CHAPTER 1 – Iron Deficiency with and without Anemia in the Inherited Bleeding Disorders Population
Experts: Glaivy Batsuli1; Magdalena D. Lewandowska2; Ming Y. Lim3; Amy Dunn4; Kelly Tickle5; Maureen K. Baldwin6; Rose Bender7; Jennifer Davila8; Jennifer Girard9; Brendan Hayes10; Amanda Jacobson-Kelly4; Peter Kouides 11; Michele P. Lambert12; Shilpa Nataraj1; Michelle Sholzberg13; Roona Sinha14, Hanny Al-Samkari15
Affiliations:
1Stanford University, Palo Alto, CA, USA
2Innovative Hematology/Indiana Hemophilia & Thrombosis Center, Indianapolis, IN, USA
3 Utah Center for Bleeding & Clotting Disorders at University of Utah Health, Salt Lake City, UT, USA
4Nationwide Children’s Hospital, The Ohio State University College of Medicine, Columbus, OH, USA
5Hemophilia of Georgia Center for Bleeding and Clotting Disorders at Children’s Healthcare of Atlanta, Atlanta, GA, USA
6Department of Obstetrics and Gynecology, Oregon Health & Science University, Portland, OR, USA
7Yale University School of Medicine, New Haven, CT, USA
8Hemophilia Treatment Center at Montefiore Einstein and Albert Einstein College of Medicine, Bronx, NY, USA
9University of Michigan, Ann Arbor, MI, USA
10National Bleeding Disorders Foundation, New York, NY, USA
11Mary M. Gooley Hemophilia Treatment Center, Rochester, NY, USA
12Children's Hospital of Philadelphia and The University of Pennsylvania, Philadelphia, PA, USA
13St. Michael’s Hospital, Li Ka Shing Knowledge Institute, University of Toronto, Toronto, Ontario, Canada
14University of Saskatchewan in Saskatoon, Saskatoon, SK, Canada
15Massachusetts General Hospital and Harvard Medical School, Boston, MA, USA
TOPIC 1: The Impact of Iron Deficiency
Background
An estimated 24% of the world’s population, or ~2 billion individuals, has anemia.1 Iron deficiency anemia is the most common cause of anemia and is associated with significant morbidity and mortality in high-risk populations.2 Iron deficiency without anemia is an under recognized and undertreated public health issue that affects males and females across the lifespan. Iron deficiency is especially prevalent among women and girls, affecting 40% of this population between the ages of 12-21 years when applying current standards for reference ranges of ferritin.3 Severe iron deficiency, with or without anemia, may be the first indication of an underlying inherited bleeding disorder that warrants additional evaluation, while individuals with an inherited bleeding disorder are at an increased risk of developing iron deficiency due to iron loss from bleeding.
Untreated iron deficiency ultimately evolves into iron deficiency anemia. Screening and correction of iron deficiency is critical, particularly during high-risk periods such as childhood, adolescence, and pregnancy. Diets consisting of low iron, excessive cow’s milk intake, or disordered eating can further increase one’s risk of iron deficiency. Because iron is a vital micronutrient for development, iron deficiency has been associated with motor and cognitive impairment and poor physical growth in children even in the absence of anemia.4,5 Iron deficiency can affect quality of life through reduced physical productivity and work capacity in adults.6,7 It has also been associated with decreased effectiveness of some childhood vaccines, placing children at risk for severe infections.8 It is important to recognize the wide spectrum of symptoms associated with iron deficiency (Table 1).9-12
| Table 1. Signs and Symptoms of Iron Deficiency with or without Anemia | |
| Breath holding spells in infants/toddlers | Increased bruising |
| Cheilitis (inflammation of the lips) | Insomnia |
| Chills | Pale mucous membranes |
| Cognitive decline in the elderly population | Pale skin |
| Cognitive impairment | Pagophagia (craving of ice) |
| Deformed or “spoon” shape to nails | Passing out (syncope) or almost passing out (near syncope) |
| Depressed mood or irritability | Pica (craving of non-food substances) |
| Dizziness | Restless legs syndrome |
| Dry skin or hair | Ringing in the ears |
| Exercise intolerance | Shortness of breath |
| Fatigue | Smooth or glossy appearance of the tongue |
| Hair loss | Tachycardia/palpitations |
| Headache | |
Recommendation 1.1
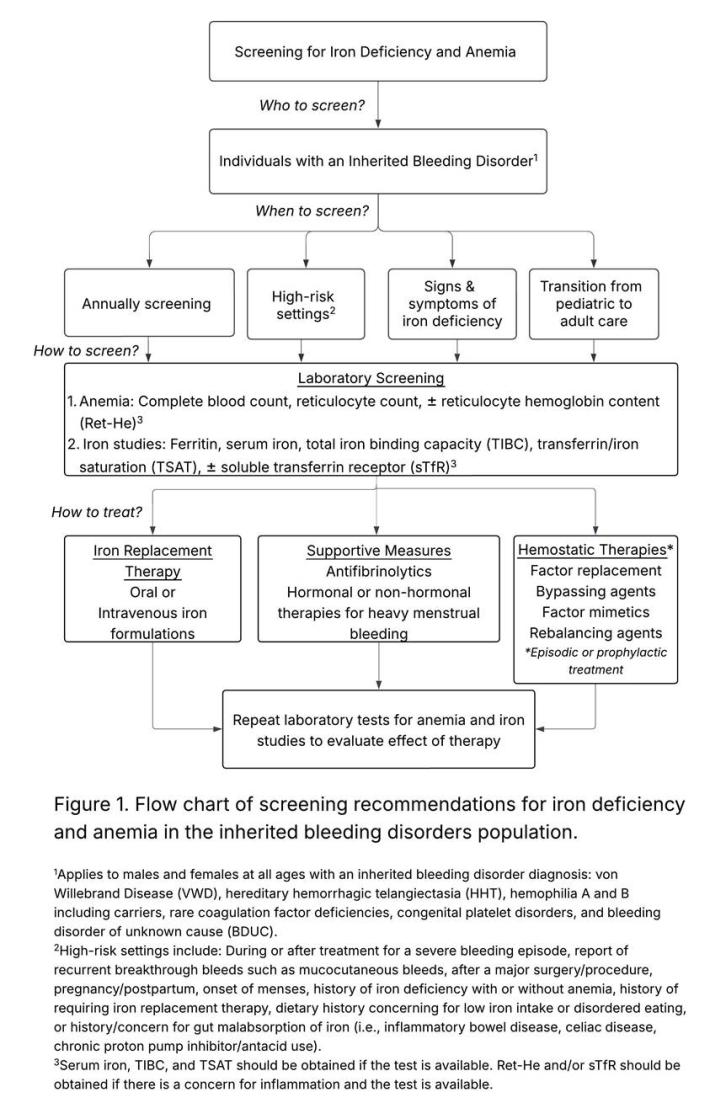
- MASAC recommends that all individuals with an inherited bleeding disorder should be routinely and periodically screened for iron deficiency and anemia irrespective of sex, age, or bleeding disorder severity (Figure 1).
- REMARK: All inherited bleeding disorder diagnoses should be considered when pursuing laboratory screening. Inherited bleeding disorders includes (in no particular order):
Hemophilia A (Factor VIII deficiency)
Hemophilia B (Factor IX deficiency)
Von Willebrand Disease (VWD)
Rare Coagulation Factor Deficiencies (e.g., Fibrinogen disorders, Factor II deficiency, Factor V deficiency, Combined Factor V and VIII deficiency, Factor VII deficiency, Factor X deficiency, Factor XI deficiency, and Factor XIII deficiency)
Congenital Platelet Disorders (e.g., Platelet storage pool disorders, Glanzmann Thrombasthenia, Bernard Soulier Syndrome, and other inherited platelet disorders)
Hemophilia A and B Carriers – defined as individuals with a factor VIII or factor IX gene mutation and normal factor VIII/IX activity levels13,14
Hereditary hemorrhagic telangiectasia (HHT)15
Bleeding Disorder of Unknown Cause (BDUC) – defined as a diagnosis of exclusion in an individual with a clinically significant bleeding tendency but normal hemostatic evaluation16
Recommendation 1.2
- MASAC recommends a multi-disciplinary approach to laboratory testing for iron deficiency and anemia including collaboration between primary care providers (PCPs) and specialists to ensure timely diagnosis, monitoring, and resolution of iron deficiency.
REMARK: This guidance is intended for clinical providers including PCPs, obstetricians/gynecologists, oncologists, and hematology-focused physicians and advanced practice providers (APPs) who provide care to individuals with inherited bleeding disorders within and outside of Hemophilia Treatment Centers (HTCs). These recommendations are also intended for individuals within the inherited bleeding disorders community to promote self-advocacy when concerns for iron deficiency are present.
TOPIC 2: Frequency of Laboratory Screening for Iron Deficiency and Anemia
Background
Due to the risk of chronic blood loss from bleeding events, the prevalence of iron deficiency and iron deficiency anemia in the inherited bleeding disorders population may be higher than the general population. Laboratory screening for iron deficiency varies widely among HTCs. Query of the American Thrombosis & Hemostasis Network (ATHN) dataset revealed that only ~4% of 10,527 females with bleeding disorders aged 13-40 years had laboratory screening for iron deficiency, but 72% of the screened individuals met criteria for iron deficiency.17 Heavy menstrual bleeding, a common bleeding symptom and cause of iron deficiency, affects up to 80% of women and girls with inherited bleeding disorders.18-20 A case-series of 14 male and female children with qualitative platelet disorders Glanzmann Thrombasthenia and Bernard Soulier Syndrome reported that while 79% of patients required iron replacement therapy for iron deficiency anemia, all the patients met the criteria for iron deficiency (ferritin <20 ng/mL) at least once.21 Lastly, a single-institution retrospective review of hemophilia B carriers and females with mild/moderate hemophilia B between 10-74 years old reported 93% of the 57 individuals had documented ferritin levels <50 ng/mL, consistent with iron deficiency.22 These studies suggest that the true prevalence of iron deficiency and anemia in the inherited bleeding disorders population is likely significantly underestimated. Despite the risk of bleeding related-complications, there are no existing recommendations or guidelines addressing screening for iron deficiency and anemia in this population.
Recommendation 2.1
- MASAC recommends that laboratory screening for iron deficiency and anemia should be performed annually in all individuals with an inherited bleeding disorder.
- REMARK: In the inherited bleeding disorders population where individuals are at a higher risk of chronic blood loss and experiencing complications from untreated iron deficiency, routine individual risk assessment in combination with laboratory screening for iron deficiency and anemia is strongly recommended on an annual basis.
- REMARK: Laboratory screening should be performed in all individuals irrespective of sex, age, or disease severity/phenotype.
Recommendation 2.2
- MASAC recommends additional laboratory screening for iron deficiency and anemia during high-risk settings (outside of routine annual screening) in all individuals with inherited bleeding disorders.
- REMARK: individuals with inherited bleeding disorders are at a higher risk for iron deficiency during high-risk settings.23,24 High-risk settings include:
During the treatment of a severe or prolonged bleeding episode*
Following hospitalization for the treatment of a bleeding episode*
History of recurrent breakthrough bleeds, particularly mucocutaneous bleeds (i.e., mouth, nose, or gastrointestinal bleeding)
Following a major surgery or report of excessive blood loss during a surgery/procedure
During pregnancy and postpartum (see Topic 3)
At the onset of menarche in adolescents
Report of heavy menstrual bleeding (defined as menstrual bleeding lasting more than 7 days, soaking through 1 or more pads/tampons in less than 2 hours, or passing large clots)
History of iron deficiency or iron deficiency anemia
History of requiring iron replacement therapy
History of Helicobacter pylori gastritis, inflammatory bowel disease, celiac disease, prior bariatric surgery, or other risk factors for iron malabsorption
Dietary history concerning low iron intake, limited access to iron-rich foods, disordered eating, avoidant/restrictive food intake disorder, etc.
Frequent or long-term use of proton pump inhibitors (PPIs), H2 blockers, and antacids
- REMARK: It is important to note that a normal or high ferritin during acute treatment for a bleed or hospitalization does not rule out iron deficiency. Ferritin is an acute phase reactant that can be falsely high or normal in the setting of inflammation. It is important to repeat laboratory testing at a later time point to confirm the presence or absence of iron deficiency in these situations.*
Recommendation 2.3
- MASAC recommends that laboratory screening for iron deficiency and anemia should be performed at any time if an individual with an inherited bleeding disorder presents with signs or symptoms suggestive of iron deficiency with or without anemia.
Recommendation 2.4
- MASAC recommends that laboratory screening for iron deficiency and anemia should be considered prior to the transition of adolescents and young adults with inherited bleeding disorders to adult providers if not previously performed.
TOPIC 3: Risk of Iron Deficiency during Pregnancy and Postpartum in the Inherited Bleeding Disorders Population
Background
Iron deficiency anemia, the most frequent cause of anemia during pregnancy, is a common but preventable complication of pregnancy affecting nearly 1 in every 5 pregnant persons in the United States.25 Each pregnancy requires approximately 1000 milligrams (mg) of total iron to support increased red blood cell (RBC) production, normal placental and fetal development, and anticipated blood loss at delivery.26 Persons with inherited bleeding disorders who become pregnant have increased risk of iron deficiency anemia, given the risk of lower baseline iron stores from blood loss. Furthermore, the diagnosis of physiologic versus pathologic cause of anemia in pregnancy poses a significant challenge on account of plasma volume expanding by 40-50% with erythrocyte mass expansion of only 15-25%. Iron deficiency anemia during pregnancy is associated with an increased risk of maternal death, postpartum depression, preterm birth, low birth weight, cesarean delivery, longer hospital stays after delivery, postpartum hemorrhage, and perinatal blood transfusion.27-30 Additionally, persons who are iron deficient during pregnancy are at higher risk of delivering infants who are iron deficient, which has been linked to neurological impairment.27,31,32 Postpartum iron deficiency anemia is underrecognized yet associated with fatigue, depression, and negative impact on cognition functioning, which can ultimately affect maternal-infant bonding and maternal care of the infant.33-37
Recommendation 3.1
- MASAC recommends that individuals with inherited bleeding disorders should be screened for iron deficiency and anemia at least once in the first trimester and again during the second or third trimester of pregnancy.
Recommendation 3.2
- MASAC recommends that individuals with inherited bleeding disorders should be screened for iron deficiency and anemia after delivery and at 6-12 weeks postpartum.
TOPIC 4: Recommended Tests for Laboratory Screening of Iron Deficiency and Anemia
Background
Laboratory screening should consider that signs and symptoms of iron deficiency can occur with or without anemia. Hemoglobin alone is not a good predictor of iron deficiency. Moreover, iron deficiency anemia does not typically present until severe iron deficiency has developed and even before microcytosis ensues. It is important to directly measure an individual’s iron status alongside tests for anemia to ensure accurate diagnosis and enable prompt treatment.
Most organizations and clinical laboratories differentiate the hemoglobin reference values used to define anemia based on biological sex in which females have a lower cut-off value of hemoglobin to be considered anemic. This is in part due to the effect of testosterone on RBC production resulting in a higher upper range of hemoglobin for males.38 The World Health Organization (WHO) defines anemia as a hemoglobin <12 g/dL in nonpregnant women and <13 g/dL in men.39 The WHO differentiates reference values for hemoglobin based on biologic sex at the onset of menarche, while the reference range recommended for hemoglobin levels prior to menarche and after menopause are equivalent between females and males. The hemoglobin reference ranges identified were primarily chosen based on mean hemoglobin values in various populations of women and men. This has led to a growing debate on whether sex-based reference ranges are appropriate, particularly in light of the lack of physiologic data demonstrating differences in oxygen uptake, oxygen carrying capacity, and oxygen delivery by sex.9,40 For instance, there is no sex-based difference in the hemoglobin threshold for preoperative or other indications for blood transfusion. In addition, mild anemia (hemoglobin <13 g/dL or hematocrit <36%) in the preoperative setting is associated with increased length of hospital stay and higher 30-day morbidity & mortality rates after major non-cardiac surgery irrespective of sex.41 As a result, sex-specific reference values for defining anemia may enable permissive iron deficiency among females, placing these individuals at increased harm from iron deficiency and anemia.41-43
For ferritin values, there are no physiologic reasons to support separate reference ranges between males and females. A recent Delphi consensus review concluded that the minimum ferritin for females should be 30 ng/mL.44 This document has been a helpful reference for caregivers and clinical providers to educate their laboratory colleagues, as many laboratories still use a female specific cutoff in the 10-13 ng/mL range compared to 30 ng/mL in males.44 However, more recent studies performed in healthy women between 18-50 years of age assessing ferritin levels that correspond with physiological upregulation of iron absorption and hepcidin levels identified a ferritin cutoff of <50 ng/mL as an indicator of iron deficiency.45,46 Parenteral iron administration in women with iron deficiency defined as a ferritin <50 ng/ml with a normal hemoglobin also resulted in improved quality of life.47
Recommendation 4.1
In individuals with inherited bleeding disorders, MASAC recommends that laboratory screening should include testing for anemia and iron studies (Table 2).
Table 2. Laboratory Tests for Anemia and Iron Studies | |
Tests for Anemia | Tests of Iron Status |
Complete blood count (CBC) | Ferritin |
Reticulocyte count | Serum Iron** |
Reticulocyte hemoglobin content (CHr or Ret-He)* | Total iron binding capacity (TIBC)** |
| Transferrin or iron saturation (TSAT)** |
| Soluble transferrin receptor (sTfR)* |
*If available and concerned about concurrent inflammation.
**Serum iron, TIBC, and TSAT are often performed in combination with ferritin. If not readily accessible, would add the serum iron, TIBC, and TSAT if concerned about concurrent inflammation or other causes of anemia.
- REMARK: Pregnant persons should be screened for iron deficiency and anemia with CBC, reticulocyte count, ferritin, and iron studies (iron, total iron binding capacity, transferrin saturation, and soluble transferrin receptor).
- REMARK: The reticulocyte hemoglobin content (CHr or Ret-He) provides an indirect measure of the functional iron available for new RBC production over the previous 3 to 4 days.48 A low Ret-He is seen in iron deficiency anemia and thalassemia. If there is concern for inflammation and clinically accessible, testing of the Ret-He should be considered. The Ret-He can help to distinguish iron deficiency anemia from anemia of inflammation in which Ret-He will be normal in anemia of inflammation. It is important to note that the Ret-He is not reliable in individuals with hemoglobinopathy (i.e., sickle cell disease, thalassemia, etc.) or hemoglobinopathy trait (i.e., sickle cell trait, thalassemia trait/thalassemia minor).
- REMARK: A low serum iron level alone is insufficient for the diagnosis of iron deficiency because a low serum iron level is also seen in anemia of inflammation. Additionally, serum iron levels can be affected by recent dietary iron intake. As such, a normal or high serum iron level does not rule out iron deficiency. If there is concern for falsely normal or high serum iron levels due to recent intake of an iron-rich meal, repeat testing can be performed when fasting.
- REMARK: The soluble transferrin receptor (sTfR) measures the receptor for iron that is bound to transferrin and is helpful in cases where anemia of inflammation versus iron deficiency anemia needs to be clarified.46 The sTfR is not an acute phase reactant (like ferritin) and values will be high in the setting of iron deficiency, but normal in anemia of chronic inflammation. It is also helpful for interpreting iron tests in the setting of class 2 or 3 obesity, but the sTfR values are age and sex dependent.
Recommendation 4.2
- MASAC recommends a hemoglobin level <13 g/dL as the cut-off for diagnosing anemia in males and females with inherited bleeding disorders at all ages.
- REMARK: This recommendation ensures that the diagnosis of iron deficiency anemia is properly identified in a high-risk population to address under-recognition of this bleeding-related complication. However, the clinical provider can take into consideration their knowledge of the individual’s personal history in combination with their clinical judgement when applying this recommendation.
- REMARK: Anemia in pregnant persons in the general population is defined as a hemoglobin <11 g/dL by the American College of Obstetrics and Gynecology.26
Recommendation 4.3
MASAC recommends a ferritin level of <50 ng/mL as the cut-off for diagnosing iron deficiency in males and females with inherited bleeding disorders at all ages.
- REMARK: Iron deficiency in pregnant persons with inherited bleeding disorders should be defined as ferritin <50 ng/mL.
- REMARK: A higher ferritin threshold is strongly recommended to prevent unrecognized or misdiagnosed iron deficiency.21,49,50
- REMARK: While there is evolving data on the optimal ferritin threshold in the general population, it is important to emphasize that there is very limited to no data on the optimal ferritin thresholds for individuals with inherited bleeding disorders. The clinical provider can take into consideration their knowledge of the individual’s personal history in combination with their clinical judgement when applying this recommendation.
- REMARK: It is important to note that ferritin is an acute phase reactant in which high ferritin or falsely normal values can be seen when there is inflammation, including autoimmunity, certain infections, cancer, or liver disease.
Recommendation 4.4
- MASAC recommends that the levels and pattern of the iron studies (i.e., ferritin, serum iron, TIBC, and TSAT) are evaluated collectively with the hemoglobin and reticulocyte count to confirm the diagnosis of iron deficiency and anemia (Table 3).
- REMARK: Ideally, iron studies should be performed when the individual is fasting. However, this is not always feasible. If there is concern for falsely normal iron studies due to recent intake of an iron-rich meal, repeat testing should be performed when fasting.
| Table 3. Summary of Reference Values for Iron Studies. | ||
Normal | Iron Deficiency | |
| Ferritin (ng/mL or mcg/L)9,45,46 | 50-300* | <50 |
| Serum iron (mcg/dL)51 | 60-150 | <60 |
| TIBC (mcg/dL)52 | 250-450 | >450 |
| Transferrin Saturation (%)51,53 | 20-60 | <20 |
| Reticulocyte hemoglobin content (pg)48,54 | >30 | ≤30 |
*The upper limit of ferritin values is 200 ng/mL for women and 300 ng/mL for men.
TOPIC 5: Diagnostic Considerations for Laboratory Screening
Background
Although individuals with an inherited bleeding disorder are at risk for blood loss from the underlying bleeding disorder, there are other potential sources of blood loss or diagnoses that may cause anemia, such as a malignant lesion, arteriovenous malformation, thalassemia trait or other RBC disorder, or anemia of inflammation. Other diagnostic considerations include the impact of anemia on coagulation laboratory tests. Clinical providers should be aware of several studies showing that anemia can “mask” the diagnosis of VWD by falsely elevating the factor VIII activity and von Willebrand factor (VWF) levels.55-57 Clinical providers should also be aware that anemia can cause platelet dysfunction, which will affect platelet function testing.58,59
Recommendation 5.1
- MASAC recommends that other causes of anemia are considered if 1) the laboratory pattern of iron indices is not consistent with iron deficiency, 2) the individual is not responsive to iron replacement therapy, or 3) there are additional concerning signs/symptoms for alternative causes of anemia when accounting for the individual’s medical and/or family history.
- REMARK: Thalassemia and other RBC disorders such as sideroblastic anemia should be considered in the evaluation of anemia. Individuals with thalassemia or thalassemia trait without concomitant iron deficiency typically have a high/normal serum iron, normal TIBC, normal TSAT, and normal/high ferritin in addition to an increased or normal RBC count. In contrast, iron deficiency is associated with a low serum iron, TSAT, ferritin, and RBC count. Additionally, the Mentzer index assessing the ratio of the RBC count to mean corpuscular volume (MCV) is a helpful calculation. An RBC count/MCV ratio <12 is typically consistent with thalassemia and a ratio >13 with iron deficiency.
Recommendation 5.2
- If another cause of anemia or specific site of blood loss is identified (i.e., gastrointestinal bleeding), MASAC recommends subspecialty consultation where applicable and further diagnostic evaluation per standards of care.
Recommendation 5.3
- MASAC recommends that diagnostic VWF panels and platelet function testing is performed in the non-anemic state, if possible. If anemia is present at the time of initial testing, repeat testing is recommended once the anemia is corrected.
TOPIC 6: Management of Iron Deficiency and Anemia
Background
The beneficial effects of treating iron deficiency anemia with oral and/or intravenous (IV) iron replacement therapy on symptom resolution and improving one’s health status are well documented. Treating iron deficiency without anemia is just as beneficial to individuals as treating iron deficiency anemia. Studies have shown that iron replacement therapy for iron deficiency without anemia can improve an individual’s cognition (particularly memory and verbal language), mood, and reduce fatigue.60,61 While an individual’s dietary intake of iron should be optimized, timely implementation of iron replacement therapies with oral or intravenous iron formulations coupled with response monitoring is critical.
Recommendation 6.1
- MASAC recommends that iron replacement with oral or IV iron formulations should be implemented in a timely manner once iron deficiency with or without anemia has been identified.
- REMARK: The use of IV iron replacement should be strongly considered in individuals with an inherited bleeding disorder with chronic blood loss that is challenging to manage with oral iron, oral iron intolerance/allergy, persistent severe symptomatic anemia (hemoglobin <10 g/dL), persistent fatigue despite a ferritin between 30-50 ng/ml, or the need for rapid correction of iron deficiency.10,47,62
- REMARK: The use of certain IV iron replacement formulations, including ferric carboxymaltose and iron polymaltose, have very high rates of treatment-emergent hypophosphatemia (upwards of 50-75% in persons receiving ferric carboxymaltose for a single course of iron repletion).63-66 This hypophosphatemia occurs due to an acquired renal lesion from these products, often lasts for weeks to months, may be severe, and is strongly resistant to oral or IV phosphate repletion. Given that people with inherited bleeding disorders, particularly those with chronic ongoing bleeding, will often require repeat courses of IV iron repletion, ferric carboxymaltose and iron polymaltose should be avoided when at all possible. Repeat infusions of these formulations may result in hypophosphatemic osteomalacia and insufficiency fractures as well as kidney stones and fatigue.67 Formulations allowing for full-dose iron repletion (1000 mg elemental iron or more) with a negligible risk of treatment-emergent hypophosphatemia are considered optimal for this population. This includes iron sucrose (primarily available for use in the pediatric population), low molecular weight iron dextran, ferumoxytol, and ferric derisomaltose.
Recommendation 6.2
- During second and third trimester of pregnancy, MASAC recommends iron replacement with IV iron formulations over oral iron formulations for rapid correction of iron deficiency in individuals with inherited bleeding disorders.
- REMARK: There is very limited data on the safety of IV iron infusions in the first trimester of pregnancy. IV iron infusions are generally deferred until the second trimester when possible. However, the decision of whether to administer intravenous iron should be individualized and made utilizing shared decision making with the pregnant individual, hematologist, and obstetrician/gynecologist.
- REMARK: During the second and third trimesters of pregnancy, rapid correction of iron deficiency is important to optimize the health of the mother and baby. This is more easily accomplished with IV iron replacement. There have been several studies that show IV iron infusions significantly increase hemoglobin, ferritin, and iron levels in pregnant individuals in the general population with iron deficiency anemia (IDA) compared to oral iron taken during pregnancy.68,69 One study of pregnant individuals with IDA found that IV iron resulted in a reduced need for blood transfusions compared to oral iron (0% vs 15%).70 Rapid correction of IDA is especially important during the second and third trimester of pregnancy, where oral iron is much less effective that IV iron at improving the baby’s cord blood ferritin level at birth.71
- REMARK: In the postpartum period, IV iron replacement is associated with higher hemoglobin and ferritin levels up to 6 weeks postpartum compared to oral iron replacement.72
Recommendation 6.3
MASAC recommends that the response to treatment should be monitored until resolution of iron deficiency and anemia, with repeated treatment as necessary.
- REMARK: The effect of iron replacement therapy should be evaluated within 1-3 months of initiation at minimum and continued until iron deficiency correction. The individual’s clinical situation and tolerance of iron replacement therapy should be considered when determining the follow up interval and frequency of monitoring.
Recommendation 6.4
MASAC recommends assessment for the need of supportive measures or hemostatic therapies (either episodically or prophylactically) to prevent chronic blood loss and iron deficiency in the inherited bleeding disorders population.
- REMARK: In inherited bleeding disorders, it is important to address the source of bleeding symptoms that may be contributing to iron deficiency through supportive measures (i.e., antifibrinolytic agents for mucosal bleeds; topical measures or surgical interventions for nosebleed prevention) and/or hemostatic therapies (i.e., factor replacement therapies, bypassing agents, factor mimetics, or rebalancing agents) depending on the bleeding severity and underlying diagnosis.49
- REMARK: For individuals with heavy menstrual bleeding, supportive therapies such as hormone-based therapies or non-hormone based therapies should be strongly considered to minimize bleeding at least until iron repletion.
- REMARK: In some circumstances, chronic blood loss resulting in iron deficiency may justify the need to initiate prophylactic hemostatic therapies to optimize disease management, prevent further blood loss, and enable resolution of iron deficiency. Initiation of prophylactic hemostatic therapy should be discussed with clinical providers with expertise in the management of inherited bleeding disorders and preferably located at HTCs.
Recommendation 6.5
- MASAC recommends addressing any other risk factors (i.e., long-term PPI use, poor dietary intake of iron, excessive cow’s milk intake in children, etc.) that may be contributing to iron deficiency and anemia when applicable.
- REMARK: A table identifying food sources of iron is provided in the Supplemental Information.
Future Research Needs
- The purpose of the MASAC Iron Deficiency Screening Taskforce was to generate initial recommendations for clinical providers and Lived Experienced Experts (LEE) on laboratory screening for iron deficiency in the inherited bleeding disorders population across the age spectrum and regardless of sex. Individuals with inherited bleeding disorders are at high risk of developing iron deficiency resulting from bleeding episodes. However, the prevalence of iron deficiency in the inherited bleeding disorder population is not fully understood owing to the limited research dedicated to this specific topic area.
- Further research is necessary to understand the impact of iron deficiency across all inherited bleeding disorders and to help define the optimal testing frequency, laboratory cut-off values, and ideal treatment approaches.
- Further investigation on the impact of iron deficiency and anemia on coagulation laboratory testing is also recommended.
- The availability of clinical trials and real-world data affects the development of guidelines or consensus statements on the approach to or frequency of screening for iron deficiency and iron deficiency anemia in this population. Increased research dedicated to understanding the prevalence and impact of iron deficiency in the inherited bleeding disorders population across the lifespan is warranted.
- Several other organizations are developing recommendations and guidelines for diagnosing and management or iron deficiency with and without anemia that will help to further increase awareness of this issue.
REFERENCES
1. GBD 2021 Anaemia Collaborators. Prevalence, years lived with disability, and trends in anaemia burden by severity and cause, 1990-2021: findings from the Global Burden of Disease Study 2021. Lancet Haematol. 2023;10(9):e713-e734.
2. Jefferds MED, Mei Z, Addo Y, et al. Iron Deficiency in the United States: Limitations in Guidelines, Data, and Monitoring of Disparities. Am J Public Health. 2022;112(S8):S826-s835.
3. Weyand AC, Chaitoff A, Freed GL, Sholzberg M, Choi SW, McGann PT. Prevalence of Iron Deficiency and Iron-Deficiency Anemia in US Females Aged 12-21 Years, 2003-2020. JAMA. 2023;329(24):2191-2193.
4. Baker RD, Greer FR. Diagnosis and prevention of iron deficiency and iron-deficiency anemia in infants and young children (0-3 years of age). Pediatrics. 2010;126(5):1040-1050.
5. Halterman JS, Kaczorowski JM, Aligne CA, Auinger P, Szilagyi PG. Iron deficiency and cognitive achievement among school-aged children and adolescents in the United States. Pediatrics. 2001;107(6):1381-1386.
6. Pasricha SR, Low M, Thompson J, Farrell A, De-Regil LM. Iron supplementation benefits physical performance in women of reproductive age: a systematic review and meta-analysis. J Nutr. 2014;144(6):906-914.
7. Li R, Chen X, Yan H, Deurenberg P, Garby L, Hautvast JG. Functional consequences of iron supplementation in iron-deficient female cotton mill workers in Beijing, China. Am J Clin Nutr. 1994;59(4):908-913.
8. Drakesmith H, Pasricha SR, Cabantchik I, et al. Vaccine efficacy and iron deficiency: an intertwined pair? Lancet Haematol. 2021;8(9):e666-e669.
9. Martens K, DeLoughery TG. Sex, lies, and iron deficiency: a call to change ferritin reference ranges. Hematology Am Soc Hematol Educ Program. 2023;2023(1):617-621.
10. Iolascon A, Andolfo I, Russo R, et al. Recommendations for diagnosis, treatment, and prevention of iron deficiency and iron deficiency anemia. Hemasphere. 2024;8(7):e108.
11. Pasricha SR, Tye-Din J, Muckenthaler MU, Swinkels DW. Iron deficiency. Lancet. 2021;397(10270):233-248.
12. Jain R, Omanakuttan D, Singh A, Jajoo M. Effect of iron supplementation in children with breath holding spells. J Paediatr Child Health. 2017;53(8):749-753.
13. van Galen KPM, d'Oiron R, James P, et al. A new hemophilia carrier nomenclature to define hemophilia in women and girls: Communication from the SSC of the ISTH. J Thromb Haemost. 2021;19(8):1883-1887.
14. Makris M, Oldenburg J, Mauser-Bunschoten EP, Peerlinck K, Castaman G, Fijnvandraat K. The definition, diagnosis and management of mild hemophilia A: communication from the SSC of the ISTH. J Thromb Haemost. 2018;16(12):2530-2533.
15. Faughnan ME, Mager JJ, Hetts SW, et al. Second International Guidelines for the Diagnosis and Management of Hereditary Hemorrhagic Telangiectasia. Ann Intern Med. 2020;173(12):989-1001.
16. Baker RI, Choi P, Curry N, et al. Standardization of definition and management for bleeding disorder of unknown cause: communication from the SSC of the ISTH. J Thromb Haemost. 2024;22(7):2059-2070.
17. McCormick M, Hu J, Chandler M, Manuel M, Chrisentery-Singleton T, Ragni MV. The Iron Ladies: Prevalence and Risk Factors of Iron Deficiency in Females With Bleeding Disorders. Haemophilia. 2025.
18. Kadir RA, Economides DL, Sabin CA, Pollard D, Lee CA. Assessment of menstrual blood loss and gynaecological problems in patients with inherited bleeding disorders. Haemophilia. 1999;5(1):40-48.
19. Maas D, Saes JL, Blijlevens NMA, et al. High prevalence of heavy menstrual bleeding in women with rare bleeding disorders in the Netherlands: retrospective data from the RBiN study. J Thromb Haemost. 2023;21(10):2726-2734.
20. Winikoff R, Scully MF, Robinson KS. Women and inherited bleeding disorders - A review with a focus on key challenges for 2019. Transfus Apher Sci. 2019;58(5):613-622.
21. Lee A, Maier CL, Batsuli G. Iron deficiency anemia and bleeding management in pediatric patients with Bernard-Soulier syndrome and Glanzmann Thrombasthenia: A single-institution analysis. Haemophilia. 2022;28(4):633-641.
22. Lu Z, Machin NC. Assessment of Bleeding Severity and Prevalence of Iron Deficiency Among Hemophilia B Carriers By Factor IX Activity Levels. Blood. 2024;144(Supplement 1):2586-2586.
23. Eiduson R, Heeney MM, Kao PC, London WB, Fleming MD, Shrier LA. Prevalence and Predictors of Iron Deficiency in Adolescent and Young Adult Outpatients: Implications for Screening. Clin Pediatr (Phila). 2022;61(1):66-75.
24. Srivastava A, Santagostino E, Dougall A, et al. WFH Guidelines for the Management of Hemophilia, 3rd edition. Haemophilia. 2020;26 Suppl 6:1-158.
25. Mei Z, Cogswell ME, Looker AC, et al. Assessment of iron status in US pregnant women from the National Health and Nutrition Examination Survey (NHANES), 1999-2006. Am J Clin Nutr. 2011;93(6):1312-1320.
26. Anemia in Pregnancy: ACOG Practice Bulletin, Number 233. Obstet Gynecol. 2021;138(2):e55-e64.
27. Daru J, Zamora J, Fernández-Félix BM, et al. Risk of maternal mortality in women with severe anaemia during pregnancy and post partum: a multilevel analysis. Lancet Glob Health. 2018;6(5):e548-e554.
28. Smith C, Teng F, Branch E, Chu S, Joseph KS. Maternal and Perinatal Morbidity and Mortality Associated With Anemia in Pregnancy. Obstet Gynecol. 2019;134(6):1234-1244.
29. Klebanoff MA, Shiono PH, Selby JV, Trachtenberg AI, Graubard BI. Anemia and spontaneous preterm birth. Am J Obstet Gynecol. 1991;164(1 Pt 1):59-63.
30. Elmore C, Ellis J. Screening, Treatment, and Monitoring of Iron Deficiency Anemia in Pregnancy and Postpartum. J Midwifery Womens Health. 2022;67(3):321-331.
31. Drukker L, Hants Y, Farkash R, Ruchlemer R, Samueloff A, Grisaru-Granovsky S. Iron deficiency anemia at admission for labor and delivery is associated with an increased risk for Cesarean section and adverse maternal and neonatal outcomes. Transfusion (Paris). 2015;55(12):2799-2806.
32. Tunkyi K, Moodley J. Anemia and pregnancy outcomes: a longitudinal study. J Matern Fetal Neonatal Med. 2018;31(19):2594-2598.
33. Lee KA, Zaffke ME. Longitudinal changes in fatigue and energy during pregnancy and the postpartum period. J Obstet Gynecol Neonatal Nurs. 1999;28(2):183-191.
34. Corwin EJ, Murray-Kolb LE, Beard JL. Low hemoglobin level is a risk factor for postpartum depression. J Nutr. 2003;133(12):4139-4142.
35. Beard JL, Hendricks MK, Perez EM, et al. Maternal iron deficiency anemia affects postpartum emotions and cognition. J Nutr. 2005;135(2):267-272.
36. Congdon EL, Westerlund A, Algarin CR, et al. Iron deficiency in infancy is associated with altered neural correlates of recognition memory at 10 years. J Pediatr. 2012;160(6):1027-1033.
37. Geng F, Mai X, Zhan J, et al. Impact of Fetal-Neonatal Iron Deficiency on Recognition Memory at 2 Months of Age. J Pediatr. 2015;167(6):1226-1232.
38. Hero M, Wickman S, Hanhijärvi R, Siimes MA, Dunkel L. Pubertal upregulation of erythropoiesis in boys is determined primarily by androgen. J Pediatr. 2005;146(2):245-252.
39. WHO Guidelines Approved by the Guidelines Review Committee. WHO guideline on use of ferritin concentrations to assess iron status in individuals and populations. Geneva: World Health Organization © 2020.; 2020.
40. Solleiro Pons M, Bernert L, Hume E, et al. No sex differences in oxygen uptake or extraction kinetics in the moderate or heavy exercise intensity domains. J Appl Physiol (1985). 2024;136(3):472-481.
41. Dugan C, MacLean B, Cabolis K, et al. The misogyny of iron deficiency. Anaesthesia. 2021;76 Suppl 4:56-62.
42. Weyand AC, McGann PT, Sholzberg M. Sex specific definitions of anaemia contribute to health inequity and sociomedical injustice. Lancet Haematol. 2022;9(1):e6-e8.
43. Musallam KM, Tamim HM, Richards T, et al. Preoperative anaemia and postoperative outcomes in non-cardiac surgery: a retrospective cohort study. Lancet. 2011;378(9800):1396-1407.
44. Naveed K, Goldberg N, Shore E, et al. Defining ferritin clinical decision limits to improve diagnosis and treatment of iron deficiency: A modified Delphi study. Int J Lab Hematol. 2023;45(3):377-386.
45. Galetti V, Stoffel NU, Sieber C, Zeder C, Moretti D, Zimmermann MB. Threshold ferritin and hepcidin concentrations indicating early iron deficiency in young women based on upregulation of iron absorption. EClinicalMedicine. 2021;39:101052.
46. Tarancon-Diez L, Genebat M, Roman-Enry M, et al. Threshold Ferritin Concentrations Reflecting Early Iron Deficiency Based on Hepcidin and Soluble Transferrin Receptor Serum Levels in Patients with Absolute Iron Deficiency. Nutrients. 2022;14(22).
47. Krayenbuehl PA, Battegay E, Breymann C, Furrer J, Schulthess G. Intravenous iron for the treatment of fatigue in nonanemic, premenopausal women with low serum ferritin concentration. Blood. 2011;118(12):3222-3227.
48. Chinudomwong P, Binyasing A, Trongsakul R, Paisooksantivatana K. Diagnostic performance of reticulocyte hemoglobin equivalent in assessing the iron status. J Clin Lab Anal. 2020;34(6):e23225.
49. VanderMeulen H, Sholzberg M. Iron deficiency and anemia in patients with inherited bleeding disorders. Transfus Apher Sci. 2018;57(6):735-738.
50. Abe K, Dupervil B, O'Brien SH, et al. Higher rates of bleeding and use of treatment products among young boys compared to girls with von Willebrand disease. Am J Hematol. 2020;95(1):10-17.
51. Koerper MA, Dallman PR. Serum iron concentration and transferrin saturation in the diagnosis of iron deficiency in children: normal developmental changes. J Pediatr. 1977;91(6):870-874.
52. Faruqi A, Zubair M, Mukkamalla SKR. Iron-Binding Capacity. StatPearls: StatPearls Publishing; 2024.
53. Fertrin KY. Diagnosis and management of iron deficiency in chronic inflammatory conditions (CIC): is too little iron making your patient sick? Hematology Am Soc Hematol Educ Program. 2020;2020(1):478-486.
54. Mast AE, Blinder MA, Dietzen DJ. Reticulocyte hemoglobin content. Am J Hematol. 2008;83(4):307-310.
55. Simoneau J, Tay C, Wheeler A, et al. Association between hemoglobin values and VWF assays: a multicenter investigation. Blood Adv. 2024;8(5):1152-1154.
56. Brown MC, White MH, Friedberg R, et al. Elevated von Willebrand factor levels during heavy menstrual bleeding episodes limit the diagnostic utility for von Willebrand disease. Res Pract Thromb Haemost. 2021;5(4):e12513.
57. Carter-Febres M, Fenchel M, Pomales J, Tarango C, Mullins ES. Hemoglobin concentration and body mass index are determinants of plasma von Willebrand factor and factor VIII levels. Thromb Res. 2024;240:109061.
58. Akay OM, Akin E, Mutlu FS, Gulbas Z. Effect of Iron Therapy on Platelet Function among Iron-Deficient Women with Unexplained Menorrhagia. Pathophysiol Haemost Thromb. 2008;36(2):80-83.
59. Yıldırım ZK, Orhan MF, Büyükavcı M. Platelet function alterations and their relation to P-selectin (CD62P) expression in children with iron deficiency anemia. Blood Coagul Fibrinolysis. 2011;22(2):98-101.
60. Bruner AB, Joffe A, Duggan AK, Casella JF, Brandt J. Randomised study of cognitive effects of iron supplementation in non-anaemic iron-deficient adolescent girls. Lancet. 1996;348(9033):992-996.
61. Patterson AJ, Brown WJ, Roberts DC. Dietary and supplement treatment of iron deficiency results in improvements in general health and fatigue in Australian women of childbearing age. J Am Coll Nutr. 2001;20(4):337-342.
62. Camaschella C. Iron deficiency. Blood. 2019;133(1):30-39.
63. Auerbach M, Wolf M. Intravenous Iron Associated Hypophosphatemia: Much More Than a Laboratory Curiosity. Am J Hematol. 2025;100(5):752-754.
64. Wolf M, Rubin J, Achebe M, et al. Effects of Iron Isomaltoside vs Ferric Carboxymaltose on Hypophosphatemia in Iron-Deficiency Anemia: Two Randomized Clinical Trials. JAMA. 2020;323(5):432-443.
65. Magagnoli J, Knopf K, Hrushesky WJ, Carson KR, Bennett CL. Ferric Carboxymaltose (FCM)-Associated Hypophosphatemia (HPP): A Systematic Review. Am J Hematol. 2025;100(5):840-846.
66. Vilaca T, Velmurugan N, Smith C, Abrahamsen B, Eastell R. Osteomalacia as a Complication of Intravenous Iron Infusion: A Systematic Review of Case Reports. J Bone Miner Res. 2022;37(6):1188-1199.
67. Ifie E, Oyibo SO, Joshi H, Akintade O. Symptomatic hypophosphataemia after intravenous iron therapy: an underrated adverse reaction. Endocrinol Diabetes Metab Case Rep. 2019;2019(1).
68. Awomolo AM, McWhirter A, Sadler LC, Coppola LM, Hill MG. Intravenous infusions of ferumoxytol compared to oral ferrous sulfate for the treatment of anemia in pregnancy: a randomized controlled trial. Am J Obstet Gynecol MFM. 2023;5(9):101064.
69. Hansen R, Sommer VM, Pinborg A, et al. Intravenous ferric derisomaltose versus oral iron for persistent iron deficient pregnant women: a randomised controlled trial. Arch Gynecol Obstet. 2023;308(4):1165-1173.
70. Lewkowitz AK, Stout MJ, Cooke E, et al. Intravenous versus Oral Iron for Iron-Deficiency Anemia in Pregnancy (IVIDA): A Randomized Controlled Trial. Am J Perinatol. 2022;39(8):808-815.
71. Awomolo AM, McWhirter A, Sadler LC, Coppola LM, Hill MG. Neonatal outcomes from a randomized controlled trial of maternal treatment of iron deficiency anemia with intravenous ferumoxytol vs oral ferrous sulfate. Am J Obstet Gynecol MFM. 2023;5(9):101063.
72. Sultan P, Bampoe S, Shah R, et al. Oral vs intravenous iron therapy for postpartum anemia: a systematic review and meta-analysis. Am J Obstet Gynecol. 2019;221(1):19-29.e13.
Keywords: Anemia, bleeding, heavy menstrual bleeding, inherited bleeding disorders, iron deficiency, pregnancy
List of Abbreviations
Abbreviation | Full Term |
APPs | Advanced Practice Providers |
ATHN | American Thrombosis and Hemostasis Network |
CBC | Complete blood count |
CHr | Reticulocyte hemoglobin content |
HTC | Hemophilia Treatment Center |
IDA | Iron deficiency anemia |
IV | Intravenous |
LEE | Lived Experienced Expert |
MCV | Mean corpuscular volume |
Mg | Milligrams |
PCPs | Primary Care Provider |
PPIs | Proton pump inhibitors |
RBC | Red blood cell |
Ret-He | Reticulocyte hemoglobin content |
sTfR | Soluble transferrin receptor |
TIBC | Total iron binding capacity |
TSAT | Transferrin or iron saturation |
VWD | Von Willebrand Disease |
VWF | Von Willebrand factor |
WHO | World Health Organization |
Supplemental Table S1. Food Sources of Iron. | ||
Food | Serving Size | Iron Content (mg) |
Meats and Animal Proteins | ||
Oysters | 3 oysters | 6.9 |
Beef liver | 3 ounces | 5.8 |
Mussels | 3 ounces | 5.7 |
Duck (breast) | 3 ounces | 3.8 |
Bison | 3 ounces | 2.9 |
Lean sirloin | 3 ounces | 2.9 |
Duck egg | 1 egg | 2.7 |
Beef | 3 ounces | 2.7 |
Sardines (canned) | 3 ounces | 2.5 |
Crab | 3 ounces | 2.5 |
Clams | 3 ounces | 2.4 |
Lamb | 3 ounces | 2.0 |
Turkey leg | 3 ounces | 2.0 |
Lean ground beef | 3 ounces | 1.8 |
Shrimp | 3 ounces | 1.8 |
Organ meats (various) | 3 ounces | 1.8-19 |
Game meats (various) | 3 ounces | 1.8-8.5 |
Skinless dark chicken | 3 ounces | 1.1 |
Skinless light chicken | 3 ounces | 0.9 |
Lean pork | 3 ounces | 0.9 |
Canned salmon (with bones) | 3 ounces | 0.7 |
Egg yolk | 1 large yolk | 0.6 |
Grains | ||
Hot wheat cereal (fortified) | 1 cup | 12.8 |
Bagel (enriched) | 1 medium sized | 6.4 |
Cereal | 1 cup | 4.5-18 |
Bran | 1/2 cup | 3.0 |
Bulgur | 1 cup | 1.7 |
Rice | 1/2 cup | 1.4 |
Pretzels | 1 ounce | 1.2 |
Whole wheat bread | 1 slice | 0.9 |
White bread | 1 slice | 0.8 |
Seeds and Nuts | ||
Lentils (cooked) | 1 cup | 6.6 |
Chickpeas (garbanzo beans, cooked) | 1 cup | 4.7 |
Pumpkin seeds | 1 ounce | 4.2 |
Soybean nuts | 1/2 cup | 3.5 |
Tahini | 2 tablespoons | 2.7 |
Cashews | 1/4 cup | 2.1 |
Sesame seeds | 1/2 ounce | 2.1 |
Almonds | 1/4 cup | 1.3 |
Sunflower seeds | 1/4 cup | 1.2 |
Fruits, Vegetables, and Starches | ||
Boiled spinach | 1 cup | 6.4 |
Artichokes (cooked) | 1 cup | 5.1 |
Swiss chard | 1 cup | 4.0 |
Chrysanthemum leaves | 1 cup | 3.7 |
Stewed tomatoes (canned) | 1 cup | 3.4 |
Turnip greens | 1 cup | 3.2 |
Asparagus (raw) | 1 cup | 2.9 |
Beets | 1 cup | 2.9 |
Beet greens | 1 cup | 2.7 |
Mushrooms (cooked) | 1 cup | 2.7 |
Peas | 1 cup | 2.5 |
Green beans | 1 cup | 2.5 |
Prune juice | 3/4 cup (6 ounces) | 2.3 |
Mustard spinach (raw) | 1 cup | 2.3 |
Collard greens | 1 cup | 2.2 |
Acorn squash | 1 cup | 1.9 |
Dandelion greens | 1 cup | 1.9 |
Leeks | 1 cup | 1.9 |
Baked potato with skin | 1 medium | 1.9 |
Bok choy | 1 cup | 1.8 |
Sweet potato | 1 cup | 1.8 |
Watermelon | 1/8 medium | 1.4 |
Kale | 1 cup | 1.2 |
Raisins | 1/2 cup | 1.0-1.6 |
Dried prunes | 1/4 cup (5-6 prunes) | 1.0 |
Dried apricots | 3 pieces | 0.5 |
Zucchini | 1/2 cup | 0.3 |
Cranberry juice | 3/4 cup (6 ounces) | 0.3 |
Grapes | 1/2 cup (about 16 grapes) | 0.1 |
Proteins | ||
Lima beans | 1 cup | 4.9 |
Soybeans | 1/2 cup | 4.4 |
Black beans | 1 cup | 3.6 |
Pinto beans | 1 cup | 3.6 |
White beans | 1/2 cup | 3.4 |
Red kidney beans | 1/2 cup | 2.6 |
Navy beans | 1/2 cup | 2.2 |
Black-eyed peas | 1/2 cup | 2.2 |
Yellow beans | 1/2 cup | 2.2 |
Cranberry (roman) beans | 1/2 cup | 1.9 |
Peanut butter | 2 tablespoons | 0.6 |
Other | ||
Blackstrap molasses | 2 tablespoons | 7.2 |
Tofu | 4 ounces | 6.4 |
Tempeh | 1 cup | 4.5 |
Quinoa | 1 cup | 2.8 |
Poi (taro root) | 1 cup | 2.1 |
Veggie burger | 1 patty | 1.4 |
Vitamin C helps to improve the absorption of iron. Good sources of vitamin C including citrus fruit (i.e., orange juice), tomatoes, peppers, and broccoli.
*This list was adapted from Innovative Hematology Indiana Hemophilia & Thrombosis Center Iron Rich Foods Educational Handout, U.S. Department of Agriculture’s Agricultural Research Service. Food Data Central, 2019 (fdc.nal.usda.gov), and DeLoughery TG. Iron Deficiency Anemia. Med Clin North Am. 2017;101(2):319-332.

