a high-risk hemophilia genotype. Eighty-one patients started on episodic treatment; of these, 69 switched to prophylaxis. Twenty patients started on prophylaxis, and 2 were not assigned a regimen. Eighty-seven (84.5%) patients completed the study. Eighty-seven (84.5%), 85 (82.5%), and 81 (78.6%) patients had ≥10, ≥20, and ≥50 EDs to rFVIIIFc, respectively. Total and high-titer (≥5.00 BU/mL) inhibitor rate was 31.1% (28/90) and 15.6% (14/90), respectively, for patients with ≥10 EDs (3 patients with inhibitors and <10 EDs included). Median time to inhibitor development was 9 EDs (range: 1–53). rFVIIIFc dosing and efficacy data are in Table 1. Twenty-eight (27.2%) patients had 32 rFVIIIFc adverse events assessed as related by the investigator (FVIII inhibition, n=28; soft tissue hemorrhage, n=1; deep vein thrombosis, n=1; device-related thrombosis, n=1; papular rash, n=1). There was 1 non–treatment-related death due to intracranial hemorrhage (onset during screening period before first rFVIIIFc dose).
Conclusions:
This was the first prospective study of an EHL, rFVIIIFc, as treatment for PUPs with severe hemophilia A. Overall inhibitor development was within the expected range, although high-titer incidence was lower than that reported in the literature. The data demonstrate that rFVIIIFc was well tolerated and effective in this pediatric patient population.
Four-year safety and efficacy of N8-GP (ESPEROCT®) in previously treated adolescents/adults with hemophilia A in the completed pathfinder 2 trial
Objective:
The adolescent/adult pivotal phase 3 pathfinder 2 trial assessed N8-GP (turoctocog alfa pegol, ESPEROCT®) use for routine prophylaxis and treatment of bleeds in previously treated patients (PTPs).
Methods:
pathfinder 2 was a multi-center, multi-national, single-arm study evaluating safety, efficacy and pharmacokinetics. Adolescents/adults (aged ≥12 y) with severe hemophilia A were administered prophylaxis (50 IU/kg Q4D) in the main phase with option for eligible patients (0-2 bleeds in prior 6 months) to randomize (2:1) to 75 IU/kg Q7D or 50 IU/kg Q4D during extension 1 (24 weeks) and continue treatment into extension 2. An on-demand group was included throughout. Current analysis covers January 2012 through December 2018.
Summary:
Of the 186 PTPs (including 46 [25%] from the US) enrolled in the main phase, 150 (81%) started extension 1, 139 (75%) completed extension 1, and 128 (69%) completed the study. Mean age was 31.1 years, weight 75 kg and BMI 24.3.
The complete trial covers 785 patient-years of treatment (66,577 exposure days [ED]) during which there were 2,758 bleeds, including 1,807 (66%) spontaneous bleeds and 1,735 (63%) joint bleeds. Twelve patients treated on-demand for a mean 3.1 years reported nearly half of all bleeds (1,270, 46%), including 971 (54%) spontaneous bleeds and 627 (36%) joint bleeds. Hemostatic efficacy was rated excellent/good in 2,470 (90%) episodes; 2,614 bleeds (95%) were treated with 1-2 injections.
Of 175 patients on prophylaxis, 55 of 110 eligible were randomized in extension 1. For 177 patients treated with 50 IU/kg Q4D prophylaxis for 613 years (57,723 ED), 126 (71%) experienced 1,312 bleeds. For 61 low-bleed patients with 134 years (7,255 ED) on 75 IU/kg Q7D prophylaxis, 53 (87%) experienced 176 bleeds. Median ABRs are shown in the TABLE.
| 50 IU/kg Q4D | 75 IU/kg Q7D | |
| n | 177 | 61 |
| Mean treatment | 3.5 years | 2.2 years |
| Median ABR | 0.8 | 1.7 |
N8-GP mean trough levels were 3.1 IU/dL on 50 IU/kg Q4D and 1.0 IU/dL on 75 IU/kg Q7D.
A total of 1,827 adverse events were reported over 785 exposure years, including 63 serious adverse events. One patient with an intron 22 inversion developed a low-titer inhibitor at 93 ED and was withdrawn when it progressed to >5 BU. Non-neutralizing anti-PEG antibodies were seen at baseline in 12 patients (6.5%) prior to first N8-GP exposure and 11 (5.9%), who had negative anti-PEG at baseline, had positive antibodies after exposure.
Conclusion:
These data support the safety and efficacy of N8-GP in a controlled phase 3 trial setting in adolescents/adults. Prophylaxis with N8-GP with a consistent dose/interval (50 IU/kg Q4D) was effective in preventing bleeds; extended dosing was evaluated as successful for a subgroup of low-bleed patients. No significant safety issues were identified.
Five-year safety and efficacy of N9-GP (REBINYN®) in previously treated children with hemophilia B in the ongoing paradigm 5 trial
Modeling of Daily Administration of N8-GP (ESPEROCT®) vs Standard Half-life FVIII for Patients With Hemophilia A Participating in Sports Activities
Objective:
Daily administrations of FVIII products are considered useful for providing high FVIII coverage for active patients with hemophilia A. This analysis was performed to determine the daily dose levels required of N8-GP (turoctocog alfa pegol, ESPEROCT®) vs standard half-life (SHL) FVIII (N8, turoctocog alfa, Novoeight®) to normalize risk of activity-related bleeding for patients with hemophilia participating in daily sports activities (practices, games) of varying risk profiles.
Methods:
Patients with hemophilia engaging in physical activity have associated increased bleeding risk with sports that have increased potential for contact injuries as classified by Broderick et al (JAMA. 2012): Class 1 - no contact (eg, swimming); Class 2 - contact might occur (eg, basketball); and Class 3 - inevitable contact (eg, American football). To normalize the risk of bleeding, nominal targets of FVIII activity levels for at least 2 h/d based on Broderick et al were chosen: above 30% (Class 1), above 50% (Class 2), and above 70% (Class 3).
Pharmacokinetic (PK) simulations were performed using a one-compartment model with first-order elimination. FVIII PK profiles were simulated for the extended half-life (EHL) N8-GP based on the pathfinder 1 PK trial showing 60% prolonged half-life compared with prior SHL FVIII. For PK simulations of an SHL, N8 was used due to 104⁰F stability with PK based on the guardian clinical trial program.
Summary:
Daily doses to sustain at least 2 h/d of 30%/50%/70% activity were estimated for N8-GP (9, 15, and 21 IU/kg) and N8 (14, 23, and 33 IU/kg). Steady-state PK profile simulations of once-daily administration are shown in the Figure.
| Broderick Class 1 (>30%) | Broderick Class 2 (>50%) | Broderick Class 3 (>70%) | |
| N8-GP | |||
| Daily (weekly), IU/kg | 9 (63) | 15 (105) | 21 (147) |
| Peak/trough activity | 32%/13% | 54%/21% | 76%/29% |
| Difference from 50 IU/kg Q4D | -28% | 21% | 69% |
| N8 | |||
| Daily (weekly), IU/kg | 14 (98) | 23 (171) | 33 (231) |
| Peak/Trough activity | 36%/6% | 59%/10% | 87%/14% |
| Difference from 25 IU/kg QD | 13% | 97% | 166% |
| N8-GP vs N8 | |||
| Utilization, IU/kg | -36% | -39% | -36% |
Conclusion:
Experience with routine prophylaxis with EHL/SHL FVIIIs towards guideline recommended >1% activity does not readily translate to HCP understanding of the PK with high daily or much higher every-other-day administration required to minimize risk for the active patient. With a 1.6x (60%) prolongation in half-life for adolescents/adults, this model shows daily N8-GP to be a more efficient strategy compared with daily SHL FVIII (N8) to cover the active patient; N8-GP achieves higher trough levels with a smaller increase in overall factor consumption compared with standard prophylaxis with SHL FVIII.
Five-year safety and efficacy of N8-GP (ESPEROCT®) in previously treated children with hemophilia A in the completed pathfinder 5 trial
Objective:
The completed pediatric phase 3 pathfinder 5 trial assessed the safety and efficacy of N8-GP (turoctocog alfa pegol, ESPEROCT®) use for routine prophylaxis and treatment of breakthrough bleeds in previously treated children.
Methods:
pathfinder 5 was a multicenter, multinational, single-arm study evaluating safety, efficacy, and pharmacokinetics. Children (aged <12) with severe hemophilia A were administered prophylaxis (target 60 [50-75] IU/kg twice weekly) in the main phase (26 weeks) followed by an extension phase. Current analysis covers study initiation (February 2013) through completion (September 2018).
Summary:
Of the 68 children (34 aged 0-5, 34 aged 6-11) enrolled, 63 completed the main phase and 62 completed the extension. Most (95%) were previously on prophylaxis. The total study period amounted to 306 patient-years (32,138 exposure days); median (mean) patient exposure was 4.9 (4.5) years.
Overall, 838 adverse events (AEs) were reported; 18 serious AEs included 2 possibly/probably related to N8-GP (severe allergic reaction [1] and increasing bleeding symptoms [1]). No inhibitor development was observed in the trial. Two AEs resulted in withdrawal; a third patient with severe allergic reactions (after 4 doses) that resolved after 2 hours without any treatment met preestablished withdrawal criteria. There were no anti-PEG antibodies of clinical significance; however, 21 (31%) patients had anti-PEG antibodies at baseline (prior to exposure), and 1 patient had a single positive measurement after exposure at a titer <1.
Overall, 55 patients (81%) reported 330 bleeds during the study; most were traumatic (67%). The success rate for hemostasis was 84% (excellent/good); 71% were treated with 1 injection, and 88% of patients were successfully treated with 1-2 injections. Median (mean) utilization for bleeds was 68 (95) IU/kg.
Median ABRs are shown below; estimated mean ABR was 1.1. Forty-seven percent of children had no spontaneous bleeds throughout the trial. Of 13 children with 17 target joints at baseline, 77% (main phase) and 46% (complete trial) reported no bleeds in their target joints. For those previously on prophylaxis, the mean observed ABR was 2.3 compared with the historical ABR of 6.4. The mean prophylaxis dose was 64.7 IU/kg with an interval of 3.5 days.
| Median ABR | Age 0-5 y | Age 6-11 y | Total |
| Overall | 0.6 | 0.9 | 0.8 |
| Spontaneous | 0.1 | 0.2 | 0.2 |
| Traumatic | 0.3 | 0.8 | 0.5 |
N8-GP prolonged single dose half-life by 1.9x compared with the child’s prior FVIII product. The mean trough levels on twice-weekly dosing were 0.019 IU/mL (0.016 ages 0-5, 0.024 ages 6-11).
Conclusion:
These data support the safety and efficacy of N8-GP in a controlled phase 3 trial setting in children. Prophylaxis with N8-GP using a consistent dose/interval (65 IU/kg twice weekly) was effective in preventing bleeds. No unexpected safety issues were identified.
Three-year efficacy and safety results from a phase 1/2 clinical study of AAV5-hFVIII-SQ gene therapy (valoctocogene roxaparvovec) for severe hemophilia A (BMN 270-201 study)
Objective:
Hemophilia A (HA) is an X-linked disorder caused by mutations in the gene encoding Factor VIII protein (FVIII). Gene therapy is increasingly being viewed as a viable treatment option for hemophilia. Herein, long-term clinical safety and efficacy are presented from a Phase 1/2 study of an AAV-mediated gene therapy for severe HA.
Methods:
Valoctocogene roxaparvovec is an adeno-associated virus-mediated gene therapy that delivers a functional, codon-optimized, B domain-deleted, human FVIII gene under the control of a liver-specific promoter (AAV5-hFVIII-SQ). An ongoing Phase 1/2 study continues to evaluate the safety and efficacy of valoctocogene roxaparvovec in thirteen males with severe HA. Study participants received a single intravenous injection of valoctocogene roxaparvovec at one of two dose levels (6×1013vg/kg, n=7; 4×1013vg/kg, n=6).
Summary:
Participants who received 6×1013vg/kg valoctocogene roxaparvovec showed a reduction in annualized bleeding rate (ABR) of 96%, from a pre-treatment median(mean) of 16.5(16.3) to 0.0(0.7) at year three. Participants demonstrated an absence of target joints and target joint bleeds, with 86% experiencing zero bleeds requiring FVIII treatment. ABR diminished by 92% in 4×1013vg/kg participants, from a pre-treatment median(mean) of 8(12.2) to 0(1.2) at year two. Sixty-seven percent of 4×1013vg/kg participants experienced zero bleeds requiring FVIII treatment.
FVIII usage demonstrated a reduction from pre-treatment median(mean) of 139(137) infusions to 0(5.5) at year three in 6×1013vg/kg participants, and from 156(147) to 0.5(6.8) at year two in 4×1013vg/kg participants.
In 6×1013vg/kg participants, FVIII levels reported by chromogenic assay reached a median(mean) of 60.3(64.3), 26.2(36.4), and 19.9(32.7) IU/dL at the end of one, two, and three years post-infusion, respectively. In 4×1013vg/kg participants, FVIII levels reported by chromogenic assay reached a median(mean) of 22.9(21.0) IU/dL and 13.1(14.7) IU/dL at the end of one and two years post-infusion, respectively. Although FVIII levels were measured and will be presented using both the chromogenic substrate assay and the one-stage assay, chromogenic assay results appear to more accurately represent the true level of circulating FVIII.
The safety profile of valoctocogene roxaparvovec remains favorable and unchanged, with transient, asymptomatic ALT elevations and no FVIII inhibitor development reported to-date.
Conclusions:
Following a single administration of valoctocogene roxaparvovec, participants showed sustained, clinically relevant FVIII activity that reduced self-reported bleeding and exogenous FVIII replacement use at 156 weeks and 104 weeks post-administration in 6×1013vg/kg and 4×1013vg/kg dose cohorts, respectively.
Baseline patient characteristics in ReITIrate: A prospective study of rescue ITI with recombinant factor VIII Fc fusion protein (rFVIIIFc) in patients who have failed previous ITI attempts
Objective:
Inhibitor development is the most serious complication of hemophilia A therapy. Immune tolerance induction (ITI) is the gold standard for inhibitor eradication, restoring factor VIII (FVIII) responsiveness. Retrospective data on ITI therapy using rFVIIIFc have been reported (Carcao et al. Haemophilia. 2018). The ReITIrate study (NCT03103542) was designed to prospectively evaluate success of rescue ITI with rFVIIIFc.
Methods:
ReITIrate, a prospective, interventional, multicenter, open-label study, enrolled patients with severe hemophilia A and inhibitors, who failed previous ITI attempts. The primary purpose is to describe the outcome of ITI performed with rFVIIIFc (200 IU/kg/day) within a maximum of 60 weeks. Here, patient baseline characteristics are reported using descriptive statistics and listings.
Summary:
Sixteen subjects were included in the study between November 2017 and December 2018. The median (range) age at study enrollment was 7.5 (2–46) years. Seven subjects had a known family history of inhibitors. The median (range) number of prior ITI attempts was 1 (1–3) and the median (range) total ITI duration was 51.5 (12–155) months. All subjects had previously received high-dose ITI, with 3 subjects receiving plasma products, 6 subjects receiving recombinant products, and 7 subjects receiving both recombinant and plasma products for previous courses of ITI. Four subjects received prior immunomodulatory therapy. The median (range) inhibitor titer at screening and historical peak were 11 (0.9–635) BU/mL and 127 (8–3000) BU/mL, respectively. During the 12 months prior to enrollment, the median (range) number of bleeds was 5 (0–24); 11 subjects used activated prothrombin complex concentrate (aPCC) for treatment of bleeds, 5 subjects received recombinant factor VIIa (rFVIIa), and 1 subject each received FVIII/von Willebrand factor, recombinant FVIII, and tranexamic acid. Twelve subjects received prophylaxis with bypassing agents during this period (10 aPCC, 1 rFVIIa, and 1 both products).
Conclusions:
This is the first prospective study describing rescue ITI with an extended half-life recombinant FVIII product. Enrolled subjects had multiple risk factors for poor ITI outcomes and a long duration of previous ITI. There is an unmet need for successful tolerization in such patients, allowing regular FVIII prophylaxis and potentially leading to improved clinical outcomes and quality of life.
Clinical Study to Investigate the Efficacy and Safety of Wilate During Prophylaxis in Previously Treated Patients With Von Willebrand Disease (VWD)
No evidence of germline transmission of vector DNA following intravenous administration of AAV5-hFIX to male mice
Background:
Recombinant adeno-associated viruses (rAAV) are replication-deficient, non-integrating viruses commonly used as vectors for gene therapies currently in clinical development. Systemic administration of gene therapy raises the possibility of vertical germline transmission of the vector DNA.
Aim:
Here, we investigated the possibility of germline transmission following IV administration of an AAV serotype 5 vector designed for the liver-directed expression of human Factor IX which is being studied in clinical trials for hemophilia B.
Methods:
Since hemophilia B predominantly occurs in male patients, paternal germline transmission was investigated in mice in a GLP compliant study, according to current gene therapy guidelines (EMEA/273974/2005). Male C57Bl/6 mice (n=15) each received a single intravenous infusion of 2x1014 gc/kg AAV5-hFIX and were mated 6 days later with untreated female mice (n=30). On day 20 post-treatment, males were sacrificed and the seminal vesicle, epididymis, testes and a sperm sample were collected. Successfully mated females were necropsied on day 17 of gestation and the uterus, placenta and fetuses collected for each female. Each fetus was examined for viability and externally visible abnormalities. All samples were analyzed for vector DNA by QPCR.
Results:
No effect of treatment was observed on male mating performance, fertility indices, maternal body weight, food consumption, pregnancy performance, external fetal abnormalities, or fetal weights. Vector DNA levels of up to 2x106 gc/μg gDNA were detected in male reproductive tissues (epididymis, seminal vesicle, sperm, and testes), but not in female uterus, placenta and offspring. Although vector DNA was detected in the reproductive tissues of males, there was no evidence of transmission of vector DNA to female reproductive tissues or to the fetuses.
Conclusion:
The risk of paternal germline transmission following AAV5-based vector administration is therefore considered to be low.
Bleeding types and treatments in patients with von Willebrand disease before and after diagnosis
Objective:
Von Willebrand disease (VWD) is the most common inherited bleeding disorder, however, initial diagnosis and subsequent management of patients after diagnosis remains a challenge. The aim of this study was to characterize the specialists who are treating patients before and after diagnosis of VWD. We also identified the most common bleeding types and the treatments given to these patients.
Methods:
This retrospective study analysed data from a US medical claims insurance database (IQVIA PharMetrics Plus Database) for patients who had made insurance claims for VWD (International Classification of Diseases, ninth edition [ICD-9] code: 286.4). The claims were made from January 01, 2006 to June 30, 2015. Patients with ≥2 medical claims for VWD and who were continuously enrolled for a 2-year period before and after their 1 st VWD claim were included in this study. Descriptive statistics were used to summarize patient demographic and clinical characteristics, which included bleed types, treating physician specialty, and type of VWD treatment, in both the pre- and post-diagnosis periods.
Summary:
A total of 3,756 patients were included: 73% were female, and the median age at VWD diagnosis was 34 years old (age range 2–82 years). Pre-diagnosis, the top 3 treating physician specialties were hospitalists (22%), primary care physicians (14%) and obstetrician-gynecologists (13%). Post-diagnosis, the top 3 treating physician specialties were hospitalists (14%), primary care physicians (8%) and obstetrician- gynecologists (10%). Only 6% of patients saw a specialist hematologist before VWD diagnosis for a bleeding event and this decreased to 3% after diagnosis. The number of claims made by patients for bleeding events decreased from 45% pre-diagnosis to 34% post-diagnosis. In females, heavy menses were the most common bleed type, representing 29% of pre-diagnosis claims and 21% of post-diagnosis claims. In males, epistaxis was the most common bleed type, representing 13% of pre-diagnosis claims and 8% of all post-diagnosis claims. Overall, insurance claims for medical treatments associated with VWD increased from 19% pre-diagnosis to 27% post-diagnosis. The most prescribed treatments in women were oral contraceptives, desmopressin (DDAVP) and aminocaproic acid (ACA) (pre-diagnosis: 18%, 5% and 2%, respectively; post- diagnosis: 20%, 11% and 5%, respectively). In men, the most prescribed treatments were DDAVP, ACA and von Willebrand factor (VWF) concentrates (pre-diagnosis: 5%, 4% and 2%, respectively; post-diagnosis: 9%, 6% and 4%, respectively).
Conclusions:
These data show an overall reduction in the frequency of bleeding event insurance claims after VWD diagnosis. This was coupled with an increase in treatment insurance claims for DDAVP, ACA and VWF after diagnosis. These results highlight the importance of diagnosis of VWD and treatment optimization in these patients. Also, only a minority of patients received care from a hematologist, which may impact treatment and care.
Congenital afibrinogenemia: a case report of perioperative hematological management during difficult orthopedic surgery
Background:
Congenital afibrinogenemia is an autosomal recessive bleeding disorder referring to the total absence of fibrinogen measured by an antigenic assay. The commonest manifestation of the disease is bleeding from mucosal surfaces, however musculoskeletal bleeding, gynecologic and obstetric complications, spontaneous bleeding, bleeding after minor trauma and during interventional procedures or thromboembolic episodes.
Objective:
We hereby report the only case of this disorder in Slovakia with a successful perioperative management of hemostasis during revision total hip arthroplasty.Method and results: Preoperatively, the patient received fibrinogen concentrate in the dose of 75mg/kg, this dose increased the level of fibrinogen after 2 hours to corresponding 170mg/dL. During surgery, the patient received fibrinogen concentrate in the dose of 25mg/kg. The patient was administered an intraoperative transfusions because of blood loss. Twenty-four hours after surgery, the fibrinogen concentrate was applied in the patient at the dose 37.5 mg/kg every 8 hours. One day after surgery, we administered fibrinogen concentrate at the dose of 37.5 mg/kg every 12 hours with a targeted level of fibrinogen in the interval of 130-150mg/dL. We continued to reduce the dose of fibrinogen concentrate. The patient was discharged safely at 12th day after surgery with level of fibrinogen above 50mg/dL. The administration of fibrinogen concentrate was combined with low molecular weight heparin.
Conclusion:
Our results in this patient with congenital afibrinogenemia who underwent the successful repeated total left hip arthroplasty reaffirm the recommendation to tailor treatment to ensure a hemostasis balance between the replacement of clotting factor (fibrinogen concentrate) and thromboprophylaxis.
Head-to-head pharmacokinetic comparisons of N9-GP with standard FIX and rFIXFc in patients with hemophilia B
Objective:
Nonacog beta pegol (N9-GP) and recombinant factor IX-Fc fusion protein (rFIXFc) are two modified rFIX compounds with extended half-lives compared with standard FIX products. We report results from two head-to-head, single-dose pharmacokinetic (PK) trials comparing N9-GP with standard FIX and rFIXFc in previously-treated patients (PTPs) with congenital hemophilia B (≤2% FIX).
Methods:
paradigm™1 (NCT00956345) was a first human-dose trial in PTPs investigating the safety and PK of a single N9-GP dose. Sixteen PTPs (21-55 years) received one dose of their previous FIX product, followed by one dose of N9-GP at the same dose level (25, 50, or 100 IU/kg) with ≥7 days between doses. FIX activity was assessed up to 48 hours (standard FIX) or 168 hours (N9-GP) with additional samples at 2 and 4 weeks analyzed by one-stage clotting assay (TriniCLOT™) with product-specific standard as calibrator. paradigm™7 (NCT00956345) was a multicenter, randomized, head-to-head trial where 15 patients (21-65 years) received single injections (50 IU/kg) of N9-GP and rFIXFc with ≥21 days between doses. FIX activity was assessed for up to 240 hours using a one-stage clotting assay (SynthAFax or Actin FSL) and a chromogenic assay (ROX factor IX) with normal human plasma as calibrator. The primary endpoint was area under the FIX activity–time curve from 0 to infinity, dose normalized to 50 IU/kg (AUC0-inf,norm).
Summary:
In paradigm™1, the estimated terminal half-life of N9-GP was 93 hours, 4.8 times longer than for patients’ previous product. For N9-GP, estimated incremental recovery at 30 minutes (IR30min) (1.33 IU/dL per IU/kg) was 94% and 20% higher compared with rFIX and plasma-derived FIX (pdFIX), respectively. AUC0-inf,norm with N9-GP was 10.1 times and 7.7 times higher compared with rFIX and pdFIX, respectively. Time to 3% and 1% FIX activities was 16.2 and 22.5 days, respectively. In paradigm™7, the estimated AUC0-inf,norm measured with one-stage clotting assay was 4.4 times higher for N9-GP compared with rFIXFc (9656 versus 2199 IU*h/dL). IR30min was 2.2 times higher (1.7 versus 0.8 IU/dL per IU/kg), maximum activity, dose normalized to 50 IU/kg, was 2 times higher (91% versus 45%), and FIX activity at 168 hours was 5.8 times higher (19% versus 3%). N9-GP had a longer terminal half-life (103.2 versus 84.9 hours; ratio: 1.22). Results were similar when measuring FIX activity with chromogenic assay. One patient in paradigm™1 developed transient hypersensitivity symptoms during administration of N9-GP and was excluded from PK analyses. No patient developed inhibitors in either trial, and no unexpected safety concerns were identified.
Conclusion:
These two single-dose PK trials show that N9-GP achieves higher FIX activity levels and greater AUC than pdFIX, rFIX, and rFIXFc through higher recovery and longer terminal half-life. These findings will support clinicians’ understanding of differences in PK between specific FIX products.
Long-term clinical outcomes of rFIXFc prophylaxis in adults 50 years of age or older with severe hemophilia B
Objective:
Recombinant factor IX Fc fusion protein (rFIXFc) is an extended half-life therapy approved for the treatment of children and adults with hemophilia B. B-LONG parent (NCT01027364) and B-YOND extension (NCT01425723) Phase 3 studies evaluated the safety and efficacy of rFIXFc in prevention and treatment of bleeding in previously treated subjects with severe hemophilia B. This analysis evaluated clinical outcomes for over 3 years in a subgroup aged ≥50 years using B-LONG and B-YOND interim data cut 2.
Methods:
Subjects were assigned to one of the following treatment regimens: weekly prophylaxis (WP; 20–100 IU/kg every 7 days), individualized interval prophylaxis (IP; 100 IU/kg every 8–16 days), modified prophylaxis (MP-tailored dosing if IP and WP were suboptimal), and episodic treatment (ET; on-demand dosage dependent on type and severity of bleeding episode). In B-YOND, subjects could change treatment groups at any time and may appear in more than one treatment regimen. For this subgroup analysis outcomes included inhibitor development, the annualized bleeding rate (ABR), ABRs for subjects with target joints and target joint resolution, hemophilia quality of life questionnaire for adults (Hem-A-QoL), cumulative exposure, and factor consumption.
Summary:
Overall, 26 subjects ≥50 years of age (median [range], 56 [50–71] years) in B-LONG and/or B-YOND received rFIXFc (WP, n = 13; IP, n = 7; MP, n = 3; ET, n = 8). Baseline median (interquartile range [IQR]) ABR was 1 (0–5) and 20 (12–27) for subjects who received prophylaxis and on-demand treatment regimens, respectively. No subjects developed inhibitors. On-treatment overall ABRs (median [IQR]; with n ≥5) were 2.13 (1.16–4.35; WP), 1.14 (0.48–2.64; IP), and 12.83 (8.96–20.59; ET). On-treatment target joint ABR (median [IQR]; with n ≥5) was 3.17 (1.16–4.35; WP, n=9). All 19 target joints resolved with prophylactic treatment. Mean (standard deviation) total Hem-A-QoL score changed by –3.9 (10) points from baseline to last visit for 9 subjects always on prophylactic treatment during the parent and extension studies. Subjects had a median (IQR) of 3.42 (0.98–4.31) years of treatment with rFIXFc and 90 (44.0–198) cumulative rFIXFc exposure days. Factor consumption remained stable.
Conclusions:
In subjects ≥50 years of age with severe hemophilia B, these data from over 3 years of rFIXFc prophylaxis demonstrated sustained bleed control and target joint resolution while maintaining consistent factor consumption. Results are consistent with the overall study population, suggesting that rFIXFc treatment provides long-term clinical benefits for individuals with severe hemophilia B, irrespective of age and presence of target joints.
Long-term clinical outcomes of rFVIIIFc prophylaxis in adults 50 years of age or older with severe hemophilia A
Objective:
Recombinant factor VIII Fc fusion protein (rFVIIIFc) is an extended half-life FVIII therapy approved for treatment of adults and children with hemophilia A. A-LONG parent (NCT01181128) and ASPIRE extension (NCT01454739) Phase 3 studies evaluated safety and efficacy of rFVIIIFc for prevention and treatment of bleeding episodes for previously treated subjects with severe hemophilia A. A subgroup analysis was performed using A-LONG and ASPIRE interim data cut 3 and including subjects ≥50 years of age.
Methods:
Subjects were assigned to one of the following treatment regimens: individualized prophylaxis (IP; 25–65 IU/kg rFVIIIFc every 3–5 days), weekly prophylaxis (WP; 65 IU/kg every 7 days), modified prophylaxis (MP; tailored dosing if IP and WP were suboptimal), and episodic treatment (ET; on-demand dosage dependent on type and severity of bleeding episode). Subjects could change treatment groups at any time in ASPIRE and may appear in more than one treatment regimen. For this subgroup analysis outcomes included inhibitor development, the annualized bleeding rate (ABR), ABRs for subjects with target joints and target joint resolution, hemophilia quality of life questionnaire for adults (Hem-A-QoL), modified hemophilia joint health score (mHJHS), and cumulative exposure.
Summary:
Twenty-one subjects ≥50 years of age (median [range] age, 57 [50–65] years) in A-LONG and/or ASPIRE received rFVIIIFc (IP, n = 14; WP, n = 7; MP, n = 3; ET, n = 3). Baseline median (interquartile range [IQR]) ABR was 13 (6–22) for subjects who received pre-study prophylaxis and 27 (19–43) for subjects who received pre-study ET. No subjects developed inhibitors. Median (IQR) ABRs on treatment (with n ≥5) were 1.77 (0.35–5.26; IP) and 2.01 (0.25–4.76; WP). On-treatment ABRs (median [IQR]) for subjects with target joints (with n ≥5) were 1.39 (0.00–5.26; IP) and 2.01 (0.25–4.76; WP). All 49 evaluable target joints were resolved during prophylactic treatment. Mean (standard deviation) change from baseline in total Hem-A-QoL score and mHJHS was –1.9 (10.9) and –9.2 (11.43), respectively, for subjects who were always on prophylactic rFVIIIFc treatment. Subjects had a median (IQR) of 4.05 (3.04–4.26) years of treatment with rFVIIIFc and 293 (227–364) cumulative rFVIIIFc exposure days. There was no increase in weekly factor consumption.
Conclusions:
For previously treated subjects ≥50 years of age with severe hemophilia A, rFVIIIFc prophylaxis for approximately 4 years resulted in low ABRs and improved/sustained joint health without inhibitor development and without increase in factor consumption, suggesting that rFVIIIFc prophylaxis in adults ≥50 years of age has overall clinical benefits irrespective of baseline status. These data were similar to the overall study population.
Efficacy of on-demand treatment of bleeding episodes in hemophilia B patients with extended half-life N9-GP in pivotal trials: an in-depth analysis of treatment
Objective:
N9-GP is a glycoPEGylated recombinant factor-IX (rFIX) product that provides approximately two times incremental recovery, five times half-life, and 10 times area under the plasma concentration-time curve compared with standard rFIX. Phase 3 single-dose (40 IU/kg) in adults showed incremental recovery 2.34 %/IU/kg and 17% mean FIX activity at 7 days. This analysis investigates N9-GP 40 IU/kg as a single-dose on-demand (OD) treatment for hemophilia B, focusing on predictors of a second dose.
Methods:
In the paradigm™2 pivotal trial of previously treated adult/adolescent patients (≤2% FIX), the FDA requested that a group receive OD treatment prior to US enrollment into prophylaxis. This case-by-case analysis evaluated OD treatment in relationship to bleed type/pattern and prestudy treatment regimen. Hemostatic efficacy was reported by patients on a 4-point scale.
Summary:
Fifteen patients were enrolled for OD treatment (13 severe, two moderate); 13 were previously treated OD and two with prophylaxis. Overall, 14/15 patients experienced 143 bleeds during 26 weeks, of which 120 (84%) in 13 patients were treated with one dose. Seven patients (five severe, two moderate) treated all bleeds (62) with one dose (36 ‘excellent’ and 26 ‘good’ response). The other seven patients, described below, experienced 58 bleeds (72%) treated with one dose (seven ‘excellent’, 49 ‘good’, one ‘moderate’, one not reported); their other 23 bleeds (28%) required ≥2 doses (17 ‘good’, six ‘moderate’). Two of these seven had 4/11 (36%) recurrent target joints (TJ) bleeds/rebleeds treated with additional doses. An 18-year-old previously treating with plasma-derived FIX (pdFIX), 76 IU/kg/bleed, had three right elbow TJ bleeds in two months treated with one, five, and two doses. A 27-year-old previously on prophylaxis (pdFIX 100 IU/kg every 3 days and 100 IU/kg/bleed) had two bleeds in a right ankle TJ in 2 weeks treated with two doses (including one for early rebleeding) and six doses prior to withdrawing from the study. Another four patients with bleeds requiring multiple doses had been historically treated with multiple high FIX doses (IU/kg x doses per bleed: 60×2, 80×2, 80×2, and 81×3) with prescribed dosing of 120, 160, 160, and 243 IU/kg/bleed; they reported 63 bleeds, of which 45 (71%) were treated with a single 40-IU/kg dose, 17 with two, and one with four doses. Average N9-GP dosing was 44.0, 74.7, 97.9, and 52.8 IU/kg/bleed (63%, 53%, 39%, and 78% reduction in FIX use per episode). The last patient was treated prestudy with 10 IU/kg/bleed had seven bleeds (six treated with one dose, mean 46.8 IU/kg/bleed).
Conclusions:
N9-GP 40 IU/kg was effective as a single-dose OD bleed treatment (84%). Additional dose(s) for some bleeds were associated with recurrent TJ bleeds in patients not on prophylaxis or patients previously taking multiple high doses for bleeding.
An update on cognitive and behavior function in children and young adults with hemophilia: a 25-year journey from the Hemophilia Growth and Development Study to the current eTHINK study
Objective:
The Evolving Treatment of Hemophilia’s Impact on Neurodevelopment, Intelligence and Other Cognitive Functions (eTHINK) study aims to evaluate the impact of hemophilia on neurodevelopment and cognitive function through the use of validated instruments and to identify covariates that drive differences in neuropsychological performance.
Methods:
A sample of at least 510 males aged 1-21 years (~25 per age) with hemophilia A or B (any severity, with or without inhibitors) will be enrolled in a cross-sectional, non-interventional study. Following ethics review and informed consent, data collected will include a structured developmental and hemophilia history interview, a standardized neurologic examination, and a comprehensive neuropsychological assessment of cognitive/motor development (Bayley-III), intelligence (WPPSI-IV/WASI-II), attention/processing speed (CogState™), executive function (BRIEF-P/BRIEF2/BRIEF-A), mood and behavior (BASC-3), and adaptive behavior (ABAS-3). Assessments will include objective tests as well as parent and patient self-report rating scales. Z scores will be derived from published general population norms for each instrument and analyzed to develop hemophilia population specific norms. Secondary analysis for predictors of outcome will include regression modeling and chi-square tests of top vs bottom quartile responses.
Summary:
Initiated in the early 1990s under Centers for Disease Control, Maternal and Child Health Bureau, and National Institutes of Health, the Hemophilia Growth and Development Study (HGDS) evaluated the impact of hemophilia on neurodevelopment, executive function, and intelligence. The 4-year observational study enrolled 333 patients from 14 US centers, aged 6-18 years at baseline (62% HIV+), who underwent annual/semi-annual comprehensive assessments including neurologic examination, neuroimaging (MRI), and neuropsychological assessment. Results suggested that hemophilia and HIV had independent effects at baseline and follow-up. Baseline neurologic examination findings were common, as were progressive abnormalities of gait/coordination. Imaging showed baseline CNS bleeds in 12% of patients and new CNS bleeds (2% per year), which often occurred in the absence of reported head trauma. HIV+ children were more likely to show lower scores on neuropsychological assessments. Academic/adaptive skills were lower than expected based on mean IQ, and more behavioral/emotional problems were seen, including attention abnormalities related to known/silent CNS bleeds. There was a large shift in mean scores in IQ and achievement for the children with more severe hemophilia. Six small studies published between 1996 and 2009 reported impacts on academic achievement, attention, and behavior.
Conclusions:
HGDS established 25 years ago that hemophilia and HIV have independent effects on cognitive and behavioral function in children with hemophilia. Since then, standards of care in hemophilia treatment have changed significantly, but no follow-up studies have investigated whether these changes have affected the profile of neurocognitive outcomes in hemophilia. We therefore designed the eTHINK study to provide valuable insights into whether subgroups of children and young adults with hemophilia remain at risk for impaired neuropsychological outcomes.
PROTECT VIII Extension Trial Interim Data: Safety of >5 Years of Treatment With BAY 94-9027
Objective:
BAY 94-9027 is an extended–half-life recombinant factor VIII (FVIII) product. In the PROTECT VIII study, BAY 94-9027 provided effective protection against bleeds and was well tolerated with twice-weekly, every-5-day, and every-7-day prophylaxis in patients with severe hemophilia A. We report interim safety data from the PROTECT VIII extension study evaluating long-term outcomes in patients using BAY 94-9027 prophylaxis for >5 years .
Methods:
Previously treated patients aged 12 to 65 years with severe hemophilia A were enrolled in PROTECT VIII, in which they received BAY 94-9027 for 36 weeks on demand or as twice-weekly (30–40 IU/kg), every-5-day (45–60 IU/kg), or every-7-day (60 IU/kg) prophylaxis. Patients could subsequently participate in an extension study with the same or a different regimen. Adverse events (AEs), anti-PEG antibodies, inhibitor development, renal safety, and plasma PEG levels were evaluated during the extension phase.
Summary:
One hundred twenty-one of 134 patients from PROTECT VIII continued in the extension study receiving BAY 94-9027 either on demand (n=14) or as prophylaxis (n=107). At data cutoff (January 2018), patients aged 15 to 67 years at time of analysis (median age, 40 y) had a median (range) of 1420 (297–1965) days in the trial since enrollment and a median (range) of 223 (23–563) exposure days . Prophylaxis patients were treated either twice weekly (n=23), every 5 days (n=33), every 7 days (n=23), or switched frequency during the extension (n=28) . Overall, 9 patients (7.4%) experienced treatment-related AEs during the extension classified as either mild (n=4), moderate (n=4), or severe (n=1) . Two patients (1.7%) experienced 3 SAEs considered to be treatment-related (elevated liver function tests in a patient with hepatitis C ; 2 incidences of back pain); these 2 patients discontinued the study. Transient low-titer anti-PEG antibodies were detected at a single visit in 8 patients but were not associated with clinical events. No patients developed FVIII inhibitors or had sustained levels of detectable PEG in plasma . No specific changes in renal parameters were observed.
Conclusions:
During the ongoing PROTECT VIII extension, BAY 94-9027 prophylaxis was well tolerated for >5 years, and no patients developed FVIII inhibitors.
Effective Long-term Prophylaxis with BAY 94-9027 in Previously Treated Children: Interim Results of the PROTECT VIII Kids Extension Study
Objective:
BAY 94-9027 is an extended–half-life recombinant factor VIII (FVIII) product. In the PROTECT VIII Kids trial, BAY 94-9027 was efficacious for the prevention and treatment of bleeding episodes in previously treated children with severe hemophilia A. We report interim long-term efficacy and safety data from the PROTECT VIII Kids extension study.
Methods:
In PROTECT VIII Kids, previously treated patients (PTPs) aged <12 years with severe hemophilia A received BAY 94-9027 prophylaxis twice weekly (25‒60 IU/kg), every 5 days (45‒60 IU/kg), or every 7 days (60 IU/kg). Patients completing ≥50 exposure days (EDs) and ≥6 months in the main study or a 12-week safety substudy (part 2) that enrolled PTPs aged <6 years could continue in the optional extension for an additional ≥50 EDs.
Summary:
Fifty-nine of 73 patients treated with BAY 94-9027 in PROTECT VIII Kids (main study or part 2) continued in the extension (median [range] age at enrollment in the main study, 5.0 [2–11] years). At data cutoff (January 2018), patients had a median (range) of 1456 (351–1665) days in the trial (main study or part 2 plus extension). Patients in the extension received prophylaxis twice weekly (n=20), every 5 days (n=20), every 7 days (n=8), or switched prophylaxis frequency during the extension (variable frequency; n=11). Median (range) dose/infusion was 52.1 (19–62) IU/kg. Median annualized bleeding rate (ABR) for total bleeds was 1.8 for all patients and 0.8, 1.1, 2.1, and 3.2 for those treated twice weekly, every 5 days, every 7 days, or with varying frequency, respectively. Median ABR for joint bleeds in all patients was 0.7. During the extension, 3 patients (5.1%) experienced treatment-related adverse events (AEs) classified as mild (n=1), moderate (n=1), or severe (n=1). One patient discontinued because of a serious AE that was not related to treatment. No confirmed FVIII inhibitors or anti-PEG antibodies were observed; no patients had sustained levels of detectable PEG in plasma.
Conclusions:
Long-term treatment (up to ~4.5 years) with BAY 94-9027 prophylaxis was efficacious and well tolerated in previously treated pediatric patients with severe hemophilia A.
Long-term Benefit of BAY 81-8973 Prophylaxis in Children With Severe Hemophilia A: Interim Analysis of the LEOPOLD Kids Extension Study
Objective:
BAY 81-8973 (Kovaltry®) is a full-length, unmodified recombinant human factor VIII (FVIII) for prophylaxis and treatment of bleeds in patients with hemophilia A. Safety and efficacy of BAY 81-8973 in children, adolescents, and adults were established in the LEOPOLD clinical trials. This analysis reports interim data from the LEOPOLD Kids extension study for patients with ≥100 exposure days (EDs) to BAY 81-8973 in the main study plus extension study.
Methods:
In LEOPOLD Kids, boys aged ≤12 years with severe hemophilia A and ≥50 EDs to FVIII received BAY 81-8973 (25–50 IU/kg) ≥2 times/wk for ≥50 EDs. Patients completing the main study could enroll in an ongoing extension study for ≥100 EDs.
Summary:
Of 51 patients who completed the main study, 46 (90.2%) entered the extension study (aged <6 years, n=22; aged 6–12 years, n=24). Patients were treated for a median (range) of 1494 (175–1989) days and accumulated 546 (67–1011) EDs in the extension study. Median (quartile [Q]1; Q3) dose per prophylaxis infusion was 37.7 (33.1; 41.8) and 30.9 (29.1; 34.9) IU/kg for younger and older patients, respectively; annual prophylaxis dose was 4984 (3679; 6529) and 4089 (3283; 5555) IU/kg. Median (Q1; Q3) annualized number of total bleeds was 2.0 (0.2; 4.2) and 1.8 (0.5; 3.0) for younger and older patients, respectively; annualized total bleed rate was 3.0 (0; 6.0) and 0 (0; 6.4) for these patients in the main study. Median (Q1; Q3) annualized total bleeds within 48 hours of prophylaxis infusion was 0.8 (0; 1.7) and 1.0 (0.1; 1.6) in younger and older patients in the extension study. Response was excellent/good in 337/405 bleeds (83.2%); data were missing for 22 (5.4%) bleeds. Most bleeds (93.5%) were mild/moderate and were spontaneous (42.4%) or trauma related (53.6%). One patient experienced a mild treatment-related serious adverse event (transient very low FVIII inhibitor titer concurrent with acute infection and positive immunoglobulin G anticardiolipin) and remained in the extension study. No change in treatment was required, and the patient was clinically well.
Conclusions:
Data from the LEOPOLD Kids extension study show that BAY 81-8973 provides safe and effective long-term prophylaxis in children with severe hemophilia A treated for a median of 4.1 years, confirming safety results observed in the main study.
Effective Protection for >5 Years With BAY 94-9027 Prophylaxis: PROTECT VIII Extension Trial Interim Results
Objective:
In the PROTECT VIII phase 2/3 trial, the extended–half-life recombinant factor VIII product BAY 94-9027 provided effective bleed protection with twice-weekly, every-5th-day, and every-7th-day prophylaxis for 36 weeks. Herein, we report interim efficacy data from the PROTECT VIII extension study in patients receiving BAY 94-9027 prophylaxis for up to 5.4 years.
Methods:
In the PROTECT VIII trial, previously treated males aged 12 to 65 years with severe hemophilia A received BAY 94-9027 for 36 weeks on demand or for prophylaxis either twice weekly (30–40 IU/kg), every 5 days (45–60 IU/kg), or every 7 days (60 IU/kg). Patients could continue in the extension study using the same or a different regimen. Bleeds were recorded in electronic patient diaries, and annualized bleeding rates (ABRs) were calculated.
Summary:
Of 134 patients enrolled in PROTECT VIII, 121 patients aged 15 to 67 years (median age, 40 years) at the data cutoff (January 2018) continued in the extension with either on-demand treatment (n=14) or prophylaxis (n=107). At the time of analysis, patients had spent a median of 3.9 years (range, 297–1965 days) in the study since enrollment, with a median of 223 (range, 23–563) exposure days. Median (quartile [Q] 1; Q3) ABR for total bleeds during the extension study was 34.1 (20.3; 36.6) for on-demand patients and 1.6 (0.3; 4.6) for prophylaxis patients. Median (Q1; Q3) ABR for joint bleeds was 0.9 (0; 3.3) for prophylaxis patients during the extension. Median (Q1; Q3) ABR for total bleeds during the extension was similar for patients receiving prophylaxis twice weekly (1.7 [0.8; 3.6]; n=23), every 5 days (1.2 [0; 4.6]; n=33), and every 7 days (0.7 [0; 1.6]; n=23); among patients who switched prophylaxis frequency during the extension (n=28), total ABR was 3.1 (1.2; 6.2). Compared with the main study, ABR during the extension was further reduced in patients who remained in their treatment arm, including patients receiving prophylaxis every 7 days (median [Q1; Q3] main study ABR for total bleeds, 0.96 [0; 4.3]) . Of patients receiving prophylaxis, 20.6% had zero bleeds during the extension. No safety issues were identified.
Conclusions:
Good bleeding control was maintained with BAY 94-9027 prophylaxis with extended intervals of every 5 days and every 7 days throughout the PROTECT VIII extension study for up to >5 years.
BAY 94-9027 Maintains Hemostasis During Major Surgery in Adults and Adolescents With Severe Hemophilia A: PROTECT VIII Results
Objective:
BAY 94-9027 is an extended–half-life recombinant factor VIII (FVIII) product. Efficacy in maintaining hemostasis during major surgery was evaluated in a subset of patients with severe hemophilia A in the phase 2/3 PROTECT VIII study.
Methods:
Patients aged 12–65 years requiring major surgery during PROTECT VIII or its ongoing extension were included in the analysis. Patients with severe hemophilia A undergoing major surgery who were not enrolled in PROTECT VIII but met all study inclusion and exclusion criteria also were eligible to participate. BAY 94-9027 dosing during the perioperative period was based on preoperative pharmacokinetic measurements and adjusted at the physician’s discretion. Types of procedures and duration of surgeries, BAY 94-9027 consumption on the day of surgery, intraoperative blood loss, surgeon assessment of hemostasis during surgery, and need for blood transfusion were evaluated.
Summary:
17 patients (median [range] age, 37 [13–61] years) underwent 20 major surgeries, including 15 orthopedic surgeries (9 joint replacements [hip, n=1; knee, n=6; ankle, n=2], 2 open synovectomies, 3 arthroscopies, and 1 thigh hematoma evacuation), 3 complex dental extractions, 1 penile prosthesis, and 1 inguinal hernia repair at data cutoff (January 2015). Median (range) surgical duration was 102 (17‒217) minutes, and median (range) total dose used on day of surgery, including preoperative, intraoperative, and postoperative infusions on that day, was 72.4 (43‒136) IU/kg. Median (range) number of FVIII infusions on the day of surgery was 2 (1–3), with 40% of procedures requiring only 1 infusion (preoperative) on that day. Following a median (range) presurgical dose of 52.1 (41–64) IU/kg, the median (range) FVIII level (chromogenic assay; measured in a central laboratory) immediately before the second infusion was 71.6 (44–140) IU/dL; median (range) time between the presurgical and second infusions was 12.3 (3.6–50.0) hours. Hemostasis during surgery was good (13/20; 65%) or excellent (7/20; 35%) for all procedures. Intraoperative blood loss was within expected ranges for all surgeries (median [range], 50 [0–1000] mL), and blood transfusions were required in 4 patients undergoing knee surgeries.
Conclusions:
BAY 94-9027 is efficacious in maintaining hemostasis during major surgeries in adolescents and adults with severe hemophilia A. Excellent or good hemostasis with blood loss as expected was achieved in all surgical procedures, which included major orthopedic surgeries (75% of all procedures), with 40% of patients requiring only a single infusion of BAY 94-9027 on the day of surgery.
Achievement of therapeutic levels of factor VIII activity following gene transfer with valoctocogene roxaparvovec (BMN 270): Long-term efficacy and safety results in patients with severe hemophilia A
Objective:
As a single gene disorder of Factor VIII (FVIII), hemophilia A (HA) is an ideal candidate for gene therapy. We present results from an ongoing Phase 1/2 study of valoctocogene roxaparvovec (BMN 270; AAV5-FVIII-SQ) gene transfer in patients with severe HA.
Methods:
As of 16 April 2018, 13 subjects (6E13 vg/kg, n=7; 4E13 vg/kg, n=6) received a single intravenous dose of valoctocogene roxaparvovec, an AAV5 vector containing a B-domain-deleted FVIII gene. Safety, efficacy, immunogenicity, and other endpoints are being evaluated.
Summary:
FVIII activity is presented as median levels over 4-week intervals. In the 6E13 cohort, FVIII activity plateaued by Week 20 post-valoctocogene roxaparvovec, with median levels between Weeks 20-104 in the non-hemophilic range ([range] 46-122 IU/dL); Week 104 median FVIII activity was 46 IU/dL ([range] 6-145 IU/dL). In the 4E13 cohort, median [range] FVIII activity increased to just below the normal range (NR) at Week 52 [n=6]: 32 [3-59] IU/dL. Prior FVIII prophylaxis subjects had median [interquartile range, IQR] annualized FVIII infusions decline from 139 [122-157] (6E13) and 156 [126-183] (4E13) to 0 [0-0.4] and 0 [0-1] 4 weeks post-infusion through last follow-up; median [IQR] annualized bleeding rates post-infusion were 0 [0-0] in both cohorts (no bleeding episodes in 5 subjects in each cohort). Mild, grade 1, asymptomatic alanine aminotransferase (ALT) increases were reported in six of seven 6E13 and four of six 4E13 subjects; one 4E13 subject had a grade 2 ALT increase. Peak ALT levels ranged from 44-141 U/L (upper limit of normal=43 U/L). All subjects had a normal ALT level at last follow-up and all subjects were off of corticosteroid therapy. No subjects developed inhibitors to FVIII.
Conclusions:
Gene transfer with valoctocogene roxaparvovec in subjects with severe HA resulted in sustained, clinically relevant FVIII activity that reduced self-reported bleeding and exogenous FVIII use 2 years post-infusion in the 6E13 cohort. FVIII activity in the 4E13 cohort was maintained at the upper range of mild HA 1 year post-infusion. Both doses enabled achievement of long-term therapeutic levels of FVIII activity and prevention of hemophilia-related bleeding with a favorable safety profile.
Bypassing agent (BPA) use for the treatment of bleeds in persons with Hemophilia A (PwHA) with inhibitors before and after emicizumab prophylaxis in the HAVEN 1 study
Background:
Emicizumab was approved by the FDA in 2017 for routine prophylaxis in PwHA with inhibitors. HAVEN 1, a phase III study in adolescent and adult PwHA with inhibitors, demonstrated that emicizumab prophylaxis significantly reduced annualized treated bleed rate by 87% (P<0.001) vs no prophylaxis. In this retrospective, post-hoc analysis, we examined the use of BPAs to treat breakthrough bleeds before and after emicizumab initiation in HAVEN 1.
Methods:
HAVEN 1 patients were included from emicizumab-treated Arms A (previously treated with only episodic BPA) and C (previously treated with prophylactic BPA) and who had participated in the non-interventional study (NIS). In both studies, bleed and treatment data collection were comparable. In the study protocol, no guidance for the treatment of bleeds was provided; and hemostatic efficacy was not measured, thus optimal treatment of bleeds cannot be accurately assessed. Additionally, only data before October 7th, 2016 are included in this analysis to better represent treatment patterns before amended BPA guidance was provided. We describe the total number of patients and bleeds, number of infusions per bleed, and the cumulative dose/kg per bleed before and after emicizumab initiation.
Results:
This analysis (48 total patients) included 24 patients each from Arm A and Arm C who participated in the NIS prior to enrollment in the HAVEN 1 trial. On average, patients received numerically fewer activated prothrombin complex concentration (aPCC) infusions with lower cumulative doses while on emicizumab as compared to prior to emicizumab administration. In Arm A, 11 bleeds were treated with aPCC resulting in an average of 1.2 aPCC infusions/ bleed and an average cumulative aPCC dose/ bleed of 95.9 U/kg while on emicizumab as compared to 136 bleeds resulting in an average of 1.7 infusions/ bleed and cumulative dose 134 U/kg prior to emicizumab. Similar findings were seen in Arm C (bleeds, aPCC infusion/cumulative dose numbers: 14,2.3/166.6 U/kg on emicizumab compared to 205, 2.6/189 U/kg prior to emicizumab.) Fewer bleeds were treated with rFVIIa and no clear trend was seen regarding how rFVIIa was used to treat bleeds. In Arm A, 11 bleeds were treated with rFVIIa resulting in an average of 1.5 infusions/ bleed and cumulative dose 212.2 µg/kg on emicizumab, vs 97 bleeds, 1.4 infusions/ bleed and cumulative dose 181.6 µg/kg prior. In Arm C, 14 bleeds were treated with rFVIIa resulting in 4.4 infusions/ bleed and cumulative dose 555.6 µg/kg on emicizumab vs 58 bleeds, 8.6 infusions/ bleed and cumulative dose 1829 µg/kg prior.
Conclusions:
Treatment of bleeds with aPCC in HAVEN 1 resulted in numerically fewer infusions and lower cumulative doses of aPCC per bleed while on emicizumab when compared to bleeds treated prior to emicizumab initiation. Treatment of bleeds with rFVIIa showed no clear trend.
Change in cost and units consumed by people with factor VIII and factor IX deficiency after switching from a standard half-life product to an extended half-life product
Objective:
To examine whether a difference exists in factor consumption and cost after a person with factor VIII or factor IV switches from a standard half-life (SHL) factor product to an extended half-life (EHL) factor product managed by a single specialty pharmacy.
Methods:
All electronic medical records of patients with factor VIII or IX deficiencies who filled factor prescriptions with a single specialty pharmacy were reviewed. Data, including regimen, dosing and label instructions for fills of all recombinant and plasma-derived factor VIII and factor IX products between September 2013 and December 2017 were examined retrospectively. Adult patients (age ≥ 18 years) who switched from an SHL to an EHL product with 6 months of fill data available, for both periods before and after the initial EHL prescription was dispensed, were eligible for inclusion. Total cost was calculated using average wholesale price (AWP) using MediSpan (accessed June 1, 2018).
Summary:
Sixty-three (63) people with factor VIII deficiency and 29 people with factor IX deficiency met study criteria by having a prescription for factor and switching from an SHL product to an EHL product, as well as 6 months of dispensing prior to and after the change. The mean cost of therapy with an SHL factor VIII product for 6 months was $272,192 versus $455,260 for 6 months after switching to an EHL factor VIII product, an increase of 67%. The mean number of units of an SHL factor VIII product dispensed was 141,934 units compared to 181,688 units of an EHL factor VIII product per person, which is an increase of 28%. The mean cost of therapy with an SHL factor IX product for 6 months was $234,146 versus $474,858 for the 6 months after switching to an EHL factor IX product, an increase of 100%. The mean number of units of an EHL factor IX product dispensed was 147,730 units compared to 124,509 of an EHL factor IX product, a decrease of 16%.
Conclusion:
EHL products have several reported advantages over SHL products including longer interval between infusions, less frequent troughs (that may lead to decreased risk of bleeding episodes) and improved adherence. This analysis of dispensing data from a single specialty pharmacy indicates that change of prescription from SHL products to EHL products is associated with a higher cost of treatment. There are many more elements in factor prescribing that could contribute to future discussion.
Integrated efficacy and safety analysis of Phase 2 and 3 studies with glecaprevir/pibrentasvir in patients with a history of bleeding disorders and chronic hepatitis C virus genotype 1–6-infection
Objective:
Hepatitis C virus (HCV) infection is a significant health problem for patients with bleeding disorders due to the absence of highly effective HCV screening of blood products prior to the early 1990s. The aim of this analysis was to evaluate the safety and efficacy of the ribavirin-free regimen of glecaprevir/pibrentasvir (G/P) among patients with a history of bleeding disorders.
Methods:
Data from 11 Phase 2 and 3 registrational studies conducted across 18 countries worldwide were included. HCV genotype (GT) 1–6-infected patients with compensated liver disease received G/P for 8, 12 or 16 weeks. The sustained virologic response 12 weeks after end of treatment (SVR12) rate and safety are reported.
Results:
Sixty-two patients with history of bleeding disorders were identified, the most common disorder being hemophilia (37%, [23/62]). The remaining 63% (39/62) of patients had other disorders, including Von Willebrand disease. The cohort was 76% (47/62) male and 89% (54/62) white race with a mean age of 50.5 years (standard deviation ± 12.1). Additionally, 23% (14/62) had compensated cirrhosis. The HCV genotype breakdown among the patients was: GT1, 39%; GT2, 18%; GT3, 21%; GT4, 13%, GT5, 8%; GT6, 2%. In addition to bleeding disorders, a medical history of anemia was reported in 16% (10/62) of patients. SVR12 was achieved in 100% (62/62) of patients. Adverse events (AEs) were reported in 68% (42/62) of patients, and 37% (23/62) of patients experienced an AE assessed as possibly related to study drugs. Serious AEs (SAE) were reported in 3 (5%) patients, none being assessed as related to the study drugs; 2 of these SAEs (1 per patient) were associated with the underlying bleeding disorder. There was 1 (2%) AE leading to discontinuation of study drug (dyspepsia); the patient went on to achieve SVR12. There was 1 (2%) non-treatment emergent death reported, occurring 60 days after the last dose of study drug. Post-baseline grade ≥3 laboratory abnormalities occurred in 3 patients: one each in hemoglobin, aspartate aminotransferase and bilirubin (all n = 1; 2%), without concurrent elevations in alanine aminotransferase.
Conclusions:
G/P achieved a 100% SVR12 rate in patients with a history of bleeding disorders and demonstrated a favorable safety profile, thus providing support for treatment of chronic HCV infection with the G/P regimen in this patient population.
A Multicenter, Retrospective Data Collection Study on the Compassionate Use of a Plasma-Derived Factor X Concentrate to Treat Patients with Hereditary Factor X Deficiency
Objective:
Report results of an open-label international study that collected retrospective data on compassionate use of high-purity plasma-derived FX concentrate (pdFX) in subjects with hereditary factor X (FX) deficiency (FXD).
Methods:
This study included subjects with hereditary FXD (irrespective of severity) who received compassionate use pdFX as routine prophylaxis (RP), on-demand (OD) treatment, short-term prevention, and/or perisurgical hemostatic cover. Dosing was at the investigator’s discretion and tailored to each patient. Data from date of first compassionate use dose until data cutoff (31 December 2015) were collected retrospectively.
Summary:
All 15 enrolled subjects from 12 study centers received ≥1 pdFX dose for compassionate use. Of these, 13 subjects were aged ≥12 years (mean, 22.8 years) and 2 were aged <12 years, 8 (53.3%) were female, 12 (80.0%) were white, 3 (20.0%) were Asian. All subjects had moderate or severe FXD (FX activity [FX:C] <5 IU/dL).Of the 15 patients, 7 received only RP, 7 received only OD, and 1 alternated between OD and RP. The 8 subjects on RP received a total of 1239 RP infusions (mean, 154.9 infusions/subject, range 39–492), with a mean dose/infusion/subject of 32.5 IU/kg. The 2 subjects aged <12 years received larger RP doses than the 6 older subjects (mean doses/infusion/subject of 51.1 vs 26.3 IU/kg).Twelve subjects (8 OD, 4 RP; all aged ≥12 years) reported 88 bleeds (34 minor, 7 major, and 47 not rated); 37 bleeds were menorrhagic, 28 were traumatic, 17 were spontaneous, 4 were other, and 2 had unknown cause. pdFX efficacy was rated as effective for the 79 bleeds (including 1 subdural hematoma) treated with OD pdFX. Mean pdFX dose was 22.2 IU/kg/infusion/subject, with a mean of 9.5 infusions/subject to treat a bleed. More bleeds occurred in the OD than in the RP population.Two subjects underwent 1 dental procedure each, with only 1 presurgical pdFX dose required per patient; a third surgery, a portacath insertion, required 6 infusions to prevent postoperative bleeding. Two successful pregnancies/childbirths were also reported, with no abnormal bleeding complications or efficacy/safety concerns reported.The mean duration of compassionate use was 87.6 weeks for the 15 subjects, with a range of 15–211 weeks (0.3–4.0 years). Over the 1373 infusions administered across 25.2 subject-years, investigators rated overall pdFX efficacy as excellent in 14 (93.3%) subjects and good in 1 (6.7%) subject. No adverse drug reactions, safety concerns, infusion site reactions, tolerability issues, or inhibitor development were reported during pdFX compassionate use.
Conclusions:
The higher bleed rate in OD versus RP use and the treatment duration (up to 4 years) support the efficacy and safety of pdFX demonstrated in prospective clinical studies and its continued use in the treatment of subjects with hereditary FXD.
Staying on TRAQ: Determining transition readiness from pediatric to adult care in adolescents and young adults with hemophilia
Advancements in medical care for the hemophilia patients has created the need for an active and intentional process of transition from pediatric oriented health care to adult oriented health care. Instruments to measure transition “readiness” have not been validated in the adolescent and young adult (AYA) hemophilia population. The primary aim of this study was to identify the baseline state of transition readiness of hemophilia males in their ability to take charge of their own care as they transition from pediatrics to adult care using a validated Transition Readiness Assessment Questionnaire (TRAQ). Our secondary objective was: 1) To compare transition readiness between young adults who are transitioned to an adult hemophilia clinic in a separate facility versus those who continue to receive care in the same facility but transitioned to an adult provider.
Methods:
For this purpose, we conducted a cross-sectional study at the Hemophilia study of Western New York (HCWNY), Buffalo (same facility) and at Children’s Hospital of Michigan (CHM), Detroit (separate facility). Inclusion criteria: 1) males who are currently 16-21 years old, 2) diagnosis of hemophilia A and B regardless of severity, 3) ability to read English at a grade 8 level. TRAQ is a 20-item, 5- domain patient-reported assessment of health and health care self-management skills with possible scores ranging from 1 (low) to 5 (optimal). Parents/legal guardians of patients aged 16 to 17 and patients aged 18-21 were mailed a letter explaining the study in detail. The willing participants were directed to the questionnaire link. The responses were anonymous which were directly imported into excel spread sheet without collecting any identifying information of the participants.
Results:
A total of 13 individuals at the two sites participated. Amongst these, there were 5 from the HCWNY site, 1 with mild hemophilia and 1 patient reported unknown severity. The mean overall TRAQ score was 4+0.8. The mean scores for the different subscales were: managing medications (4.2+0.8), appointment keeping (4.1+0.9), tracking health issues (3.6+1.2), talking with providers (4.4+1.2) and managing daily activities (4.1+1.1). No differences in the overall and the subscales scores was noted between the two centers (Wilcoxon rank sum for all p>0.05).
Conclusions:
Our results suggest that the hemophilia youth in our population appear to have good readiness to transition from pediatric to adult care. We did not find a difference between the two different clinical care settings. The tracking health issues portion of TRAQ demonstrated the least readiness. Our study demonstrates that transition readiness assessments can be implemented in the hemophilia treatment centers, which can be used to guide clinical care.
Efficacy, safety and pharmacokinetics of once-weekly prophylactic emicizumab (ACE910) in pediatric persons (<12 years) with hemophilia A with inhibitors: interim analysis of single-arm, multicenter, open-label, phase 3 study (HAVEN 2)
Objectives:
Emicizumab, a novel bispecific humanized monoclonal antibody promotes coagulation by bridging FIXa and FX to replace the function of missing activated FVIII, and has potential to address unmet medical needs in pediatric persons with hemophilia A (PwHA) with inhibitors. This study assessed efficacy, safety and pharmacokinetics of once-weekly subcutaneous emicizumab prophylaxis in pediatric PwHA with inhibitors.
Methods:
The study (NCT02795767) enrolled PwHA with inhibitors aged <12 years (or 12–17 years if <40 kg) previously treated with bypassing agents to receive emicizumab prophylaxis for ≥52 weeks. Emicizumab was administered subcutaneously at 3 mg/kg/week for 4 weeks, followed by 1.5 mg/kg/week thereafter. Efficacy objectives included bleed rate, and comparison of the bleed rate on emicizumab prophylaxis vs historical bleed rate obtained from a prospective, non-interventional study (NIS; NCT02476942). The NIS collected detailed, high-quality real- world data on bleeds and safety outcomes from a cohort of pediatric PwHA with inhibitors treated according to local, routine clinical practice. Participants from the NIS could subsequently enter the HAVEN 2 study, which permitted intra-individual comparisons.
Summary:
This interim analysis included 20 PwHA with inhibitors aged 3–12 years (median 8.5); 19 aged <12 years were included in the efficacy analyses. The median observation time was 12.1 weeks (range 7–14). In total, 18/19 (94.7%) participants had zero treated bleeds and 12/19 (63.2%) did not bleed while on study. Overall, 14 bleeds were reported in 7 participants, with none occurring in a joint or muscle. No participants have required up- titration of emicizumab. A substantial reduction in ABR on study vs ABR on prior treatment with bypassing agents (non-interventional study) was observed in 8 participants included in the intra-individual comparison; all 8 participants reported zero bleeds with emicizumab prophylaxis (efficacy period 85–99 days). Emicizumab was well tolerated; most common AEs were mild injection-site reactions (15%) and nasopharyngitis (15%). Three unrelated serious AEs were observed (mouth hemorrhage, appendicitis, catheter site infection). No thromboembolic or thrombotic microangiopathy events were reported. No anti-drug antibodies were detected. Mean trough emicizumab concentrations of >50 μg/mL were achieved after 4 loading doses of 3 mg/kg/week and sustained with maintenance doses of 1.5 mg/kg/week, and were consistent across age groups and body weight.
Conclusion:
Emicizumab prophylaxis was well tolerated and prevented/reduced bleeds in pediatric PwHA with inhibitors. Clinically meaningful reductions in ABR were observed compared with ABR on prior treatment with bypassing agents. The pharmacokinetic profile of emicizumab was similar to that seen in adolescent/adult PwHA with inhibitors. These interim data show the potential for emicizumab to reduce the disease and treatment burden for pediatric PwHA with inhibitors.
Efficacy, safety and pharmacokinetics of emicizumab (ACE910) prophylaxis in persons with hemophilia A with inhibitors: randomized, multicenter, open-label, phase 3 study (HAVEN 1)
Objectives:
Emicizumab, a bispecific humanized monoclonal antibody in development to address unmet medical needs in persons with hemophilia A with inhibitors (PwHAwI), bridges FIXa and FX to replace the function of missing FVIIIa, needed for effective hemostasis. This study assessed efficacy, safety and PK of emicizumab prophylaxis in PwHAwI.
Methods:
Study NCT02622321 was conducted at 43 centers/sites, and enrolled PwHAwI ≥12 y.o. Participants (pts) receiving prior episodic bypassing agents (BPAs) were randomized (2:1) to emicizumab prophylaxis vs no prophylaxis (Arm A vs B). Primary endpoint compared treated bleed rates in Arm A vs B. PwHAwI receiving prior prophylactic BPA received emicizumab prophylaxis in Arm C. Emicizumab was injected subcutaneously at 3 mg/kg/wk for 4 wks, and 1.5 mg/kg/wk thereafter.
Summary:
109 male PwHAwI were enrolled; median age 28 (range 12–75) yrs. Median (range) emicizumab treatment exposure was 24.0 (3.0–47.9) wk overall and 29.5 (3.3–47.9) wk in Arm A. Statistically significant, clinically meaningful reductions (87%) in treated bleed rates were observed between emicizumab prophylaxis vs no prophylaxis (Arm A vs B) (annualized bleeding rate [95% confidence interval] 2.9 [1.69 to 5.02] versus 23.3 [12.33 to 43.89], P<0.0001), and in all secondary bleed-related endpoints (spontaneous, joint, target joint, and all bleeds). Of note, a 79% reduction in treated bleed rate was seen with emicizumab prophylaxis (Arm C) vs BPA prophylaxis prior to study entry in a non-interventional study (NCT02476942; intra-individual comparison, P=0.0003). Overall, 67.3% of PwHAwI on emicizumab prophylaxis had zero treated bleeds. Statistically significant, clinically meaningful improvements in health-related quality of life (HRQoL) and health status were seen for Arm A vs B. Emicizumab was well tolerated. Total of 198 adverse events (AEs) were reported in 103 pts; most common AEs were injection-site reactions (15%), and 12 serious AEs were reported in 9 (8.7%) pts. Thrombotic microangiopathy and thrombosis (2 pts each in primary analysis) were reported and associated with high cumulative aPCC doses averaging >100 U/kg daily for >24 hr prior to event onset. No events occurred with emicizumab prophylaxis alone. Both TMA events resolved once aPCC treatment was stopped, and the thrombotic events did not require anticoagulation; 2 pts resumed emicizumab without sequelae (1 with TMA, 1 with thrombosis). No antidrug antibodies were detected. Mean trough emicizumab concentrations >50 μg/mL were achieved after 4 loading doses (3 mg/kg/wk) and sustained with maintenance doses of 1.5 mg/kg/wk.
Conclusion:
Emicizumab prophylaxis prevented or substantially reduced bleeds in PwHAwI and meaningfully improved HRQoL. Emicizumab had acceptable safety without excess thrombotic risk in the absence of concomitant aPCC. PK levels were sustained with once- weekly maintenance doses. These promising data could support a paradigm shift in the management and lives of PwHAwI.
Estimating the prevalence of symptomatic, undiagnosed von Willebrand disease: analysis of medical insurance claims data
Objectives:
Von Willebrand disease (VWD) is a common inherited bleeding disorder, but awareness among healthcare professionals is low. Timely and proper diagnosis integral for reducing VWD burden, access to proper therapies, and avoidance of improper medication. Hence, we sought to estimate the prevalence of undiagnosed VWD among commercially insured patients in the United States with a recent history of bleeding events.
Methods:
Patients with a VWD diagnosis who were users of or candidates for von Willebrand factor were identified from the IMS PharMetrics Plus Database (2006─2015). We constructed a unary patient-finding model based on 12 prediagnosis variables that best defined this population, and applied to a set of undiagnosed patients with recent bleeding events from the same database. ‘Best fit’ (confidence level 5/6) and ‘good fit’ (confidence level 3/4) patients were identified. Prevalence of symptomatic undiagnosed VWD in the commercially insured population was estimated from the best-fit and good-fit population size (projection factor 10.4).
Summary:
Overall, 507 668 undiagnosed patients with recent bleeding events were identified (86% female, 14% male). Application of the VWD model identified 3318 best-fit and 37 163 good-fit patients; 91% of best-fit patients were females aged <46 years, with heavy menstrual bleeding the most common claim. Projection to the full commercially insured US population provided an estimate of 35 000 - 387 000 symptomatic, undiagnosed patients with VWD.
Conclusion:
There is a high prevalence of symptomatic, undiagnosed VWD (undiagnosed bleeding disorder patients that likely have VWD) in the commercially insured population. This data underscores the importance of improved disease education to both patients and the first line treaters, including OBGYN, emergency room, and pediatricians. Enhanced awareness of VWD symptoms and their impact, and of screening and testing procedures, may improve diagnosis of VWD and reduce the disease burden.
Physician practice patterns in the US show significant variation in how PK parameters are currently used to personalize care for US hemophilia A patients
Introduction & Objective:
Standard approaches to prophylaxis may be further improved by taking into account individuals’ pharmacokinetic (PK) profile, thereby increasing the likelihood of therapeutic success [Valentino 2012, Valentino 2014, Lissitckov 2017]. One study demonstrated an association between more time spent above higher FVIII levels (20 and 30%) and lower bleeding rates [Valentino 2016]. Furthermore, a consensus statement proposes to target specific factor levels to tailor treatment for different patient profiles [Iorio 2017]. Additionally, new extended half-life treatments provide physicians with another option to personalize therapy. As more data and therapeutic options become available, it is important to understand physicians’ current perceptions and practice patterns with respect to personalizing haemophilia A care in the US.
Materials & Methods:
Physicians in the US who treat persons with severe hemophilia A and provided informed consent were eligible to complete a cross-sectional, double-blinded web-based survey to evaluate physicians’ perceptions and practice patterns with respect to measuring PK and if/how they personalize FVIII treatment with this information, when and to what extent. This abstract presents results from the first half of the survey which focused on ways in which physicians measure FVIII pharmacokinetics and personalize care.
Results:
Ninety physicians completed the survey. The top three most important considerations for personalizing therapy in general were bleeding history, patient goals, and physical activity. The most commonly cited reason for conducting a PK assessment was product switch (74%) while the most common barrier was patient willingness/availability (60%). Physicians reported using a PK-based approach to personalize treatment in 25% (median) of their severe patients. Of physicians who use PK, trough levels (91%), half-life (58%), peak levels (56%) and Area Under the Curve (22%) were used. While 12% of these physicians reported using all PK parameters, 18% only used trough levels. 23% reported using peak, trough and half-life in combination. Most physicians (89%) indicated using PK data to adjust dose and 50% also used it as a patient education opportunity.
Conclusions:
There was significant variability across respondents as to how PK is assessed and how PK parameters are used in treatment decisions, suggesting an opportunity to increase awareness and use of PK-guided personalization to ultimately improve patient care. Additional education on the definitions and details of PK-guided dosing could help improve overall adoption of this treatment strategy and align the community on what it means to personalize therapy using PK information. More research studying the association between PK-guided prophylaxis and outcomes is encouraged to better understand how best to personalize PK-based prophylaxis for different patient scenarios.
An Integrated Safety and Efficacy Analysis of Sofosbuvir-Based Regimens in Patients with Hereditary Bleeding Disorders
Objective:
Patients with hereditary bleeding disorders have been included in Phase 2 and 3 clinical trials of sofosbuvir (SOF), ledipasvir/sofosbuvir (LDV/SOF), sofosbuvir/velpatasvir (SOF/VEL), and sofosbuvir/velpatasvir/voxilaprevir (SOF/VEL/VOX) as well as in a dedicated study (n = 120) in this patient population. This integrated analysis evaluates the safety and efficacy of SOF-based regimens in HCV-infected patients with hereditary bleeding disorders.
Methods:
HCV-infected patients with a medical history of a hereditary bleeding disorder who participated in a SOF-based Phase 2 or 3 study were included in this pooled analysis. Medical history term(s) used to identify patients with bleeding disorders included variations of Hemophilia A or B, Von Willebrand’s Disease, Factor Deficiencies, or conditions associated with hemophilia.
Summary:
A total of 201 patients (74 GT1, 10% GT2, 14% GT3, 2% GT4, <1% GT5) with bleeding disorders were identified across 19 studies. The majority were male (91%), Caucasian (82%), IL28B non-CC (70%), HCV treatment-naïve (55%), and without cirrhosis (74%). Hemophilia A (65%) and B (26%) were the most common bleeding disorders. SVR12 results are shown in the below table by treatment regimen and genotype. The most frequently reported adverse events (>10%) were fatigue and headache; majority were mild or moderate in severity. One patient (<1%) discontinued LDV/SOF due to an adverse event and 11 patients (5%) experienced a serious adverse event. Hemarthrosis, muscle hemorrhage, epistaxis, hematoma, and hematuria were the only hemorrhagic events that occurred in >1 patient. Grade 3 or 4 laboratory abnormalities were infrequent with anemia and hyperbilirubinemia the most frequent Grade 3 laboratory abnormality consistent with RBV administration.
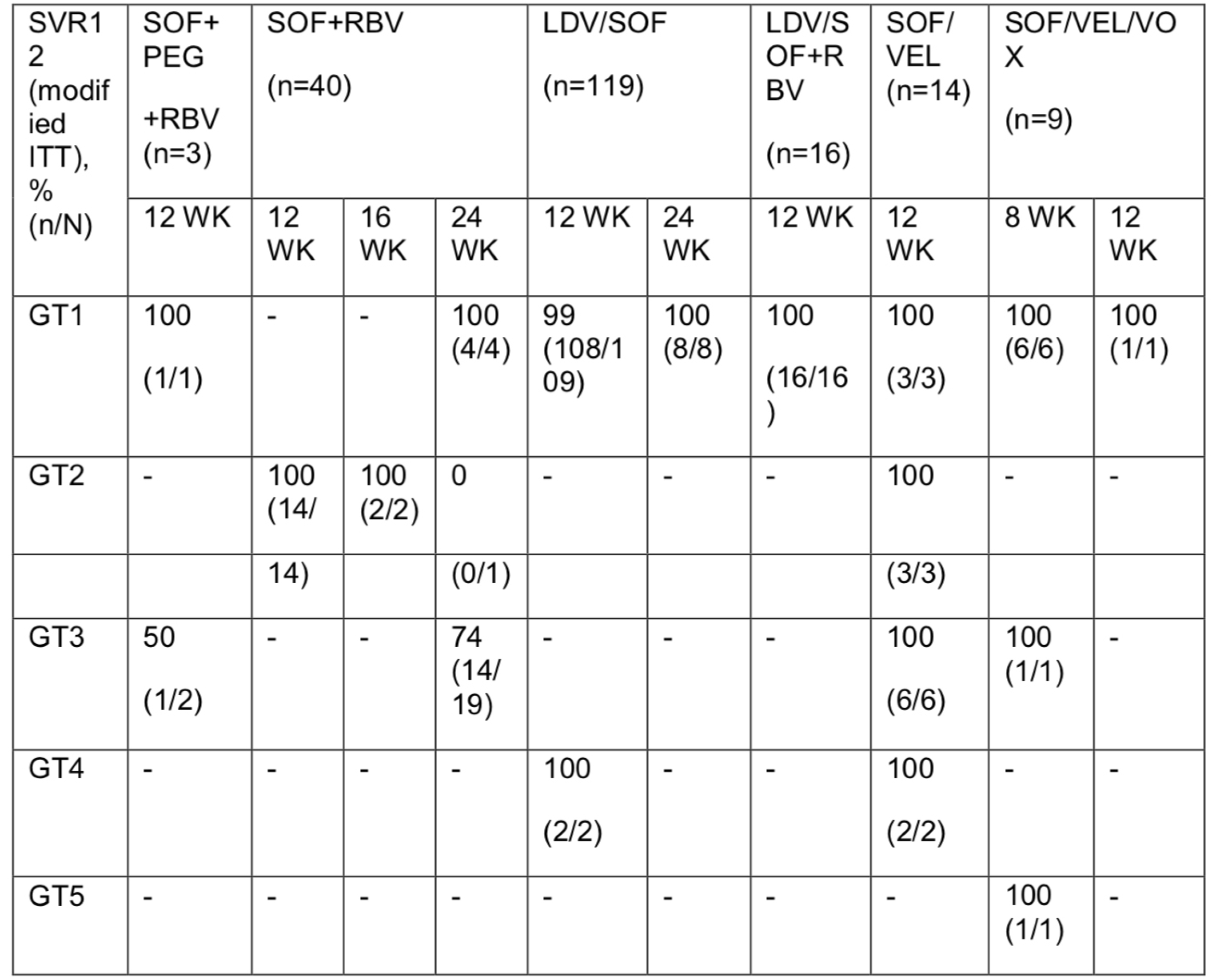
Conclusions:
SOF-based regimens led to high rates of SVR in genotype 1–5 HCV infected patients with bleeding disorders. SOF-based regimens were safe and well tolerated with no new toxicity specific to patients with bleeding disorders emerging
.
Efficacy, safety, and pharmacokinetics of a high-purity plasma-derived factor X (pdFX) concentrate in the prophylaxis of bleeding episodes in children <12 years with moderate to severe congenital factor X deficiency (FXD)
Background:
Congenital FXD is a rare bleeding disorder characterized by spontaneous joint and mucocutaneous bleeding and gastrointestinal or intracranial hemorrhage. pdFX is a US- and EU-approved treatment for congenital FXD, but data in children <12 years have been unavailable.
Aims:
To investigate pdFX efficacy, safety, and pharmacokinetics in children <12 years with moderate to severe congenital FXD.
Methods:
In this 6-month, open-label, multicenter, phase 3, prospective study in children <12 years, all subjects had a confirmed diagnosis of moderate to severe congenital FXD (basal FX:C <5%), severe bleeding history, or an F10 gene mutation causing a documented severe bleeding type. Subjects received routine prophylaxis at recommended 40–50 IU/kg twice weekly to maintain trough FX:C levels ≥5%. Each investigator assessed efficacy based on standardized criteria and presence of breakthrough bleeding. All subjects provided informed consent and the protocol was approved by appropriate independent ethics committees.
Results:
Mean age of the 9 trial completers was 6.8 years. Eight subjects had severe and 1 had moderate FXD. Overall, 537 prophylactic infusions were administered; mean dose/child was 38.6 IU/kg. Ten bleeds in 3 of 9 children were reported: 6 minor, 3 major, 1 unassessed. Investigators rated overall pdFX efficacy as excellent in all subjects. Overall mean incremental recovery was 1.74 IU/dL per IU/kg. FX trough levels were maintained >5% after visit 4 (days 29–42) in all subjects.
A total of 28 treatment-emergent adverse events (TEAEs) were reported in 8 children; none were considered pdFX related. No significant changes were noted in vital signs, physical exams, or laboratory measurements. No evidence of inhibitor development was seen.
Conclusion:
pdFX is efficacious in the prophylaxis of bleeding episodes in subjects <12 years with moderate to severe FXD. Safety profile in this population is consistent with previous results in subjects ≥12 years.
Funding: Bio Products Laboratory
Hereditary factor X (FX) deficiency in women and girls: treatment with a high purity plasma-derived factor X concentrate
Background:
A high-purity plasma-derived FX concentrate (pdFX) has been developed for treatment of hereditary FX deficiency, an autosomal recessive disorder.
Aim:
This post hoc analysis describes the pharmacokinetics, safety, and efficacy of pdFX in 10 women and girls with hereditary FX deficiency.
Methods:
In this open-label study, subjects (10 women/girls, 6 men/boys) aged ≥12 years with moderate or severe FX deficiency (basal plasma FX activity ≤5 IU/dL) were enrolled and received 25 IU/kg pdFX for on-demand treatment of bleeding episodes or preventative use for up to 2 years. All subjects provided informed consent and the protocol was approved by appropriate independent ethics committees.
Results:
Nine women and girls had severe and 1 had moderate FX deficiency, were aged 25.5 (median; range 14–58) y, and received a total of 267 pdFX infusions (178 for on-demand and 89 for preventative treatment). Men and boys (5 severe and 1 moderate FX deficiency) received a total of 159 pdFX infusions (64 on-demand; 95 preventative). The mean number of infusions per subject per month was higher among women and girls (2.48) than males (1.62). The mean pdFX incremental recovery was similar between women/girls and men/boys (2.05 vs 1.91 IU/dL per IU/kg, respectively), as was mean half-life (29.3 and 29.5 h, respectively). Among women and girls, 132 assessable bleeding episodes (61 heavy menstrual bleeding, 47 joint, 15 muscle, and 9 other) were treated with pdFX. Women and girls reported a treatment success rate (ie, subject rating of “excellent” or “good” response to pdFX) of 98%, comparable to the 100% treatment success rate among men and boys. After study completion, 2 subjects received pdFX for hemostatic cover during obstetric delivery. Additional infusion, bleed, and safety data will be presented.
Conclusion:
These results show that, in women and girls with moderate or severe hereditary FX deficiency, who experience reproductive tract and other bleeding events, pdFX was safe and effective. The pharmacokinetic profile of pdFX in women and girls was similar to that of men and boys.
Funding: Bio Products Laboratory
Updated results from a dose-escalation study in adults with severe or moderate-severe hemophilia B treated with AMT-060 (AAV5-hFIX) gene therapy: up to 1.5 years follow-up
Background:
Gene transfer for hemophilia offers the potential to convert the disease from a severe to mild phenotype with a single treatment. AMT-060 consists of an AAV5 vector containing a codon-optimized wildtype hFIX gene under control of a liver-specific promoter.
Objective:
This phase 1/2 study investigates the safety and efficacy of AMT-060 at 2 dose levels in adults with moderate-severe or severe hemophilia B.
Methods:
Multi-national, open-label, dose-escalating study in patients with factor IX (FIX) activity ≤2% of normal, and a severe bleeding phenotype (prophylactic exogenous FIX; or ondemand exogenous FIX, plus ≥4 bleeds/year or hemophilic arthropathy). Patients received either 5x1012 gc/kg (n=5) or 2×1013 gc/kg (n=5) of AMT-060 iv. Efficacy assessments include endogenous FIX activity (measured ≥10 days after use of exogenous FIX); reduction of exogenous FIX use; and annualized spontaneous bleeding rates. Safety assessments include treatment related adverse events, immunological and inflammatory biomarkers.
Summary:
There were no screening failures due to AAV5 neutralizing antibodies. Mean FIX activity in the lower dose cohort was 5.2% (min-max, 3.0-6.8%; n=4; 1 patient remaining on prophylaxis excluded) during 1 year of follow-up, and 6.9% (min-max, 3.1-12.7%; n=5) in the higher dose cohort during 26 weeks follow-up. Eight of 9 patients on FIX prophylaxis discontinued use after AMT-060 infusion. Follow-up to up to 1.5 years will be presented, with annualized reduction of exogenous FIX use and spontaneous bleeding rates. Mild, temporary elevations in ALT were observed in 3 patients with higher mean FIX activity (6.3-12.7%; 1 in the lower and 2 in the higher dose cohort). Each received a tapering course of prednisolone. ALT elevations were not associated with changes in FIX activity or a capsid-specific T-cell response.
Conclusions:
Patients continue to show sustained clinical benefit and endogenous FIX activity with no T-cell activation ≥1 year after a single infusion of AMT-060.
An Evaluation of the Switch from Standard Factor VIII Prophylaxis to Prophylaxis with an Extended Half-Life, Pegylated, Full-length Recombinant Factor VIII (BAX 855)
Objective:
This report assesses the changes in bleeding patterns as previously treated severe hemophilia A patients were switched from their pre-study standard factor VIII (FVIII) prophylactic treatment regimen to prophylaxis with BAX 855 - an extended half-life, pegylated, full-length recombinant FVIII built on ADVATE - during their participation in the pivotal trial.
Methods:
Patients’ informed consent and appropriate ethics committee approvals were obtained. At baseline, eligible patients reported their pre-study treatment regimen and average annualized bleeding rate (ABR) for the previous 3-6 months. Patients assigned to the prophylactic arm were to receive 45±5 IU/kg of BAX 855 twice weekly for ≥50 exposure days or approximately 6 months. This per-protocol analysis included 101 treated patients in the prophylactic arm.
Summary:
The overall mean (SD) ABR for these treated patients was 3.7 (4.7), which was lower than the ABR for the 17 patients treated on-demand during the study (40.8 [16.3] - who were all treated on-demand pre-study). Of the 101 subjects in BAX 855 prophylactic arm, 82 were treated on prophylaxis and 19 were treated on-demand during the pre-study period. The BAX 855 prophylactic dosing frequency was reduced by a mean (SD) of 26.7% (27.9) compared to the frequency reported in the pre-study period, which is approximately equivalent to one less prophylactic infusion per week when using BAX 855 for prophylaxis. The mean dose per prophylactic infusion was higher for the BAX 855 treatment regimen compared to pre-study (44.53 vs 38.12 IU/kg), but FVIII consumption per week was lower during the study compared to pre-study (83.96 vs 97.79 IU/kg/week). More patients treated on BAX 855 prophylaxis during the study treatment period had zero bleeding episodes compared to during their pre-study period: 37.8% vs 23.2%. The mean ABR was lower for subjects on BAX 855 prophylaxis compared to pre-study: 4.13 vs 9.74.
As expected, the 19 patients treated on-demand pre-study had a higher mean ABR (31.53) and all experienced bleeding (ie, none had zero hemarthroses during the pre-study period) compared to during their BAX 855 prophylactic treatment period (mean ABR of 1.68 and 47.4% had zero bleeding episodes).
Conclusions:
These results further demonstrate the benefit of BAX 855 prophylaxis (45±5 IU/kg twice weekly) and support its efficacy profile for the prevention of bleeding when used twice weekly, which suggests that fewer infusions may be needed to maintain prophylactic efficacy.
Low Annualized Bleeding Rates in Phase 3 Studies of Recombinant Factor VIII Fc Fusion Protein (rFVIIIFc) in Subjects with Target Joints at Baseline
Introduction and Objectives:
The phase 3 A-LONG and Kids A-LONG studies demonstrated the safety and efficacy of rFVIIIFc for the control and prevention of bleeding episodes in subjects with severe hemophilia A. This subgroup analysis of A-LONG and Kids A-LONG examined bleeding frequency with rFVIIIFc prophylaxis in subjects with target joints at baseline.
Methods:
A-LONG (≥12 y) and Kids A-LONG (<12 y) subjects with a history of target joints (a major joint with ≥3 bleeding episodes in a 6-month period) who were previously treated with FVIII and received on-study rFVIIIFc prophylaxis were identified. Self-reported pre-study 12- month bleeding history and on-study annualized bleeding rate (ABR) for all bleeds were evaluated.
Results:
93/141 subjects in A-LONG and 13/69 subjects in Kids A-LONG had target joints at baseline. The most prevalent locations of pre-study target joints identified at baseline among subjects in A-LONG were the elbow (61.7%) and ankle (59.6%). Similarly, in Kids A-LONG, these were the ankle (69.2%) and elbow (30.8%). A total of 59.7% and 52.4% of subjects receiving individualized (Arm 1) and weekly (Arm 2) rFVIIIFc prophylaxis in A-LONG, respectively, and 53.8% of subjects receiving rFVIIIFc prophylaxis in Kids A-LONG with target joints at baseline had no target joint bleeding episodes on-study. Median on-study ABRs with rFVIIIFc prophylaxis tended to be lower than pre-study bleeding rates in subjects with target joints at baseline (Table).
Conclusions:
For subjects with severe hemophilia A who had target joints at baseline, rFVIIIFc prophylaxis lowered bleeding rates across age groups compared with pre-study FVIII treatment.
Efficacy of a Recombinant Factor IX Investigational Product, IB1001 (trenonacog alfa) for Perioperative Management in Hemophilia B Patients
Objective:
To evaluate efficacy of IB1001 for perioperative management of bleeding under surgical circumstances in hemophilia B patients.
Methods:
The efficacy of IB1001 for perioperative management has been evaluated in a prospective, open-label, multicenter international study where 17 subjects (16 male PTPs and one female hemophilia B carrier) underwent 20 major surgical procedures. The PTPs were defined as patients with a minimum of 150 exposures to another factor IX preparation. The subjects had severe or moderately severe (factor IX level ≤ 2 IU/dL) hemophilia B without inhibitors, with exception of one mild hemophilia B female carrier. Efficacy of IB1001 was based on the surgeon’s assessment including estimation of blood loss as ‘less than expected’, ‘expected’, or ‘more than expected’ at the time of surgery and assessment of hemostasis at 12 and 24 hours post-surgery as ‘superior’, ‘adequate’, or ‘poorly controlled’. Transfusion requirements were also monitored.
Summary:
Thirteen major procedures in 12 male subjects were managed by bolus regimen, and 6 major procedures in 4 male subjects were managed by continuous infusion regimen. Mean loading dose for 13 major procedures managed by bolus regimen was 120 ± 11.4 IU/kg and mean maintenance bolus dose (given every 12 hours) was 59.7 ± 12.2 IU/kg. During the 24 hours following surgery, factor IX levels were successfully maintained over 60%, as intended. Factor IX levels at pre-infusion were 59.7% ± 15.9% at 12 hours after surgery and 54.4% ± 16.5% at 24 hours after surgery. For a major procedure in one female carrier, the bolus dose was 110 IU/kg, while the mean maintenance dose was 44.9 ± 7.0 IU/kg. Mean loading dose for 6 major procedures managed by continuous infusion regimen was 95.4 ± 14.5 IU/kg and the mean maintenance infusions were 7.1 ± 4.0 IU/kg/hr. In all major procedures, blood loss at the time of surgery was ‘expected’ or ‘less than expected’ as assessed by the surgeon. IB1001 was rated by the surgeon as ‘superior’ or ‘adequate’ in controlling hemostasis post-surgery, including 8 knee arthroplasties, two elbow arthroplasties, one knee amputation, one percutaneous Achilles tendon lengthening, one open inguinal hernia repair, one tibiotalar fusion, two arthroscopic synovectomies, three debridements and one total hysterectomy/bilateral oopherectomy. There were no transfusions required perioperatively.
Conclusions:
At the time of surgery, blood loss was expected or less than expected after IB1001 treatment, while post-surgery effective hemostasis control was achieved following IB1001 treatment in hemophilia B patients.
Efficacy of a Recombinant Factor IX Investigational Product, IB1001 (trenonacog alfa) in Previously Treated Patients with Hemophilia B
Objective:
To evaluate efficacy of IB1001 with respect to breakthrough bleeding during prophylaxis and with respect to control of hemorrhaging in prophylaxis and on-demand treatment regimens in previously treated patients (PTPs) with hemophilia B.
Methods:
The efficacy of IB1001 has been evaluated in a prospective, open-label, uncontrolled multicenter international study in which a total of 68 male PTPs between 7 and 64 years of age received IB1001 either as prophylaxis or on-demand treatment. All subjects had severe or moderately severe (factor IX level ≤ 2 IU/dL) hemophilia B without inhibitors. There were 61 subjects who received prophylaxis [predominantly secondary or tertiary prophylaxis as defined by World Federation of Hemophilia (WFH) guidelines] and 12 were treated on-demand during the study conduct. Both, the subjects and the study investigators rated the efficacy of IB1001 in management of bleeding episodes.
Summary:
The mean number of exposure days (ED) was 138.2 (median: 127.5 ED), including 55 subjects with ≥ 50 ED and 45 subjects with ≥ 100 ED. Subjects on prophylaxis received a mean intravenous dose of 55.0 ± 12.8 IU/kg IB1001 twice weekly, while subjects in the on- demand regimen received a mean dose of 60.0 ± 18.2 IU/kg IB1001. In the prophylaxis group, median annualized bleed rate (ABR) was 1.52, and in the on-demand group, median ABR was 16.39. There were 19/61 (31.1%) subjects on IB1001 prophylaxis who reported no bleeding episodes. A total of 508 bleeding episodes were treated with IB1001; 286 bleeds were recorded for patients on prophylaxis and 222 in the on-demand regimen. Majority of the bleeds (70.9%) resolved after a single IB1001 infusion, while 13% of the bleeds required two infusions. A minority of the bleeds (4.7%) required 5 or more infusions; these bleeds were predominantly related to trauma, target joints and/or muscle bleeds. Subjects rated efficacy of IB1001 as ‘excellent’ or ‘good’ in 84% of all treated bleeding episodes. Of 414 subject visits where the efficacy of IB1001 was evaluated by the study investigators, 92% were rated as ‘effective’ in prevention and treatment of bleeding by IB1001.
Conclusions:
IB1001 is effective for the treatment of hemophilia B either as secondary or tertiary prophylaxis or on an episodic (on-demand) basis. Based on clinical trial data, IB1001 is effective in controlling breakthrough or episodic bleeding episodes.
Recombinant von Willebrand factor in severe von Willebrand disease: a prospective clinical trial
Objectives:
This trial evaluated the hemostatic efficacy, pharmacokinetics, and safety of a recombinant VWF (rVWF) in adults with severe von Willebrand disease (VWD) (type 3 [VWF:Ag ≤ 3 IU/dL], severe type 1 [VWF:RCo< 20 IU/dL], 2A [VWF:RCo< 20 IU/dL], 2N [FVIII:C<10% ], 2B, or 2M).
Methods:
Bleeds were to be treated initially with rVWF and rFVIII (1.3:1 ratio), followed by rVWF alone (as long as FVIII:C >40%). Hemostatic efficacy was evaluated using a pre- defined 4-point rating scale (none=4, moderate=3, good=2, excellent=1). PK parameters for rVWF vs. rVWF:rFVIII were assessed in a randomized crossover design, and a 6-month repeated PK evaluation for rVWF alone.
Summary:
Of 31 subjects assigned to bleed treatment, 22 (17 type 3, 4 type 2A and 1 type 2N) experienced 192 bleeding episodes (several in multiple locations) that were treated with rVWF: 106 occurred in mucosal tissue (including 32 menorrhagic, 42 nasopharyngeal, 26 mouth/oral bleeds), 59 joint bleeds, 6 gastrointestinal bleeds, and 37 in other locations. Treatment success (mean efficacy rating <2.5) was achieved in all 22 subjects (100%; Clopper-Pearson exact 90% confidence interval: 87.3 to 100.0). ‘Excellent’ ratings were given for 186/192 (96.9%) bleeds (119/122 minor, 59/61 moderate, 6/7 severe, 2/2 unknown severity) and the remaining 3.1% were ‘good’. One infusion was effective in 81.8% of bleeds (median [range]: 1 [1-4] overall; 2 [1-3] for severe bleeds). The subject’s own assessment of treatment efficacy (an exploratory endpoint), was ‘excellent’ within 8 hours after the first infusion for 125/134 (93.3%), ‘good’ for 8/134 (6.0%) and ‘moderate’ for 1/134 (0.7%) bleeding episodes. A substantial increase in FVIII:C and sustained stabilization (> 40% by 6 hours, rising to >80% 24 hours post-infusion) was observed after infusion with rVWF. PK parameters for VWF Ristocetin cofactor (VWF:RCo, a surrogate for the platelet-dependent function of rVWF) were similar when rVWF was infused alone (mean T1/2 21.9 h vs. 19.6 h with rVWF:rFVIII). Eight adverse events (tachycardia, infusion site paraesthesia, ECG t wave inversion, dysgeusia, generalized pruritis, hot flush, chest discomfort and increased heart rate) in 5 subjects were assessed as related to rVWF. No inhibitor or anti-VWF binding antibody development was observed, and there were no thrombotic events or severe allergic reactions.
Conclusions:
rVWF was safe, well-tolerated and effective in the treatment of a variety of bleeding presentations in severe VWD. The sustained stabilization in FVIII:C after the initial infusion enables subsequent infusion of rVWF without rFVIII, when multiple infusions are required.
Kids B-LONG: Safety, Efficacy, and Pharmacokinetics of Long-Acting Recombinant Factor IX Fc Fusion Protein (rFIXFc) in Previously Treated Children with Hemophilia B
Objective:
Kids B-LONG was an open-label phase 3 study that evaluated the safety, efficacy, and pharmacokinetics (PK) of rFIXFc in previously treated children (aged <12 years; ≥50 prior exposure days [EDs] to FIX) with severe hemophilia B (≤2 IU/dL endogenous FIX) and no history of FIX inhibitors.
Methods:
Participants initiated prophylactic treatment with 50–60 IU/kg rFIXFc once-weekly; dose and interval adjustments were based upon PK data and bleeding frequency. The primary endpoint was development of inhibitors (neutralizing antibodies). Key secondary outcomes included PK and annualized bleeding rate (ABR).
Summary:
The study enrolled 30 participants (<6 years of age, n=15; 6 to <12 years of age, n=15); 90% completed the study. Prestudy, all participants received FIX prophylaxis (23/30 were dosing ≥2x/week). The median time on study was 49.4 weeks; 24 participants had ≥50 rFIXFc EDs. No participant developed inhibitors to rFIXFc. The pattern of adverse events reported was typical of the population studied. There were no serious allergic reactions and no thrombotic events. No serious adverse events were assessed by the investigator as related to rFIXFc. The terminal half-life (geometric mean [95% CI]) of rFIXFc was 66.5 (55.9, 79.1) hours in the <6 years cohort (n=11) and 70.3 (61.0, 81.2) hours in the 6 to <12 years cohort (n=13). The geometric mean (95% CI) half-life of prestudy BeneFIX was 18.2 (15.5– 21.3) hours in the <6 years cohort (n=11) and 19.2 (17.6–20.9) hours in the 6 to <12 years cohort (n=9). Median (IQR) ABR was 1.97 (0.00, 3.13) overall, and 0.00 (0.00, 1.16) for spontaneous bleeds; 33.3% of participants reported no bleeds on study. At study end, 97% of participants were dosing once weekly. The median (IQR) total weekly prophylactic dose with rFIXFc was 59.4 (53.0, 64.8) IU/kg and 57.8 (51. 7, 65.0) IU/kg, in the <6 years and 6 to <12 years cohorts, respectively. The prestudy FIX median (IQR) total weekly prophylactic dose was 110.0 (58.0, 188.0) IU/kg and 100.0 (58.0, 120.0) IU/kg in the <6 years and 6 to <12 years cohorts, respectively. 75.0% of bleeding episodes were controlled with 1 infusion; 91.7% with 1 or 2 infusions (median average dose per infusion: 68.22 IU/kg).
Conclusions:
rFIXFc was safe and effective for the prevention and treatment of bleeding in children with severe hemophilia B. Study participants achieved low bleeding rates with extended-interval rFIXFc prophylaxis, while reducing their weekly prophylactic factor consumption compared with their prior FIX regimen.
Pegylated full-length recombinant factor VIII (BAX 855) for prophylaxis in previously treated adolescent and adult patients with severe hemophilia A
Objective:
To assess the pharmacokinetics (PK) and efficacy of prophylactic treatment with BAX 855 - a novel polyethylene glycol (peg)ylated full-length recombinant factor VIII, built on the rAHF-PFM (ADVATE) protein - by age group in previously-treated male patients with severe hemophilia A.
Methods:
Adolescent (12 to <18 years) and adult (18 to 65 years) subjects received 45 ± 5 IU/kg BAX 855 twice weekly as prophylaxis for approximately 6 months. PK was assessed in a subgroup (n=25 planned, including ≥6 adolescents) for one ADVATE infusion, then for BAX 855 at the initial infusion and after ≥50 exposure days (EDs). Efficacy was assessed in all subjects.
Summary:
Twenty-six subjects (8 adolescents, 18 adults) were included in the PK evaluation, and 121 (23 adolescents, 98 adults) were included in the efficacy analysis (all subjects assigned to treatment, i.e. full analysis set). The extended half-life (T1/2) and mean residence time (MRT) of BAX 855 (initial dose) compared to ADVATE were demonstrated by fold increases in the means of 1.4 and 1.5, respectively in both adolescents and in adults, using the one-stage clotting assay. The initial and repeat PK assessments of BAX 855 showed similar results. Consistent trends were observed when PK was determined using the chromogenic assay. The arithmetic mean (SD) annualized bleeding rate (ABR) during prophylaxis with BAX 855 was higher in adolescents than in adults (6.2 [6.1] versus 3.2 [4.2]). ABRs for injury-related bleeding episodes (BEs) were higher in adolescents (arithmetic mean [SD]:2.3 [3.2] versus 1.7 [3.1] in adults), thus contributing to the overall higher ABR in this group. Joint ABRs were lower in adolescents (arithmetic mean [SD]: 1.8 [2.5] versus 3.2 [8.8] in adults) with an inverse relationship for non-joint ABRs. Six adolescents (26.1%) and 39 adults (40.2%) had zero BEs. A total of 48 BEs occurred in adolescents (20 minor; 26 moderate; 2 severe); 182 occurred in adults (69 minor; 103 moderate; 10 severe). The hemostatic efficacy of BAX 855 was rated excellent or good at resolution for the majority of BEs in both age groups (93.8% in adolescents; 92.9% in adults).
Conclusions:
Although fewer adolescents than adults were included in the study, the data suggest that BAX 855 is efficacious in both age groups for twice-weekly prophylaxis and control of BEs. As expected, joint ABR was higher in the adult group.
Long-term safety and efficacy of recombinant factor VIII Fc (rFVIIIFc) for the treatment of severe hemophilia A: United States subgroup interim analysis of the ASPIRE study
Objective:
The ongoing rFVIIIFc extension study, ASPIRE (clinicaltrials.gov #NCT01454739), evaluates the long-term safety and efficacy of rFVIIIFc in adults, adolescents, and children with severe hemophilia A. Here we report interim outcomes for United States (US) subjects in ASPIRE.
Methods:
Eligible subjects could enroll in ASPIRE upon completing A-LONG or Kids A-LONG. There were 4 treatment groups: individualized prophylaxis (25-65 IU/kg every 3-5 days, or 20-65 IU/kg on D1, 40-65 IU/kg on D4 if twice weekly); weekly prophylaxis (65 IU/kg every 7 days); modified prophylaxis (to further personalize and optimize treatment when needed); or episodic treatment. Subjects could change treatment groups at any time. Subjects <12 yrs participated only in individualized and modified prophylaxis groups. Primary endpoint: development of inhibitors. Secondary outcomes included annualized bleeding rate (ABR) and rFVIIIFc exposure days (EDs).
Summary:
Sixty subjects (49 from A-LONG; 11 from Kids A-LONG) enrolled. As of the interim data cut (6 Jan 2014), the median time on ASPIRE was 80.37 (A-LONG) and 15.94 (Kids A-LONG) wks; 82% (A-LONG) and 27% (Kids A-LONG) of subjects had ≥100 cumulative rFVIIIFc EDs. 7/49 A-LONG subjects changed treatment groups upon enrollment into or during ASPIRE; 2 Kids A-LONG subjects switched from individualized to modified prophylaxis upon enrollment into ASPIRE. Median ABRs were low with rFVIIIFc prophylaxis (Table). Overall, most subjects treated prophylactically during the parent study did not experience changes to their total weekly prophylactic dose or dosing interval during ASPIRE. For subjects who enrolled from A-LONG and Kids A-LONG, 94% and 100% of all bleeding episodes during ASPIRE, respectively, were controlled with 1 infusion. In the overall study population, no inhibitors were observed during ASPIRE; adverse events were typical of the general adult and pediatric hemophilia A populations.
Conclusion:
Interim data from US subjects in ASPIRE are consistent with those of the phase 3 parent studies and the overall ASPIRE interim analysis. Results from ASPIRE confirm the longer-term safety of rFVIIIFc and the maintenance of a low ABR with extended interval prophylactic dosing in individuals with severe hemophilia A.
Increase or Maintenance of Physical Activity in Patients Treated with Recombinant Factor IX Fc Fusion Protein (rFIXFc) in the B-LONG and Kids B-LONG Studies
Introduction and Objectives:
In the phase 3 B-LONG and Kids B-LONG studies, subjects with severe hemophilia B receiving rFIXFc prophylaxis had low annualized bleeding rates (ABRs), with decreased weekly factor consumption and fewer infusions compared with pre- study FIX treatment. This report evaluated the effect of rFIXFc on subjects’ physical activity across a variety of age groups using a subject-reported assessment.
Methods:
Eligible subjects for B-LONG (≥12 y) and Kids B-LONG (<12 y) were previously treated males with severe hemophilia B (≤2 IU/dL endogenous FIX activity). Subjects in B- LONG were enrolled into 1 of 4 treatment arms: Arm 1, weekly prophylaxis; Arm 2, individualized interval prophylaxis; Arm 3, episodic treatment; or Arm 4, perioperative management (not included in this analysis). All subjects in Kids B-LONG started on weekly prophylaxis. There were no restrictions regarding physical activity. Physical activity assessments were conducted at Weeks 4, 16, 26, 39, 52, and end of study (B-LONG) and Weeks 3, 12, 24, 36, 50, and end of study (Kids B-LONG). At each visit after their first rFIXFc dose, subjects were asked to rate their activity level relative to their prior study visit as: more (or more intensive), fewer (or less intensive), or about the same amount of physical activities. To summarize each subject’s change in physical activity over the course of the study compared to baseline, subjects’ reports were classified into four groups: less, the same, more, or undetermined.
Results:
Overall, 123 and 30 subjects enrolled in B-LONG and Kids B-LONG, respectively. The majority of subjects in B-LONG reported more or the same amount of physical activity, and few subjects reported less physical activity during the study (less, the same, more, undetermined in Arm 1 [n=60], 7%, 42%, 35%, 17%; Arm 2 [n=25], 16%, 28%, 48%, 8%; Arm 3 [n=27], 15%, 26%, 30%, 30%, respectively). Results were generally similar for subjects in Kids B-LONG (for subjects aged <6 y [n=15], 13%, 27%, 47%, 13%; for subjects aged 6 to <12 y [n=15], 7%, 13%, 67%, 13%).
Conclusions:
ABRs were low in B-LONG and Kids B-LONG despite similar or increased physical activity levels reported by the majority of subjects. These results suggest that people with severe hemophilia B across a variety of age groups may maintain or increase their physical activity levels with rFIXFc, while also reducing infusion frequency and weekly factor consumption, without compromising efficacy.
Patients Treated with Recombinant Factor VIII Fc Fusion Protein (rFVIIIFc) Reported Increased or Maintained Physical Activity in the A-LONG and Kids A-LONG Studies
Introduction and Objectives:
In the phase 3 A-LONG and Kids A-LONG studies, subjects with severe hemophilia A receiving rFVIIIFc prophylaxis 1-2 times/week had low annualized bleeding rates (ABRs), with comparable pre-study and on-study weekly factor consumption for subjects previously on FVIII prophylaxis. This report evaluated the effect of rFVIIIFc on subjects’ physical activity across a variety of age groups using a subject-reported assessment.
Methods:
Subjects eligible for A-LONG (≥12 y) and Kids A-LONG (<12 y) were previously treated males with severe hemophilia A (<1 IU/dL endogenous FVIII activity). Subjects in A- LONG were enrolled into 1 of 3 arms: Arm 1, individualized prophylaxis; Arm 2, weekly prophylaxis; or Arm 3, episodic treatment. All subjects in Kids A-LONG received rFVIIIFc prophylaxis. There were no restrictions regarding physical activity. Physical activity assessments were conducted at Weeks 7, 14, 28, 38, 52, and end of study (A-LONG) and Weeks 2, 7, 12, 17, 22, 26, and end of study (Kids A-LONG). At each visit after their first rFVIIIFc dose, subjects were asked to report any changes in their activity levels relative to their prior study visit as: more (or more intensive), fewer (or less intensive), or about the same amount of physical activities. To summarize each subject’s change in physical activity during the study compared to baseline, subjects’ reports were classified into four groups: less, the same, more, or undetermined.
Results:
A total of 165 and 71 subjects enrolled in A-LONG and Kids A-LONG, respectively. Overall, the majority of subjects in A-LONG reported more or the same amount of physical activity, and few subjects reported less physical activity during the study (less, the same, more, undetermined in Arm 1 [n=117], 8%, 36%, 51%, 5%; Arm 2 [n=23], 9%, 48%, 39%, 4%; Arm 3 [n=23], 9%, 52%, 26%, 13%, respectively). Results were generally similar for subjects in Kids A-LONG (for subjects aged <6 y [n=35], 3%, 26%, 66%, 6%; for subjects aged 6 to <12 y [n=34], 9%, 26%, 56%, 9%).
Conclusions:
The majority of subjects in A-LONG and Kids A-LONG reported similar or increased physical activity levels during the studies, while maintaining low ABRs. These self- reported data suggest that subjects across a variety of age groups with severe hemophilia A who are transitioning to rFVIIIFc may maintain or increase physical activity levels, while reducing infusion frequency and maintaining similar weekly factor consumption, without compromising efficacy.
SPINART 3-Year Analyses: Patient- and Joint-Level Changes in Colorado Adult Joint Assessment Scale and Magnetic Resonance Imaging Scores With Bayer’s Sucrose-Formulated Recombinant Factor VIII (rFVIII-FS) in Adolescents and Adults
Introduction:
Efficacy and safety of routine prophylaxis vs on-demand treatment with Bayer’s sucrose-formulated recombinant factor VIII (rFVIII-FS) in patients with severe hemophilia A were evaluated in the randomized, controlled SPINART study.
Aim:
Patient- and joint-level changes at year 3 in magnetic resonance imaging (MRI) and Colorado Adult Joint Assessment Scale (CAJAS) scores were compared to investigate if individual joint data revealed results that may have been obscured in previously reported patient-level analyses.
Methods:
SPINART included males aged 12–50 years with severe hemophilia A, ≥150 exposure days to FVIII, no inhibitors, and no prophylaxis for >12 months in the past 5 years. Patients were randomized 1:1 to rFVIII-FS on demand or prophylaxis (25 IU/kg 3x/wk). Changes from baseline to year 3 were evaluated for 6 index joints (knees, ankles, elbows) using the Extended MRI (eMRI) scale and CAJAS. Percentages of patients or joints with improved, unchanged, or worsened scores were evaluated.
Summary:
Of the 84 patients in SPINART, eMRI and CAJAS change from baseline data were available for 62 (prophylaxis, n=32; on demand, n=30) and 76 patients (n=39; n=37) and for 386 (n=197; n=189) and 446 joints (n=224; n=222), respectively. Categoric analysis of CAJAS data at year 3 showed a higher percentage of patients treated prophylactically vs on demand with improved scores (64.1% vs 43.2%) and a lower percentage with worsened scores (28.2% vs 51.4%); with eMRI, the percentage improved was smaller (12.5% vs 6.7% improved; 75.0% vs 73.3% worsened). At individual joints, improved, unchanged, and worsened CAJAS scores in patients treated with prophylaxis vs on demand were 46.0% vs 33.3%, 22.3% vs 24.3%, and 31.7% vs 42.3%; eMRI values were 8.1% vs 3.7%, 61.9% vs 69.8%, and 29.9% vs 26.5%.
Conclusions:
These data suggest that in adults and adolescents with severe hemophilia A, joint function as measured by CAJAS is more likely to improve after 3 years of routine prophylaxis with rFVIII-FS than joint structure as measured by MRI.
Interim results of the B-YOND study evaluating long-term safety and efficacy of recombinant factor IX Fc (rFIXFc) in children with severe hemophilia B
Objective:
The ongoing rFIXFc extension study, B-YOND (clinicaltrials.gov #NCT01425723) , evaluates the long-term safety and efficacy of rFIXFc for the treatment of severe hemophilia B. Here we report interim safety and efficacy data for children aged <12 yrs enrolled in B- YOND.
Methods:
Upon completing Kids B-LONG, eligible subjects could enroll in one of the 3 prophylactic treatment groups in B-YOND: weekly (20 to 100 IU/kg every 7 days), individualized (100 IU/kg every 8 to 16 days, or twice monthly), or modified (a prophylaxis regimen different from weekly or individualized prophylaxis). Subjects could change treatment groups at any point in the study. The primary endpoint was development of inhibitors. Secondary outcomes included annualized bleeding rate (ABR) and rFIXFc exposure days (EDs).
Summary:
At the time of the interim data cut (17 October 2014), 23 subjects had completed Kids B-LONG; all enrolled in B-YOND (<6 yrs of age cohort, n=9; 6 to <12 yrs of age cohort, n=14). As of the interim data cut, 2 subjects had completed and 21 subjects continued in B-YOND (median time on study: 47.7 weeks). From the start of Kids B-LONG to the B-YOND interim data cut, the median time on rFIXFc was 95.3 weeks, with a median of 94 cumulative rFIXFc EDs. All subjects were on weekly prophylaxis in Kids B-LONG; 5 subjects changed treatment groups at the start of or during B-YOND. In the weekly prophylaxis group, the median (IQR) average weekly prophylactic dose was 64 (52, 66) IU/kg and 63 (59, 64) IU/kg in the <6 yrs and 6 to <12 yrs of age cohorts, respectively. The median (IQR) dosing interval among subjects on individualized prophylaxis was 10 (10, 11) days. As of the interim data cut, no inhibitors were observed, there were no reports of anaphylaxis or serious hypersensitivity reactions associated with rFIXFc, and no thrombotic events. Adverse events were typical of the pediatric hemophilia B population; no subject discontinued the study due to an adverse event. Median ABRs were low in both age cohorts (Table). Overall, 95% of bleeding episodes were controlled with 1 or 2 infusions.
Conclusions:
Interim data in children with severe hemophilia B participating in B-YOND confirm the long-term safety of rFIXFc and the maintenance of a low ABR with extended-interval prophylactic dosing.
Pediatric Venous Thrombosis Associated With Staphylococcal Infections: A Single Institutional Experience
Introduction:
Since the emergence of community-acquired methicillin-resistant S. aureus (MRSA), severe manifestations of infection are encountered more frequently. Venous thrombosis (VT) has been previously reported in children with S. aureus infections. This study reviews our institutional experience and outcomes of children with VT and staphylococcal infections.
Methods:
A retrospective analysis of 15 pediatric patients (≤ 18 years) treated for VT and staphylococcal infections at Children’s Memorial Hermann Hospital between January 1, 2010 and December 31, 2014 was performed.
Results:
Fifteen patients were included (10 males, 5 females) with a median age at diagnosis of 8 years (range 2 mo-17yrs). Underlying infections included: osteomyelitis (n=7), soft tissue infection (n=3), aortitis (n=1), meningitis (n=1), septic arthritis (n=1), central line infection (n=1), septicemia (n=1). Primary presentation included: swelling (n=12), pain (n=11), tenderness (n= 8), and color changes (n=3). One patient had prior history of VT. Family history was positive for VT in one patient. Isolated organisms included: MRSA (n=9), MSSA (n=3), polymicrobial (n=2), and Staphylococcus non- aureus (n=1). Eight patients had central venous catheters (CVC) and 5 of them had thrombosis at the CVC site. VT sites identified by Doppler US (DUS) included: upper extremity (n= 5), lower extremity (n= 8), and neck (n=2). All thrombophilia work up was negative. The median D-dimer at diagnosis was 3 ug/ml (range: 0.31- 6.54 ug/ml). Median time elapsed between infection and VT diagnosis was 5.5 days (range: 0-35 days). All patients received anticoagulation using LMWH (1mg/kg/dose) except one who had superficial thrombophlebitis and was managed conservatively. Median time to achieve therapeutic anti- factor Xa level was 2.5 days (range 1- 6 days). Median duration of anticoagulation was 3 months. Three out of 13 patients (23%) had resolution of thrombosis within one week of anticoagulation. Four other patients had thrombus resolution by DUS at 3-6 months. Five patients did not have DUS at 3-6 months. None of the 15 patients required hospital re- admission for bleeding or thrombotic complications.
Discussion:
Staphylococcal infections may increase the risk of VT in children. Nine out of 15 patients (60%) with VT had a documented MRSA infection, which appears to confer an even higher risk for development of VT. Over half of the patients responded favorably to anticoagulation with resolution of VT within 6 months.
Conclusions:
A high index of suspicion for VT is warranted in children with Staphyloccoccal infections (particularly MRSA) to promptly diagnose and treat. This approach may improve outcomes and minimize complications including septic emboli and post-thrombotic syndrome. Prophylactic anticoagulation in presence of MRSA infection could be considered in future studies.
Keywords: Children, staphylococcal infection, venous thrombosis

