Clinical trials are one of the most important parts of the research process to find new ways to treat diseases. But this critical work is not possible without people like you.
Understanding clinical trials
During clinical trials, new or improved treatments are tested to find out if they can effectively treat a condition, or if they might work better than the treatments that are already available.
There are two main types of research trials:
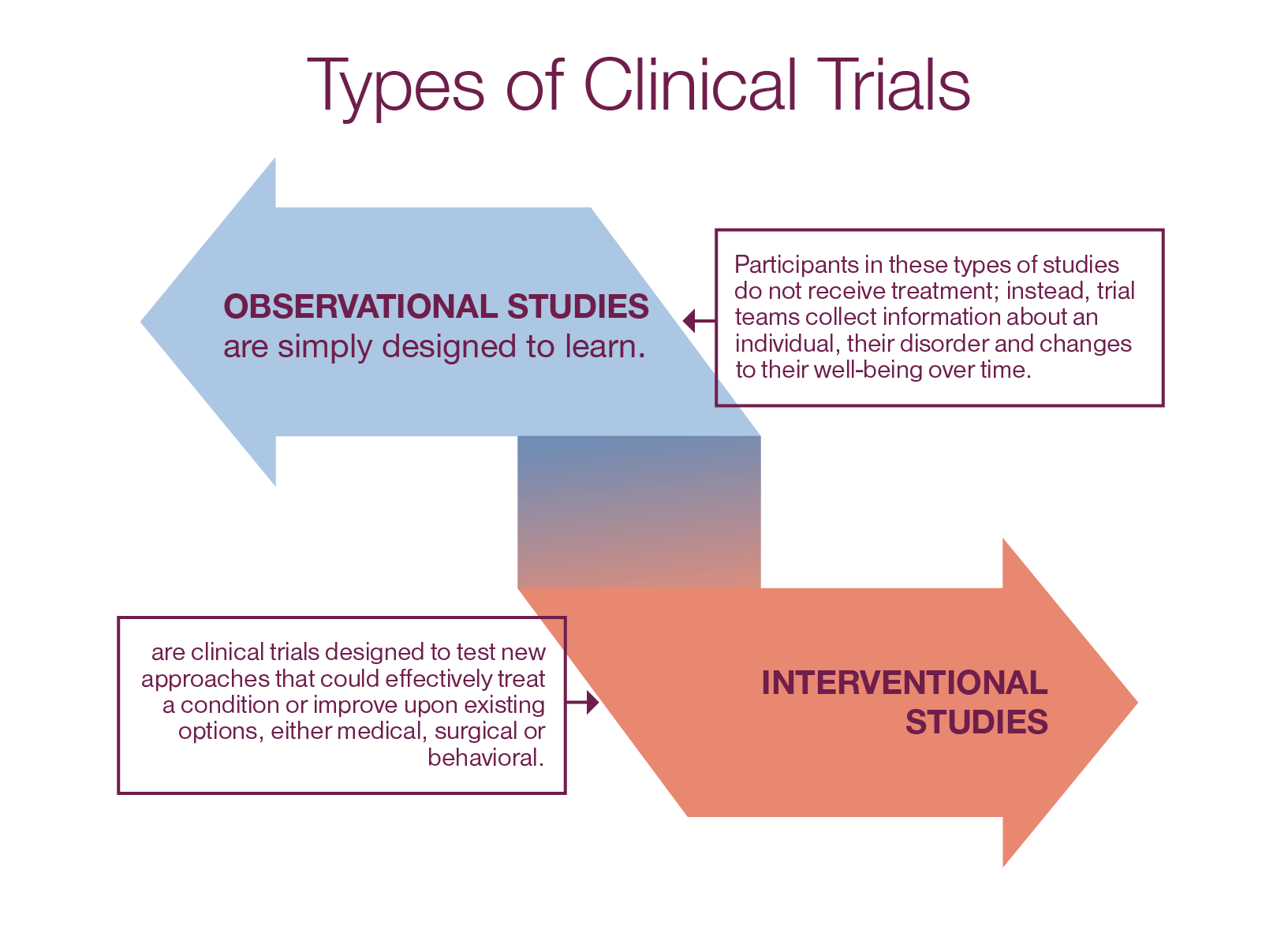
Clinical trials are often sponsored by companies, research institutions or collaborations (referred to in this kit as the “trial sponsor”). These trials are needed before a medicine can receive regulatory approval by the U.S. Food and Drug Administration (FDA) and become available for prescription..
Have you heard about trial “phases”?
New treatments or approaches are studied through different “phases” of clinical trials. These include:
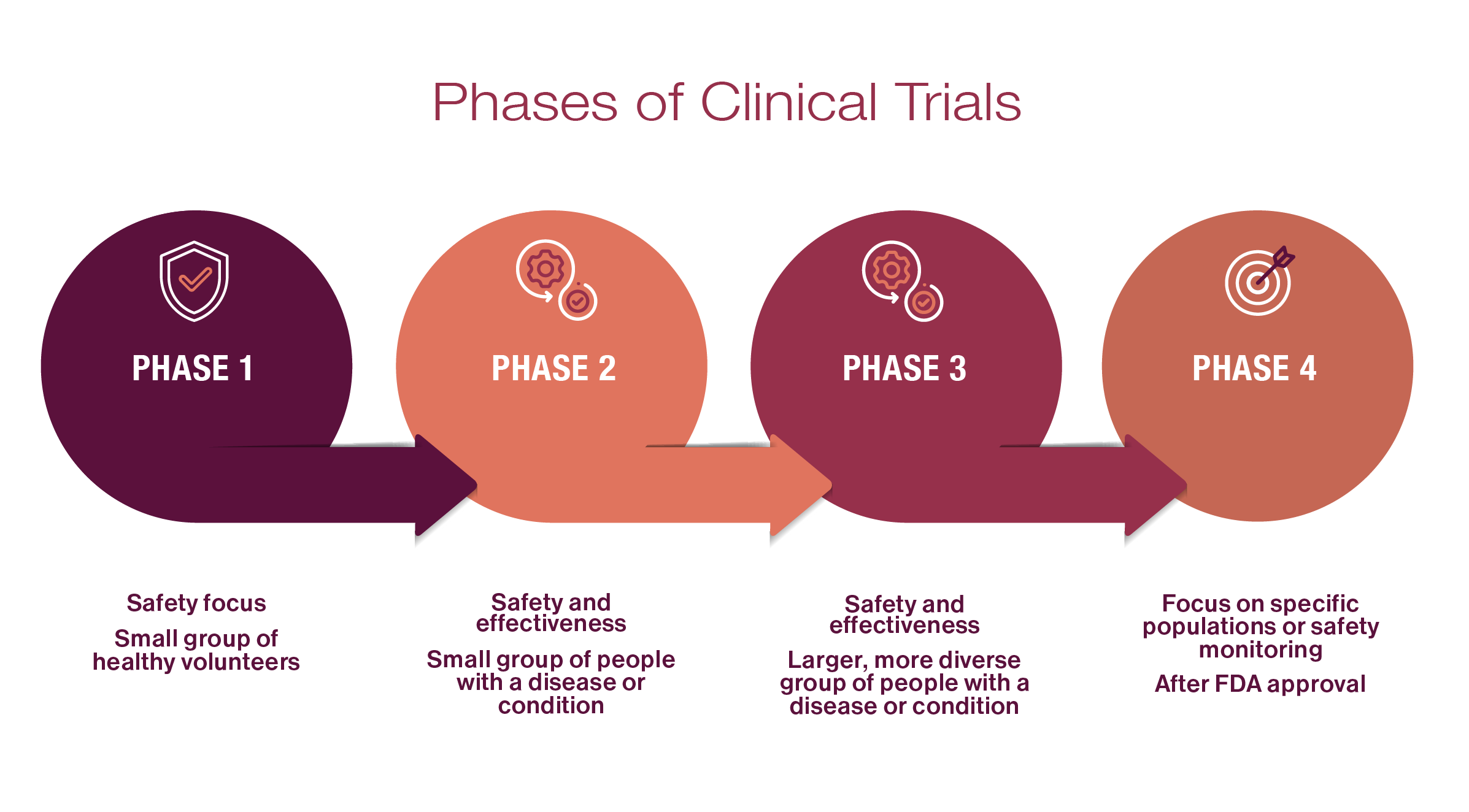
After experiments and evaluations are completed in a laboratory environment, these early trials enroll a small group of healthy volunteers to test the safety of new treatments and try to find the best dosage level.
With safety information from the first phase, a treatment then goes to a slightly larger second phase of trial, among a population of people with a certain bleeding disorder. This type of trial still monitors safety, but also studies how well the treatment works for that condition.
Once researchers collect the data from Phases 1 and 2 and see it may be useful, a sponsor may conduct a larger trial among more people of varying ages or at different stages of their disorder. In some cases, the treatment will be tested in combination with other treatments. If the treatment is found to work well with low rates of side effects, and the FDA agrees with the results, then the medicine can be approved to treat patients.
Once a treatment is approved and available, sometimes the trial sponsor will conduct more trials to continue to monitor how well it works in certain types of patients or to better understand its safety over time.
Read more about the clinical trial phases
What is a “protocol”?
A clinical trial protocol is like a blueprint for the trial. It’s a plan that describes all of the necessary steps and activities for the trial to meet its goal. The protocol includes topics like:
- The reason why the trial is being conducted
- The types of people who should participate and the number of participants needed
- The series of tests, procedures, or treatment and dosage to be followed by the research physicians
- The length of the trial, or how long treatment should continue to find out how well it works and whether it causes side effects
- And the information that will be collected about the participants
Trial controls protect participants
Today, clinical trials are very carefully organized and monitored to protect participants. Take time to learn the measures for your trial and ask any questions so you feel comfortable with the process.
Clinical trials have processes to make sure they are fair and have managed most risks. These include:
Institutional Review Boards (IRBs): You may see the term IRB in the protocol or in your consent forms. IRBs are committees made up of different types of experts including community members who are familiar with the clinical trial process. They review the trial protocol and all trial information before the trial begins to make sure the information is clear and complete in order to protect participants.
Data Safety Monitoring Board (DSMB): The DSMB is a committee of experts who are not involved in the trial or the center where it is being conducted. This group is brought together to review the findings throughout the trial and decide whether it should continue, change or stop. This could be for many reasons. Some trials are adjusted or stopped because the treatment under evaluation is working very well, is not working at all, or if there are unexpected side effects that need to be reviewed.
Personal data: The trial sponsor must also explain how your information is stored and who has access to it. This is covered in the confidentiality section of the informed consent process. Read more about the consent process to learn about the study process and ask questions of your trial team.
Why participate? Who should consider a trial
Joining a clinical trial is a big decision. For many people, taking part in a trial means they can gain access to the newest treatments that are not yet widely available, or to learn more about their own disorder through regular testing and monitoring. For others, they see this as a way to be a part of the latest research and help improve care for others in the future.
Why clinical trials are important |
|
“As a person with a bleeding disorder or a caregiver, you are vital to improving the care for people living with blood disorders and bleeding disorders today and for the future. Understanding research and your role as a participant in research is important to make progress toward cures for people with all bleeding disorders.” - Len Valentino |
Considerations for participation
Your Hemophilia Treatment Center (HTC) might have mentioned a clinical trial for people with your bleeding disorder, and you are curious if it might be a good idea for you.
It’s important to know that the trial protocol outlines certain requirements to join the trial. This might include your age, your condition (e.g., for hemophilia, if you have an inhibitor) and your treatment history. These are chosen based on what the trial sponsor wants to understand about the treatment and who may benefit from it.
You can find more about eligibility in the next module, which talks about the screening process.
Why I participate |
|
“Look at clinical trials as a possibility of improving your life and improving the lives of others. I know that it's made an incredible change in my life. By participating in these clinical trials, you're part of a legacy of people who are doing good work for those that follow behind you.” - Randy Curtis |
Diversity matters in clinical trials
This is a topic that is often discussed when designing clinical trials today. While many bleeding disorders affect people of a wide range of ethnic and racial backgrounds, most clinical trials do not reflect the diversity of our community. But it’s really important that we better understand how your background might affect the course of your disorder or how well a treatment works for you.
Most clinical trials now have resources to help individuals whose primary language is not English. Materials about these trials can be translated into many languages, and many trial facilities have trained professionals to help translate during conversations.
NOTE: It’s important to know that family members can be present to help explain the conversation, but medical information must be given by a trained medical professional.
When it might make sense not to participate
If you do not meet the eligibility criteria, that trial may not be right for you. But talk with your HTC, because there may be other trials to consider. Even if you are eligible, you may have personal reasons why taking part may not be a good fit for you right now.
Ask yourself:
- Am I ready to give enough time to this trial while I manage my other personal commitments? Can I make time in my daily routine for more visits and tests?
- Can I manage the expectations of me in the trial, such as recording my bleeds and reporting any symptoms?
- Do I have the right support systems in place to allow for travel, child care, work absence and other considerations?
- What do I hope to achieve by joining a trial?
- Do I understand and am I comfortable with the potential risks of participating in this trial?
- If you are a parent considering a trial for your child: Am I on the same page with my child about what’s involved, and can we make time for this in our lives?
Key Questions to Ask |
|
Highlights to Remember:
- The phases of clinical trials are designed to make sure a treatment is effective and its safety is very clear before it is available for prescription.
- Every trial has steps in the process to protect participants from risks and make sure they clearly understand the process and goals.
- Participating in clinical trials is very important, but there may be personal reasons to decide against it. Talk with your HTC and your family or seek resources like the NHF to decide whether a trial is right for you.

